Abstract
Key message
Corymbia citriodora subsp. variegata altered the expression of secondary metabolites in response to Paropsis atomaria larval feeding and mechanical wounding but did not alter leaf toughness and leaf trichomes.
Abstract
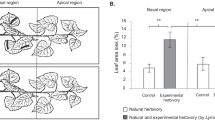
Plants have evolved strategies against herbivore pressure, relying on constitutive or induced traits that create physical and chemical barriers, which may influence herbivore performance. We evaluated the physical and chemical responses of Corymbia citriodora subsp. variegata, an important hardwood plantation taxon, to feedings by Paropsis atomaria larvae, a pest that causes severe defoliation in young trees. This was undertaken to obtain insights into plant–herbivore interactions, aiming to identify parameters that may improve plant fitness and/or protection that may benefit pest management in forestry plantations. Seedlings of C. citriodora subsp. variegata were submitted to the following treatments: no damage, mechanical wounding, and P. atomaria larval feeding damage. Foliar samples were collected during and after treatment at day 15 and 22, respectively, from damaged and undamaged leaves to detect: local, systemic, or delayed induced responses. Leaf samples were analysed to determine whether there were induced physical (leaf toughness, trichome density) or chemical (secondary metabolite profiles) responses to damage. No physical response in any of the treatments was evident in C. citriodora subsp. variegata foliage at day 15 or 22. Systemic chemical responses were observed for the larval feeding and mechanical treatments, with, a delayed response evident in the larval feeding treatment only. The proportion of long chain hydrocarbons were reduced in these treatments relative to the control, whereas the proportion of monounsaturated hydrocarbons and monoterpenes increased. When analysed across treatments, larval mortality was negatively correlated with long chain hydrocarbons and positively correlated with monounsaturated hydrocarbons. These findings suggest that CCV systemic chemical responses induced by both larval feeding and mechanical damage but only larval feeding induced delayed response, suggesting that CCV responses to herbivory may be associated with plant defence strategies.






Similar content being viewed by others
References
Abasolo M, Lee DJ, Brooks L, Raymond C, Shepherd M (2014) Genetic control of flowering in spotted gum, Corymbia citriodora subsp. variegata and C. maculata. Aust J Bot 62:22–35. https://doi.org/10.1071/bt13223
Agrawal AA (1999) Induced responses to herbivory in wild radish: effects on several herbivores and plant fitness. Ecology 80:1713–1723. https://doi.org/10.1890/0012-9658(1999)080[1713:IRTHIW]2.0.CO;2
Aleixo S, do Nascimento MT, Barroso DG (2016) Allelopathic potential of Corymbia citriodora (Hook.) KDHill & LASJohnson. on the establishment of seedlings of tree species in the Atlantic Forest. Iheringia Serie Botanica 71:277–282
Anselmo-Moreira F, Teixeira-Costa L, Ceccantini G, Furlan C (2019) Mistletoe effects on the host tree Tapirira guianensis: insights from primary and secondary metabolites. Evol Mech Chem Base Ecol Interact 29:11–24. https://doi.org/10.1007/s00049-018-0272-6
Badenes-Perez FR, Gershenzon J, Heckel DG (2014) Insect attraction versus plant defense: young leaves high in glucosinolates stimulate oviposition by a specialist herbivore despite poor larval survival due to high saponin content. PLoS ONE. https://doi.org/10.1371/journal.pone.0095766
