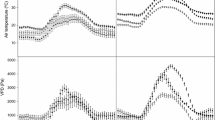
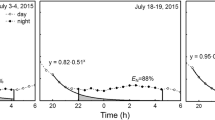
Abstract
The aim of this study was to examine the diurnal and seasonal variations in the sensitivity of leaf lamina (K lam) hydraulic conductance to irradiance in bur oak (Quercus macrocarpa Michx.) and trembling aspen (Populus tremuloides Michx.), which vary in their responses of K lam to irradiance. K lam was determined using the high-pressure method and the measurements were carried out in June, August and September. The irradiance response of K lam in bur oak was present throughout the day and declined in senescing leaves. In trembling aspen, K lam declined from morning to late afternoon and drastically decreased before the onset of leaf senescence, but it was not sensitive to irradiance. In both tree species, the capacity of the petioles to supply water to leaf lamina changed during the day in accordance with the ability of the leaf lamina to transport water. Petiole hydraulic conductivity (K pet) declined during the season in bur oak leaves, while it tended to increase in trembling aspen leaves. There was no correlation between the K lam values and air temperature or light intensity at the time of leaf collection. For trembling aspen, K pet was negatively correlated with the air temperature suggesting sensitivity to drought. We conclude that the water transport properties of petioles and leaf lamina in the two studied tree species reflect their ecological adaptations. Trembling aspen leaves have high hydraulic conductivity and high stomatal conductance regardless of the irradiance level, consistent with the rapid growth and high demand for water. In contrast, the increased lamina hydraulic conductivity and stomatal conductance under high irradiance in bur oak trees reflect a water conservation strategy.





Similar content being viewed by others
References
Aasamaa K, Sõber A (2001) Hydraulic conductance and stomatal sensitivity to changes of leaf water status in six deciduous tree species. Biol Plant 44:65–73. doi:10.1023/A:1017970304768
Aasamaa K, Sõber A (2005) Seasonal courses of maximum hydraulic conductance in shoots of six temperate deciduous tree species. Funct Plant Biol 32:1077–1087. doi:10.1071/FP05088
Aasamaa K, Sõber A, Rahi M (2001) Leaf anatomical characteristics associated with shoot hydraulic conductance, stomatal conductance and stomatal sensitivity to changes of leaf water status in temperate deciduous trees. Aust J Plant Physiol 28:765–774. doi:10.1071/PP00157
Brodribb TJ, Holbrook NM (2003a) Changes in leaf hydraulic conductance during leaf shedding in seasonally dry tropical forest. New Phytol 158:295–303. doi:10.1046/j.1469-8137.2003.00736.x
Brodribb TJ, Holbrook NM (2003b) Stomatal closure during leaf dehydration, correlation with other leaf physiological traits. Plant Physiol 132:2166–2173. doi:10.1104/pp.103.023879
Brodribb TJ, Holbrook NM (2004) Diurnal depression of leaf hydraulic conductance in a tropical tree species. Plant Cell Environ 27:820–827. doi:10.1111/j.1365-3040.2004.01188.x
Brodribb TJ, Holbrook NM, Zwieniecki MA, Palma B (2005) Leaf hydraulic capacity in ferns, conifers and angiosperms: impacts on photosynthetic maxima. New Phytol 165:839–846. doi:10.1111/j.1469-8137.2004.01259.x
Brodribb TJ, Feild TS, Jordan GJ (2007) Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol 144:1890–1898. doi:10.1104/pp.107.101352
Cochard H, Venisse JS, Barigah TS et al (2007) Putative role of aquaporins in variable hydraulic conductance of leaves in response to light. Plant Physiol 143:122–133. doi:10.1104/pp.106.090092
