Abstract
Key message
Molecular analysis of a zeta subfamily GST gene from T. hispida involved in ABA and methyl viologen tolerance in transgenic Arabidopsis and Tamarix.
Abstract
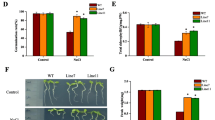
Glutathione S-transferase (GST) genes are important for the improvement of plant abiotic stress tolerance, and our previous study demonstrated that the ThGSTZ1 gene from Tamarix hispida improves plant salt and drought tolerance. To further understand the role of ThGSTZ1 in the response of plants to abscisic acid (ABA) and oxidative stress, three ThGSTZ1-overexpressing transgenic Arabidopsis thaliana lines were analyzed in the current study. The results showed that the transgenic lines exhibited higher biomass accumulation, higher activities of GST and other protective enzymes, and less reactive oxygen species (ROS) and cell damage than wild-type (WT) plants under ABA and methyl viologen (MV) stress. In addition, the analysis of a transgenic T. hispida line transiently expressing ThGSTZ1 confirmed these results. The activities of GST, glutathione peroxidase, and superoxide dismutase were markedly higher in the ThGSTZ1-overexpressing lines compared with the control lines under both ABA and MV treatments, and the transgenic lines also exhibited a lower degree of electrolyte leakage (EL) and a decreased H2O2 content. All these results suggested that ThGSTZ1 can also improve plant ABA and oxidation tolerance by regulating ROS metabolism and that ThGSTZ1 represents an excellent candidate gene for molecular breeding to increase plant stress tolerance.







Similar content being viewed by others
References
Asada K (2006) Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol 141:391–396
Bailey-Serres J, Mittler R (2006) The roles of reactive oxygen species in plant cells. Plant Physiol 141:311
Bianchi MW, Roux C, Vartanian N (2002) Drought regulation of GST8, encoding the Arabidopsis homologue of ParC/Nt107 glutathione transferase/peroxidase. Physiol Plant 116:96–105
Chen W, Singh KB (2002) The auxin, hydrogen peroxide and salicylic acid induced expression of the Arabidopsis GST6 promoter is mediated in part by an ocs element. Plant J. 19:667–677
Chen W, Chao G, Singh KB (1996) The promoter of a H2O2-inducible, Arabidopsis glutathione S-transferase gene contains closely linked OBF-and OBP1-binding sites. Plant J. 10:955–966
Chen JH, Jiang HW, Hsieh EJ, Chen HY, Chien CT, Hsieh HL, Lin TP (2012) Drought and salt stress tolerance of an Arabidopsis glutathione S-transferase U17 knockout mutant are attributed to the combined effect of glutathione and abscisic acid. Plant Physiol 158:340–351
Dinler BS, Antoniou C, Fotopoulos V (2014) Interplay between GST and nitric oxide in the early response of soybean (Glycine max L.) plants to salinity stress. J Plant Physiol 171:1740–1747
Dixon D, Hartmann D, Kolaczyk E, Samimi J, Diehl R, Kanbach G, Mayer-Hasselwander H, Strong A (1998) Evidence for a galactic gamma-ray halo. New Astron 3:539–561
Dixon DP, Lapthorn A, Edwards R (2002) Plant glutathione transferases. Genome Biol 3:1–10
Edwards R, Dixon DP, Walbot V (2000) Plant glutathione-S-transferases: enzymes with multiple functions in sickness and in health. Trends Plant Sci 5:193–198
Ezaki B, Suzuki M, Motoda H, Kawamura M, Nakashima S, Matsumoto H (2004) Mechanism of gene expression of Arabidopsis glutathione S-transferase, AtGST1, and AtGST11 in response to aluminum stress. Plant Physiol 134:1672–1682
Fath A, Bethke P, Beligni V, Jones R (2002) Active oxygen and cell death in cereal aleurone cells. J Exp Bot 53:1273–1282
Frova C (2003) The plant glutathione transferase gene family: genomic structure, functions, expression and evolution. Physiol Plant 119:469–479
Galle A, Csiszar J, Benyo D, Laskay G, Leviczky T, Erdei L, Tari I (2013) Isohydric and anisohydric strategies of wheat genotypes under osmotic stress: biosynthesis and function of ABA in stress responses. J Plant Physiol 170:1389–1399