Baldwin IT (1990) Herbivory simulations in ecological research. Trends Ecol Evol 5:91–93. https://doi.org/10.1016/0169-5347(90)90237-8
Barbosa LC, Filomeno CA, Teixeira RR (2016) Chemical variability and biological activities of Eucalyptus spp. essential oils. Molecules 21:33. https://doi.org/10.3390/molecules21121671
Björkman C, Dalin P, Ahrné K (2008) Leaf trichome responses to herbivory in willows: induction, relaxation and costs. New Phytol 179:176–184. https://doi.org/10.1111/j.1469-8137.2008.02442.x
Boland DJ, Brophy JJ, House APN (1991) Eucalyptus leaf oils: use, chemistry, distillation, and marketing. Inkata Press, Melbourne
Butler JB, Freeman JS, Potts BM, Vaillancourt RE, Grattapaglia D, Silva-Junior OB, Simmons BA, Healey AL, Schmutz J, Barry KW, Lee DJ, Henry RJ, King GJ, Baten A, Shepherd M (2018) Annotation of the Corymbia terpene synthase gene family shows broad conservation but dynamic evolution of physical clusters relative to Eucalyptus. Heredity 121:87–104. https://doi.org/10.1038/s41437-018-0058-1
Carne PB (1966) Ecological characteristics of the eucalypt-defoliating chrysomelid Paropsis atomaria Ol. Aust J Zool 14:647–672. https://doi.org/10.1071/ZO9660647
Carnegie A, Lawson S, Smith T, Pegg G, Stone C, McDonald J (2008) Healthy hardwoods: a field guide to pests, diseases and nutritional disorders in subtropical hardwoods. Forest and Wood Products Australia, Victoria
Clarke K, Gorley R (2015) Getting started with PRIMER v7. PRIMER-E: Plymouth, vol Plymouth Marine Laboratory
Coley PD (1983) Herbivory and defensive characteristics of tree species in a lowland tropical forest. Ecol Monogr 53:209–233. https://doi.org/10.2307/1942495
Edwards PB (1982) Do waxes on Eucalyptus leaves provide protection from grazing insects? Aust J Ecol 7:347–352. https://doi.org/10.1111/j.1442-9993.1982.tb01309.x
Edwards PB, Wanjura WJ, Brown WV (1993) Selective herbivory by Christmas beetles in response to intraspecific variation in Eucalyptus terpenoids. Oecologia 95:551–557
Eyles A, Bonello P, Ganley R, Mohammed C (2010) Induced resistance to pests and pathogens in trees. New Phytol 185:893–908. https://doi.org/10.1111/j.1469-8137.2009.03127.x
Fabisch T, Gershenzon J, Unsicker S (2019) Specificity of herbivore defense responses in a woody plant, black poplar (Populus nigra). J Chem Ecol 45:162–177. https://doi.org/10.1007/s10886-019-01050-y
Feeny P (1976) Plant apparency and chemical defense. In: Wallace JW, Mansell RL (eds) Biochemical interaction between plants and insects, vol 10. Springer, Boston, pp 1–40
Freeman BC, Beattie GA (2008) An overview of plant defenses against pathogens and herbivores. Plant Health Instr. https://doi.org/10.1094/PHI-I-2008-0226-01
Fürstenberg-Hägg J, Zagrobelny M, Bak S (2013) Plant defense against insect herbivores. Int J Mol Sci 14:10242–10297. https://doi.org/10.3390/ijms140510242
Hayes RA, Nahrung HF, Lee DJ (2013) Consequences of Corymbia (Myrtaceae) hybridisation on leaf-oil profiles. Aust J Bot 61:52–59. https://doi.org/10.1071/BT12224
Heil M (2014) Herbivore-induced plant volatiles: targets, perception and unanswered questions. New Phytol 204(2):297–306. https://doi.org/10.1111/nph.12977
Henery ML, Wallis IR, Stone C, Foley WJ (2008) Methyl jasmonate does not induce changes in Eucalyptus grandis leaves that alter the effect of constitutive defences on larvae of a specialist herbivore. Oecologia 156:847–859. https://doi.org/10.1007/s00442-008-1042-x
