Abstract
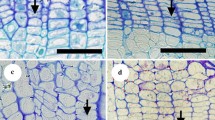
A dye injection method was used to elucidate the xylem water-conducting pathways of 34 broadleaved evergreen trees growing in southern Japan: two semi-ring-porous, 26 diffuse-porous, five radial-porous and one non-vessel species. The large earlywood vessels in semi-ring-porous species have a water transport function in only the outermost annual ring, as in deciduous ring-porous species. On the other hand, the small vessels in semi-ring-porous species maintain the water transport function in many outer annual rings. For the other xylem-type species, the many vessels in many outer annual rings have a water transport function. In diffuse-porous species, we categorized the water-conducting pattern within the annual rings into two types: d1 type, where water travels through vessels in the whole region; and d2 type, where water travels mainly through the earlywood vessels. The pattern in radial-porous species is similar to that in the d1 type; the pattern in non-vessels species is similar to that in the d2 type. The vessel diameter in radial-porous species is similar to that of the earlywood vessels of semi-ring-porous species. These results suggest that the conduit diameter size is only one of many factors determining the water-conducting pathways of broadleaved evergreen species.




Similar content being viewed by others
References
Anfodillo T, Sigalotti GB, Tomasi M, Semenzato P, Valentini R (1993) Applications of a thermal imaging technique in the study of the ascent of sap in woody species. Plant Cell Environ 16:997–1001
Axelrod DI (1966) Origin of deciduous and evergreen habits in temperate forests. Evolution 20:1–15
Baker H, James WO (1933) The behaviour of dyes in the transpiration stream of sycamores (Acer pseudoplatanus L.). New Phytol 32:245–260
Cavender-Bares J, Holbrook NM (2001) Hydraulic properties and freezing-induced cavitation in sympatric evergreen and deciduous oaks with contrasting habitats. Plant Cell Environ 24:1243–1256
Cavender-Bares J, Cortes P, Rambal S, Joffre R, Miles B, Rocheteau A (2005) Summer and winter sensitivity of leaves and xylem to minimum freezing temperatures: a comparison of co-occurring Mediterranean oaks that differ in leaf lifespan. New Phytol 168:597–612
Chabot BF, Hicks DJ (1982) The ecology of leaf life spans. Ann Rev Ecol Syst 13:229–259
Chaney WR, Kozlowski TT (1977) Patterns of water movement in intact and excised stems of Fraxinus americana and Acer saccharum seedlings. Ann Bot 41:1093–1100
Christman MA, Sperry JS, Adler FR (2009) Testing the ‘rare pit’ hypothesis for xylem cavitation resistance in three species of Acer. New Phytol 182:664–674
Cochard H, Tyree MT (1990) Xylem dysfunction in Quercus: vessel sizes, tyloses, cavitation and seasonal changes in embolism. Tree Physiol 6:393–407
Davis SD, Sperry JS, Hacke UG (1999) The relationship between xylem conduit diameter and cavitation caused by freezing. Am J Bot 86:1367–1372
DeFries RS, Hansen MC, Townshend JRG, Janetos AC, Loveland TR (2000) A new global 1-km dataset of percentage tree cover derived from remote sensing. Glob Change Biol 6:247–254
Denne MP (1988) Definition of latewood according to Mork (1928). IAWA Bull New Ser 10:59–62
Ellmore GS, Ewers FW (1986) Fluid flow in the outermost xylem increment of a ring-porous tree, Ulmus americana. Am J Bot 73:1771–1774
Ewers FW (1985) Xylem structure and water conduction in conifer trees, dicot trees, and lianas. IAWA Bull New Ser 6:309–317
Ewers FW, Cochard H, Tyree MT (1997) A survey of root pressures in vines of a tropical lowland forest. Oecologia 110:191–196
Feild TS, Brodribb T, Holbrook NM (2002) Hardly a relict: freezing and the evolution of vesselless wood in Winteraceae. Evolution 56:464–478
Fujikawa S, Suzuki T, Ishikawa T, Sakurai S, Hasegawa Y (1988) Continuous observation of frozen biological-materials with cryo-scanning electron microscope and freeze-replica by a new cryo-system. J Electron Microsc 37:315–322