Fleet DSV (1950) The cell forms, and their common substance reactions, in the parenchyma–vascular boundary. Bull Torrey Bot Club 77:340–353
Fromard L, Babin V, Fleurat-Lessard P, Fromont JC, Serrano R, Bonnemain JL (1995) Control of vascular sap pH by the vessel-associated cells in woody species (physiological and immunological studies). Plant Physiol 108:913–918. doi:10.1104/pp.108.3.913
Hachez C, Heinen R, Draye X, Chaumont F (2008) The expression pattern of plasma membrane aquaporins in maize leaf highlights their role in hydraulic regulation. Plant Mol Biol 68:337–353. doi:10.1007/s11103-008-9373-x
Hacke U, Sauter JJ (1996) Drought-induced xylem dysfunction in petioles, branches, and roots of Populus balsamifera L. and Alnus glutinosa (L.) Gaertn. Plant Physiol 111:413–417. doi:10.1104/pp.111.2.413
Hacke UG, Stiller V, Sperry JS, Pittermann J, McCulloh KA (2001) Cavitation fatigue. Embolism and refilling cycles can weaken the cavitation resistance of xylem. Plant Physiol 125:779–786. doi:10.1104/pp.125.2.779
Hamerlynck E, Knapp AK (1994) Stomatal responses to variable sunlight in bur oak (Quercus macrocarpa Michx.) leaves with different photosynthetic capacities. Int J Plant Sci 155:583–587
Johnson DM, Woodruff DR, Mcculloh KA, Meinzer FC (2009) Leaf hydraulic conductance, measured in situ, declines and recovers daily: leaf hydraulics, water potential and stomatal conductance in four temperate and three tropical tree species. Tree Physiol 29:879–887. doi:10.1093/treephys/tpp031
Knapp AK (1992) Leaf gas exchange in Quercus macrocarpa (Fagaceae): rapid stomatal responses to variable sunlight in a tree growth form. Am J Bot 79:599–604
Lo Gullo MA, Nardini A, Trifilo P, Salleo S (2005) Diurnal and seasonal variations in leaf hydraulic conductance in evergreen and deciduous trees. Tree Physiol 25:505–512. doi:10.1093/treephys/25.4.505
Moshelion M, Becker D, Biela A et al (2002) Plasma membrane aquaporins in the motor cells of Samanea saman: diurnal and circadian regulation. Plant Cell 14:727–739. doi:10.1105/tpc.010351
Nardini A, Tyree MT, Salleo S (2001) Xylem cavitation in the leaf of Prunus laurocerasus and its impact on leaf hydraulics. Plant Physiol 125:1700–1709. doi:10.1104/pp.125.4.1700
Nardini A, Salleo S, Raimondo F (2003) Changes in leaf hydraulic conductance correlate with leaf vein embolism in Cercis siliquastrum L. Trees 17:529–534. doi:10.1007/s00468-003-0265-z
Nardini A, Salleo S, Andri S (2005) Circadian regulation of leaf hydraulic conductance in sunflower (Helianthus annuus L. cv Margot). Plant Cell Environ 28:750–759. doi:10.1111/j.1365-3040.2005.01320.x
Roden JS, Pearcy RW (1993) Photosynthetic gas exchange response of poplars to steady-state and dynamic light environments. Oecologia 93:208–214. doi:10.1007/BF00317673
Sack L, Frole K (2006) Leaf structural diversity is related to hydraulic capacity in tropical rain forest trees. Ecology 87:483–491. doi:10.1890/05-0710
Sack L, Holbrook NM (2006) Leaf hydraulics. Annu Rev Plant Biol 57:361–381. doi:10.1146/annurev.arplant.56.032604.144141
Sack L, Melcher PJ, Zwieniecki MA, Holbrook NM (2002) The hydraulic conductance of the angiosperm leaf lamina: a comparison of three measurement methods. J Exp Bot 53:2177–2184. doi:10.1093/jxb/erf069
Sack L, Cowan PD, Jaikumar N, Holbrook NM (2003) The ‘hydrology’ of leaves: co-ordination of structure and function in temperate woody species. Plant Cell Environ 26:1343–1356. doi:10.1046/j.0016-8025.2003.01058.x