Jones TH, Potts BM, Vaillancourt RE, Davies NW (2002) Genetic resistance of Eucalyptus globulus to autumn gum moth defoliation and the role of cuticular waxes. Can J For Res (Revue Canadienne De Recherche Forestiere) 32:1961–1969. https://doi.org/10.1139/x02-118
Kaitaniemi P, Ruohomaki K, Ossipov V, Haukioja E, Pihlaja K (1998) Delayed induced changes in the biochemical composition of host plant leaves during an insect outbreak. Oecologia 116:182–190. https://doi.org/10.1007/s004420050578
Karban R, Baldwin IT (1997) Induced responses to herbivory. University of Chicago Press, Chicago. https://doi.org/10.7208/chicago/9780226424972.001.0001
Karban R, Agrawal AA, Thaler JS, Adler LS (1999) Induced plant responses and information content about risk of herbivory. Trends Ecol Evol 14:443–447. https://doi.org/10.1016/S0169-5347(99)01678-X
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144. https://doi.org/10.1126/science.291.5511.2141
King DJ, Gleadow RM, Woodrow IE (2004) Terpene deployment in Eucalyptus polybractea; relationships with leaf structure, environmental stresses, and growth. Funct Plant Biol 31(5):451–460. https://doi.org/10.1071/FP03217
King DJ, Gleadow RM, Woodrow IE (2006) The accumulation of terpenoid oils does not incur a growth cost in Eucalyptus polybractea seedlings. Funct Plant Biol 33:497–505. https://doi.org/10.1071/fp05304
Koricheva J, Nykänen H, Gianoli E (2004) Meta-analysis of trade-offs among plant antiherbivore defenses: are plants jacks-of-all-trades, masters of all? Am Nat 163:E64–E75. https://doi.org/10.1086/382601
Külheim C, Padovan A, Hefer C, Krause ST, Köllner TG, Myburg AA, Degenhardt J, Foley WJ (2015) The Eucalyptus terpene synthase gene family. BMC Genom 16:18. https://doi.org/10.1186/s12864-015-1598-x
Lamari L (2008) Assess 2.0: image analysis software for plant disease quantification. American Phytopathological Society, St. Paul
Lee DJ (2007) Achievements in forest tree genetic improvement in Australia and New Zealand 2: development of Corymbia species and hybrids for plantations in eastern Australia. Aust For 70:11–16. https://doi.org/10.1080/00049158.2007.10676256
Lee DJ, Huth JR, Brawner JT, Dickinson GR (2009) Comparative performance of Corymbia hybrids and parental species in subtropical queensland and implications for breeding and deployment. Silvae Genetica 58:205–212. https://doi.org/10.1515/sg-2009-0026
Lee DJ, Huth JR, Osborne DO, Hogg BW (2010) Selecting hardwood taxa for wood and fibre production in Queensland’s subtropics. Aust For 73:106–114. https://doi.org/10.1080/00049158.2010.10676316
Li H, Madden JL, Potts BM (1997) Variation in leaf waxes of the Tasmanian Eucalyptus species—I. Subgenus Symphyomyrtus. Biochem Syst Ecol 25:631–657. https://doi.org/10.1016/S0305-1978(97)00044-6
Maffei ME, Mithofer A, Boland W (2007) Before gene expression: early events in plant–insect interaction. Trends Plant Sci 12:310–316. https://doi.org/10.1016/j.tplants.2007.06.001
Malishev M, Sanson GD (2015) Leaf mechanics and herbivory defence: how tough tissue along the leaf body deters growing insect herbivores. Austral Ecol 40:300–308. https://doi.org/10.1111/aec.12214
Matsuki M, Foley WJ, Floyd RB (2011) Role of volatile and non-volatile plant secondary metabolites in host tree selection by Christmas beetles. J Chem Ecol 37:286–300. https://doi.org/10.1007/s10886-011-9916-5
McCall AC, Fordyce JA (2010) Can optimal defence theory be used to predict the distribution of plant chemical defences? J Ecol 98:985–992. https://doi.org/10.1111/j.1365-2745.2010.01693.x