Gilbert SG (1940) Evolutionary significance of ring porosity in woody angiosperms. Bot Gaz 102:105–120
Givnish TJ (2002) Adaptive significance of evergreen vs. deciduous leaves: solving the triple paradox. Silva Fenn 36:703–743
Greenidge KNH (1958) Rates and patterns of moisture movement in trees. In: Thimann KV (ed) The physiology of forest trees. Ronald, New York, pp 19–41
Hacke UG, Sperry JS, Wheeler JK, Castro L (2006) Scaling of angiosperm xylem structure with safety and efficiency. Tree Physiol 26:689–701
Hallé F, Oldeman RAA, Tomlinson PB (1978) Tropical trees and forests: an architectural analysis. Springer, Berlin
Hirose S, Kume A, Takeuchi S, Utsumi Y, Otsuki K, Ogawa S (2005) Stem water transport of Lithocarpus edulis, an evergreen oak with radial-porous wood. Tree Physiol 25:221–228
Ide J, Nagafuchi O, Chiwa M, Kume A, Otsuki K, Ogawa S (2007) Effects of discharge level on the load of dissolved and particulate components of stream nitrogen and phosphorus from a small afforested watershed of Japanese cypress (Chamaecyparis obtusa). J For Res 12:45–56
Inoue S, Utsumi Y, Otsuki K, Okano T, Koga S, Tashiro N, Nakai T (2002) Forest and tree of Kyushu University. Kyushu University Forest, Fukuoka
Itoh T (1995) Anatomical description of Japanese hardwoods I. Wood Res Tech Notes 31:81–181
Itoh T (1996) Anatomical description of Japanese hardwoods II. Wood Res Tech Notes 32:66–176
Itoh T (1997) Anatomical description of Japanese hardwoods III. Wood Res Tech Notes 33:83–201
Itoh T (1998) Anatomical description of Japanese hardwoods IV. Wood Res Tech Notes 34:30–166
Itoh T (1999) Anatomical description of Japanese hardwoods V. Wood Res Tech Notes 35:47–175
Jarbeau JA, Ewers FW, Davis SD (1995) The mechanism of water-stress-induced embolism in two species of chaparral shrubs. Plant Cell Environ 18:189–196
Lechowicz MJ (1984) Why do temperate deciduous trees leaf out at different times? Adaptation and ecology of forest communities. Am Nat 124:821–842
Maherali H, Pockman WT, Jackson RB (2004) Adaptive variation in the vulnerability of woody plants to xylem cavitation. Ecology 85:2184–2199
Miyazawa Y, Kikuzawa K (2005) Winter photosynthesis by saplings of evergreen broad-leaved trees in a deciduous temperate forest. New Phytol 165:857–866
Pittermann J, Sperry J (2003) Tracheid diameter is the key trait determining the extent of freezing-induced embolism in conifers. Tree Physiol 23:907–914
Pratt RB, Ewers FW, Lawson MC, Jacobsen AL, Brediger MM, Davis SD (2005) Mechanisms for tolerating freeze-thaw stress of two evergreen chaparral species: Rhus ovata and Malosma laurina (Anacardiaceae). Am J Bot 92:1102–1113
Sakai A (1980) Freezing resistance of broad-leaved evergreen trees in the warm-temperate zone. Low Temp Sci Ser B 38:1–14
Sano Y, Okamura Y, Utsumi Y (2005) Visualizing water-conduction pathways of living trees: selection of dyes and tissue preparation methods. Tree Physiol 25:269–275
Scholander PF, Love WE, Kanwisher JW (1955) The rise of sap in tall grapevines. Plant Physiol 30:93–104
Sperry JS (1993) Winter xylem embolism and spring recovery in Betula cordifolia, Fagus grandifolia, Abies balsamea and Picea rubens. In: Borghetti M, Grace J, Raschi A (eds) Water transport in plants under climatic stress. Cambridge University, Cambridge, pp 86–98
Sperry JS, Sullivan JEM (1992) Xylem embolism in response to freeze-thaw cycles and water stress in ring-porous, diffuse-porous, and conifer species. Plant Physiol 100:605–613
Sperry JS, Tyree MT (1988) Mechanism of water stress-induced xylem embolism. Plant Physiol 88:581–587
Sperry JS, Holbrook NM, Zimmermann MH, Tyree MT (1987) Spring filling of xylem vessels in wild grapevine. Plant Physiol 83:414–417
Sperry JS, Donnelly JR, Tyree MT (1988) Seasonal occurrence of xylem embolism in sugar maple (Acer saccharum). Am J Bot 75:1212–1218
Sperry JS, Nichols KL, Sullivan JEM, Eastlack SE (1994) Xylem embolism in ring-porous, diffuse-porous, and coniferous trees of northern Utah and interior Alaska. Ecology 75:1736–1752