Sack L, Streeter CM, Holbrook NM (2004) Hydraulic analysis of water flow through leaves of sugar maple and red oak. Plant Physiol 134:1824–1833. doi:10.1104/pp.103.031203
Sack L, Tyree MT, Holbrook NM (2005) Leaf hydraulic architecture correlates with regeneration irradiance in tropical rainforest trees. New Phytol 167:403–413. doi:10.1111/j.1469-8137.2005.01432.x
Salleo S, Nardini A, Lo Gullo MA, Ghirardelli LA (2002) Changes in stem and leaf hydraulics preceding leaf shedding in Castanea sativa L. Biol Plant 45:227–234. doi:10.1023/A:1015192522354
Scoffoni C, Pou A, Aasamaa K, Sack L (2008) The rapid light response of leaf hydraulic conductance: new evidence from two experimental methods. Plant Cell Environ 31:1803–1812. doi:10.1111/j.1365-3040.2008.01884.x
Shabala S, Newman I (1999) Light-induced changes in hydrogen, calcium, potassium, and chloride ion fluxes and concentrations from the mesophyll and epidermal tissues of bean leaves. Understanding the ionic basis of light-induced bioelectrogenesis. Plant Physiol 119:1115–1124. doi:10.1104/pp.119.3.1115
Sinclair TR, Zwieniecki MA, Holbrook NA (2008) Low leaf hydraulic conductance associated with drought tolerance in soybean. Physiol Plant 132:446–451. doi:10.1111/j.1399-3054.2007.01028.x
Siso S, Camarero J, Gil-Pelegrin E (2001) Relationship between hydraulic resistance and leaf morphology in broadleaf Quercus species: a new interpretation of leaf lobation. Trees 15:341–345. doi:10.1007/s004680100110
Tobiessen P, Kana TM (1974) Drought-stress avoidance in three pioneer tree species. Ecology 55:667–670. doi:10.2307/1935159
Tyree MT, Patiño S, Bennink J, Alexander J (1995) Dynamic measurements of roots hydraulic conductance using a high-pressure flowmeter in the laboratory and field. J Exp Bot 46:83–94. doi:10.1093/jxb/46.1.83
Tyree MT, Nardini A, Salleo S, Sack L, El Omari B (2005) The dependence of leaf hydraulic conductance on irradiance during HPFM measurements: any role for stomatal response? J Exp Bot 56:737–744. doi:10.1093/jxb/eri045
Voicu MC, Zwiazek JJ (2010) Inhibitor studies of leaf lamina hydraulic conductance in trembling aspen (Populus tremuloides Michx.) leaves. Tree Physiol 30:193–204. doi:10.1093/treephys/tpp112
Voicu MC, Zwiazek JJ, Tyree MT (2008) Light response of hydraulic conductance in bur oak (Quercus macrocarpa) leaves. Tree Physiol 28:1007–1015. doi:10.1093/treephys/28.7.1007
Wylie RB (1952) The bundle sheath extension in leaves of dicotyledons. Am J Bot 39:645–651
Zwieniecki MA (2001) Hydrogel control of xylem hydraulic resistance in plants. Science 291:1059–1062. doi:10.1126/science.1057175
Zwieniecki MA, Melcher PJ, Feild TS, Holbrook NM (2004) A potential role for xylem–phloem interactions in the hydraulic architecture of trees: effects of phloem girdling on xylem hydraulic conductance. Tree Physiol 24:911–917. doi:10.1093/treephys/24.8.911
Zwieniecki MA, Brodribb TJ, Holbrook NM (2007) Hydraulic design of leaves: insights from rehydration kinetics. Plant Cell Environ 30:910–921. doi:10.1111/j.1365-3040.2007.001681.x
Acknowledgments
We thank the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Alberta Ingenuity Fund (AIF) for research funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Guy.
Rights and permissions
About this article
Cite this article
Voicu, M.C., Zwiazek, J.J. Diurnal and seasonal changes of leaf lamina hydraulic conductance in bur oak (Quercus macrocarpa) and trembling aspen (Populus tremuloides). Trees 25, 485–495 (2011). https://doi.org/10.1007/s00468-010-0524-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-010-0524-8