Gao C, Wang Y, Liu G, Wang C, Jiang J, Yang C (2010) Cloning of ten peroxidase (POD) genes from Tamarix hispida and characterization of their responses to abiotic stress. Plant Mol Biol Rep 28:77–89
Gao C, Zhang K, Yang G, Wang Y (2012) Expression analysis of four peroxiredoxin genes from Tamarix hispida in response to different abiotic stresses and exogenous abscisic acid (ABA). Int J Mol Sci 13:3751–3764
Hamilton DW, Hills A, Kohler B, Blatt MR (2000) Ca2+ channels at the plasma membrane of stomatal guard cells are activated by hyperpolarization and abscisic acid. Proc Natl Acad Sci USA 97:4967–4972
Hayes JD, Flanagan JU, Jowsey IR (2005) Glutathione transferases. Annu Rev Pharmacol Toxicol 45:51–88
Hoque MA, Uraji M, Banu MNA, Mori IC, Nakamura Y, Murata Y (2010) The effects of methylglyoxal on glutathione S-transferase from Nicotiana tabacum. Biosci Biotechnol Biochem 74:2124–2126
Jacobson MD (1996) Reactive oxygen species and programmed cell death. Trends Biochem Sci 21:83
Jain M, Ghanashyam C, Bhattacharjee A (2010) Comprehensive expression analysis suggests overlapping and specific roles of rice glutathione S-transferase genes during development and stress responses. BMC Genom 11:73
Jha B, Sharma A, Mishra A (2011) Expression of SbGSTU (tau class glutathione S-transferase) gene isolated from Salicornia brachiata in tobacco for salt tolerance. Mol Biol Rep 38:4823–4832
Ji X, Liu G, Liu Y, Zheng L, Nie X, Wang Y (2013) The bZIP protein from Tamarix hispida, ThbZIP1, is ACGT elements binding factor that enhances abiotic stress signaling in transgenic Arabidopsis. BMC Plant Biol 13:151
Jiang Y, Yang B, Harris NS, Deyholos MK (2007) Comparative proteomic analysis of NaCl stress-responsive proteins in Arabidopsis roots. J Exp Bot 58(13):3591–3607
Kiyosue T, Yamaguchi-Shinozaki K, Shinozaki K (1993) Characterization of two cDNAs (ERD11 and ERD13) for dehydration-inducible genes that encode putative glutathione S-transferases in Arabidopsis thaliana L. FEBS Lett 335:189–192
Kouno T, Ezaki B (2013) Multiple regulation of Arabidopsis AtGST11 gene expression by four transcription factors under abiotic stresses. Physiol Plant 148(1):97–104
Kumar S, Asif MH, Chakrabarty D, Tripathi RD, Dubey RS, Trivedi PK (2013) Expression of a rice Lambda class of glutathione S-transferase, OsGSTL2, in Arabidopsis provides tolerance to heavy metal and other abiotic stresses. J Hazard Mater 248:228–237
Kwak KJ, Kim YO, Kang H (2005) Characterization of transgenic Arabidopsis plants overexpressing GR-RBP4 under high salinity, dehydration, or cold stress. J Exp Bot 56:3007–3016
Lan T, Yang ZL, Yang X, Liu YJ, Wang XR, Zeng QY (2009) Extensive functional diversification of the Populus glutathione S-transferase supergene family. Plant Cell. 21:3749–3766
Li Q, Song J, Peng S, Wang JP, Qu GZ, Sederoff RR, Chiang VL (2014) Plant biotechnology for lignocellulosic biofuel production. Plant Biotechnol J 12:1174–1192
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33:453–467
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Mittler R, Vanderauwera S, Suzuki N, Miller G, Tognetti VB, Vandepoele K, Gollery M, Shulaev V, Van Breusegem F (2011) ROS signaling: the new wave? Trends Plant Sci 16:300–309
Moons A (2003) Osgstu3 and Osgtu4, encoding tau class glutathione S-transferases, are heavy metal-and hypoxic stress-induced and differentially salt stress-responsive in rice roots. FEBS Lett 553:427–432
Moons A (2005) Regulatory and functional interactions of plant growth regulators and plant glutathione S-transferases (GSTs). Vitam Horm 72:155–202
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49:249–279
Pantin F, Simonneau T, Muller B (2012) Coming of leaf age: control of growth by hydraulics and metabolics during leaf ontogeny. New Phytol 196:349–366
Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406:731–734
Pitzschke A, Schikora A, Hirt H (2009) Mapk cascade signalling networks in plant defence. Curr Opin Plant Biol 12(4):421–426