Mitchell C, Brennan RM, Graham J, Karley AJ (2016) Plant defense against herbivorous pests: exploiting resistance and tolerance traits for sustainable crop protection. Front Plant Sci 7:8. https://doi.org/10.3389/fpls.2016.01132
Mithöfer A, Boland W (2012) Plant defense against herbivores: chemical aspects. Annu Rev of Plant Biol 63(1):431–450. https://doi.org/10.1146/annurev-arplant-042110-103854
Moreira X, Lundborg L, Zas R, Carrillo-Gavilán A, Borg-Karlson A-K, Sampedro L (2013) Inducibility of chemical defences by two chewing insect herbivores in pine trees is specific to targeted plant tissue, particular herbivore and defensive trait. Phytochemistry 94:113–122
Müller C, Riederer M (2005) Plant surface properties in chemical ecology. J Chem Ecol 31:2621–2651. https://doi.org/10.1007/s10886-005-7617-7
Nahrung HF, Waugh R (2012) Eriophyid mites on spotted gums: population and histological damage studies of an emerging pest. Int J Acarol 38:549–556. https://doi.org/10.1080/01647954.2012.709277
Nahrung HF, Dunstan PK, Allen GR (2001) Larval gregariousness and neonate establishment of the eucalypt-feeding beetle Chrysophtharta agricola (Coleoptera:Chrysomelidae:Paropsini). Oikos 94:358–364. https://doi.org/10.1034/j.1600-0706.2001.940217.x
Nahrung HF, Waugh R, Hayes RA (2009) Corymbia species and hybrids: chemical and physical foliar attributes and implications for herbivory. J Chem Ecol 35:1043–1053. https://doi.org/10.1007/s10886-009-9682-9
Naidoo S, Külheim C, Zwart L, Mangwanda R, Oates CN, Visser EA, Wilken FE, Mamni TB, Myburg AA (2014) Uncovering the defence responses of Eucalyptus to pests and pathogens in the genomics age. Tree Physiol 34:931–943. https://doi.org/10.1093/treephys/tpu075
Naidoo S, Christie N, Acosta JJ, Mphahlele MM, Payn KG, Myburg AA, Külheim C (2018) Terpenes associated with resistance against the gall wasp Leptocybe invasa, in Eucalyptus grandis. Plant Cell Environ 41(8):1840–1851. https://doi.org/10.1111/pce.13323
Neinhuis C, Barthlott W (1997) Characterization and distribution of water-repellent, self-cleaning plant surfaces. Ann Bot 79:667–677. https://doi.org/10.1006/anbo.1997.0400
Oates CN, Külheim C, Myburg AA, Slippers B, Naidoo S (2015) The transcriptome and terpene profile of Eucalyptus grandis reveals mechanisms of defense against the insect pest Leptocybe invasa. Plant Cell Physiol 56:1418–1428. https://doi.org/10.1093/pcp/pcv064
Paine TD, Steinbauer MJ, Lawson SA (2011) Native and exotic pests of Eucalyptus: a worldwide perspective. Annu Rev of Entomol 56(1):181–201. https://doi.org/10.1146/annurev-ento-120709-144817
Parra-O C, Bayly M, Udovicic F, Ladiges P (2006) ETS sequences support the monophyly of the eucalypt genus Corymbia (Myrtaceae). Taxon 55:653–663. https://doi.org/10.2307/25065641
Patton MF, Arena GD, Salminen JP, Steinbauer MJ, Casteel CL (2018) Transcriptome and defence response in Eucalyptus camaldulensis leaves to feeding by Glycaspis brimblecombei Moore (Hemiptera: Aphalaridae): a stealthy psyllid does not go unnoticed. Austral Entomol 57:247–254. https://doi.org/10.1111/aen.12319
Rapley LP, Allen GR, Potts BM (2004) Susceptibility of Eucalyptus globulus to Mnesampela privata defoliation in relation to a specific foliar wax compound. Chemoecology 14:157–163. https://doi.org/10.1007/s00049-004-0279-z
Rapley LP, Allen GR, Potts BM, Davies NW (2007) Constitutive or induced defences—how does Eucalyptus globulus defend itself from larval feeding? Chemoecology 17:235–243. https://doi.org/10.1007/s00049-007-0382-z