Suzuki M, Yoda K, Suzuki H (1996) Phenological comparison of the onset of vessel formation between ring-porous and diffuse-porous deciduous trees in a Japanese temperate forest. IAWA J 17:431–444
Taneda H, Tateno M (2005) Hydraulic conductivity, photosynthesis and leaf water balance in six evergreen woody species from fall to winter. Tree Physiol 25:299–306
Tateishi M, Kumagai T, Utsumi Y, Umebayashi T, Shiiba Y, Inoue K, Kaji K, Cho K, Otsuki K (2008) Spatial variations in xylem sap flux density in evergreen oak trees with radial-porous wood: comparisons with anatomical observations. Trees 22:23–30
Thomas H, Sadras VO (2001) The chapter and gratuitous disposal of resources by plants. Funct Ecol 15:3–12
Tyree MT, Cochard H (1996) Summer and winter embolism in oak: impact on water relations. Ann Sci For 53:173–180
Tyree MT, Sperry JS (1989) Vulnerability of xylem to cavitation and embolism. Annu Rev Plant Physiol Mol Biol 40:19–38
Tyree MT, Zimmermann MH (2002) Xylem structure and the ascent of sap, 2nd edn. Springer, Berlin
Tyree MT, Davis SD, Cochard H (1994) Biophysical perspectives of xylem evolution: is there a tradeoff of hydraulic efficiency for vulnerability to dysfunction? IAWA J 15:335–360
Umebayashi T, Utsumi Y, Koga S, Inoue S, Shiiba Y, Arakawa K, Matsumura J, Oda K (2007) Optimal conditions for visualizing water-conducting pathways in a living tree by the dye injection method. Tree Physiol 27:993–999
Umebayashi T, Utsumi Y, Koga S, Inoue S, Fujikawa S, Arakawa K, Matsumura J, Oda K (2008) Conducting pathways in north temperate deciduous broadleaved trees. IAWA J 29:247–263
Utsumi Y, Sano Y (2007) Cryoplaning technique for visualizing the distribution of water in woody tissues by cryoscanning electron microscopy. In: Kuo J (ed) Electron microscopy: methods and protocols, 2nd edn. Humana, New Jersey, pp 497–506
Utsumi Y, Sano Y, Ohtani J, Fujikawa S (1996) Seasonal changes in the distribution of water in the outer growth rings of Fraxinus mandshurica var. japonica: a study by cryo-scanning electron microscopy. IAWA J 17:113–124
Utsumi Y, Sano Y, Funada R, Fujikawa S, Ohtani J (1999) The progression of cavitation in earlywood vessels of Fraxinus mandshurica var japonica during freezing and thawing. Plant Physiol 121:897–904
Utsumi Y, Sano Y, Funada R, Ohtani J, Fujikawa S (2003) Seasonal and perennial changes in the distribution of water in the sapwood of conifers in a sub-frigid zone. Plant Physiol 131:1826–1833
Wheeler EA, Baas P (1991) A survey of the fossil record for dicotyledonous wood and its significance for evolutionary and ecological wood anatomy. IAWA Bull New Ser 12:275–332
Wheeler EA, Baas P (1993) The potentials and limitations of dicotyledonous wood anatomy for climatic reconstructions. Paleobiology 19:487–498
Wheeler EA, Baas P, Gasson PE (1989) IAWA list of microscopic features for hardwood identification: with an appendix on non-anatomical information. IAWA Bull New Ser 10:219–332
Wheeler EA, Baas P, Rodgers S (2007) Variations in dicot wood anatomy: a global analysis based on the insidewood database. IAWA J 28:229–258
Yamauchi F (1976) Anatomical studies of wood in Japanese Elaeagnus. Bull Natl Sci Mus Seri B (Bot) 2:107–118
Acknowledgments
The authors thank I. Murata, T. Inoue, H. Nagasawa, K. Kubota, S. Inoue, S. Osaki, T. Mabuchi, Y. Shiiba, and T. Itoh for their technical support. This work was supported in part by a Grant-in-Aid for Scientific Research (Nos. 17380096 and 19780121) from the Ministry of Education, Science and Culture, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Lovelock.
Rights and permissions
About this article
Cite this article
Umebayashi, T., Utsumi, Y., Koga, S. et al. Xylem water-conducting patterns of 34 broadleaved evergreen trees in southern Japan. Trees 24, 571–583 (2010). https://doi.org/10.1007/s00468-010-0428-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-010-0428-7