Ren H, Gao Z, Chen L, Wei K, Liu J, Fan Y, Davies WJ, Jia W, Zhang J (2007) Dynamic analysis of ABA accumulation in relation to the rate of ABA catabolism in maize tissues under water deficit. J Exp Bot 58:211–219
Ren X, Chen Z, Liu Y, Zhang H, Zhang M, Liu Q, Hong X, Zhu JK, Gong Z (2010) ABO3, a WRKY transcription factor, mediates plant responses to abscisic acid and drought tolerance in Arabidopsis. Plant J 63:417–429
Sharma R, Sahoo A, Devendran R, Jain M (2014) Over-expression of a rice tau class glutathione S-transferase gene improves tolerance to salinity and oxidative stresses in Arabidopsis. PLoS One 9(3):e92900
Sytykiewicz H (2011) Expression patterns of glutathione transferase gene (GstI) in maize seedlings under juglone-induced oxidative stress. Int J Mol Sci 12:7982–7995
Talaat IM, Khattab HI, Ahmed AM (2014) Changes in growth, hormones levels and essential oil content of Ammi visnaga L. plants treated with some bioregulators. Saudi J Biol Sci 21:355–365
Terada T, Maeda H, Okamoto K, Nishinaka T, Mizoguchi T, Nishihara T (1993) Modulation of glutathione s-transferase activity by a thiol/disulfide exchange reaction and involvement of thioltransferase. Arch Biochem Biophys 300:495–500
Torres MA, Dangl JL (2005) Functions of the respiratory burst oxidase in biotic interactions, abiotic stress and development. Curr Opin Plant Biol 8:397–403
Vollenweider S, Weber H, Stolz S, Chételat A, Farmer EE (2000) Fatty acid ketodienes and fatty acid ketotrienes: Michael addition acceptors that accumulate in wounded and diseased Arabidopsis leaves. Plant J 24:467–476
Wang Y, Ying Y, Chen J, Wang X (2004) Transgenic Arabidopsis overexpressing Mn-SOD enhanced salt-tolerance. Plant Sci 167:671–677
Wang Y, Gao C, Liang Y, Wang C, Yang C, Liu G (2010) A novel bZIP gene from Tamarix hispida mediates physiological responses to salt stress in tobacco plants. J Plant Physiol 167:222–230
Yang G, Wang Y, Xia D, Gao C, Wang C, Yang C (2014a) Overexpression of a GST gene (ThGSTZ1) from Tamarix hispida improves drought and salinity tolerance by enhancing the ability to scavenge reactive oxygen species. Plant Cell Tiss Org 117:99–112
Yang G, Wang Y, Zhang K, Gao C (2014b) Expression analysis of nine small heat shock protein genes from Tamarix hispida in response to different abiotic stresses and abscisic acid treatment. Mol Biol Rep 41:1279–1289
Zhai CZ, Zhao L, Yin LJ, Chen M, Wang QY, Li LC, Xu ZS, Ma YZ (2013) Two wheat glutathione peroxidase genes whose products are located in chloroplasts improve salt and H2O2 tolerances in Arabidopsis. PLoS One 8:e73989
Zhang Y, Wang Y, Wang C (2012) Gene overexpression and gene silencing in Birch using an Agrobacterium-mediated transient expression system. Mol Biol Rep 39:5537–5541
Zheng L, Liu G, Meng X, Li Y, Wang Y (2012) A versatile agrobacterium-mediated transient gene expression system for herbaceous plants and trees. Biochem Genet 50:761–769
Zheng L, Liu G, Meng X, Liu Y, Ji X, Li Y, Nie X, Wang Y (2013) A WRKY gene from Tamarix hispida, ThWRKY4, mediates abiotic stress responses by modulating reactive oxygen species and expression of stress-responsive genes. Plant Mol Biol 82:303–320
Acknowledgments
This work was supported by the Fundamental Research Funds for the Central Universities (2572014DA03), the National Natural Science Foundation of China (No. 31370676), the Scientific Research Fund of Heilongjiang Provincial Education Department (No. 12523017), and the Postdoctoral Science Foundation of Heilongjiang Province (No. LBH-Q10155).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Carlson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gao, C., Yang, G., Guo, Y. et al. Overexpression of ThGSTZ1 from Tamarix hispida improves tolerance to exogenous ABA and methyl viologen. Trees 30, 1935–1944 (2016). https://doi.org/10.1007/s00468-016-1422-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-016-1422-5