Sanchez-Serrano JJ (2017) Plant responses to wounding. Encyclopedia of life sciences. Wiley, Chichester. https://doi.org/10.1002/9780470015902.a0001321.pub2
Savatin DV, Gramegna G, Modesti V, Cervone F (2014) Wounding in the plant tissue: the defense of a dangerous passage. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00470
Schutze MK (2008) The significance of genetic and ecological diversity in a wide-ranging insect pest, Paropsis atomaria Olivier (Coleoptera: Chrysomelidae). Queensland University of Technology
Smith AH, Gill WM, Pinkard EA, Mohammed CL (2007) Anatomical and histochemical defence responses induced in juvenile leaves of Eucalyptus globulus and Eucalyptus nitens by Mycosphaerella infection. Forest Pathol 37:361–373. https://doi.org/10.1111/j.1439-0329.2007.00502.x
Staudt M, Lhoutellier L (2007) Volatile organic compound emission from holm oak infested by gypsy moth larvae: evidence for distinct responses in damaged and undamaged leaves. Tree Physiol 27:1433–1440. https://doi.org/10.1093/treephys/27.10.1433
Steinbauer MJ (2001) Specific leaf weight as an indicator of juvenile leaf toughness in Tasmanian bluegum (Eucalyptus globulus ssp. globulus): implications for insect defoliation. Aust For 64:32–37. https://doi.org/10.1080/00049158.2001.10676158
Stone C (2001) Reducing the impact of insect herbivory in eucalypt plantations through management of extrinsic influences on tree vigour. Austral Ecol 26:482–488. https://doi.org/10.1046/j.1442-9993.2001.01143.x
War AR, Taggar GK, Hussain B, Taggar MS, Nair RM, Sharma HC (2018) Plant defence against herbivory and insect adaptations. AoB Plants. https://doi.org/10.1093/aobpla/ply037
Waterman JM, Cazzonelli CI, Hartley SE, Johnson SN (2019) Simulated herbivory: the key to disentangling plant defence responses. Trends Ecol Evol. https://doi.org/10.1016/j.tree.2019.01.008
Wingfield MJ, Roux J, Slippers B, Hurley BP, Garnas J, Myburg AA, Wingfield BD (2013) Established and new technologies reduce increasing pest and pathogen threats to Eucalypt plantations. For Ecol Manag 301:35–42. https://doi.org/10.1016/j.foreco.2012.09.002
Zhang CL, Li XW, Chen YQ, Zhao J, Wan SZ, Lin YB, Fu SL (2016) Effects of Eucalyptus litter and roots on the establishment of native tree species in Eucalyptus plantations in South China. For Ecol Manag 375:76–83. https://doi.org/10.1016/j.foreco.2016.05.013
Acknowledgements
We would like to thank Dr Manon Griffiths and Ngoc Hoan Le for assisting with the insect collection for this study, and Dr Mervyn Shepherd (Southern Cross University) for the pre-submission review of this manuscript. This work was supported by University of the Sunshine Coast International Research Scholarship (USCIRS) awarded to FSB.
Author information
Authors and Affiliations
Contributions
FSB, RAH, HFN and DJL conceived and planned the experiments. FSB carried out the experiments, measurements and carried out sample preparation. FSB, RAH, HFN and DJL contributed to the analyses and interpretation of the results. FSB drafted the manuscript, and all authors provided critical feedback and helped shape the research, analysis and manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by LeBoldus.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bonora, F.S., Hayes, R.A., Nahrung, H.F. et al. Paropsis atomaria larval feeding induces a chemical but not a physical response in Corymbia citriodora subsp. variegata. Trees 35, 863–873 (2021). https://doi.org/10.1007/s00468-021-02086-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-021-02086-y