Abstract
Background
Eculizumab for the treatment of atypical hemolytic uremic syndrome (HUS) is a standard of care. Central nervous system (CNS) involvement in Shiga toxin–producing Escherichia coli (STEC)-HUS is associated with increased morbidity and mortality. There is no consensus on the use of plasma exchange and/or eculizumab. We report a series (n = 4) of children with CNS involvement in STEC-HUS with excellent outcomes after treatment with eculizumab only and supportive therapies.
Methods
A retrospective chart review of patients with CNS involvement in STEC-HUS is managed with supportive therapies and eculizumab only.
Results
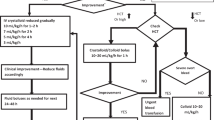
Four patients (75% female) with a median age of 5 years and 11 months (IQR: 23.5–105.5 months) were admitted to a tertiary pediatric nephrology center with CNS involvement in STEC-HUS. Neurological symptoms presented between days 2 and 7 of illness and included ataxia, altered mental status, visual symptoms, and seizures. All had an abnormal MRI brain. All received two doses of eculizumab, 1 week apart (dosing according to weight). Resolution of neurological symptoms was evident at a mean of 60 h post-administration (range: 24–72 h). All patients have complete kidney and neurological recovery at 12-month follow-up.
Conclusion
We present a case series of four children with STEC-HUS and CNS involvement, managed with eculizumab only, in lieu of plasma exchange (as per our previous policy). The marked improvement in symptoms in our cohort supports the use of eculizumab, rather than plasma exchange in the CNS involvement of STEC-HUS.


Similar content being viewed by others
Abbreviations
- AKI:
-
Acute kidney injury
- CNS:
-
Central nervous system
- EEG:
-
Electroencephalogram
- HUS:
-
Haemolytic uremic syndrome
- PCPC:
-
Pediatric cerebral performance category
- PICU:
-
Pediatric intensive care unit
- PLEX:
-
Plasma exchange
- STEC:
-
Shiga toxin–producing Escherichia coli
References
Noris M, Remuzzi G (2005) Hemolytic uremic syndrome. J Am Soc Nephrol 16:1035–1050
Lynn RM, O’Brien SJ, Taylor CM et al (2005) Childhood hemolytic uremic syndrome, United Kingdom and Ireland. Emerg Infect Dis 11:590–596
Costigan C, Raftery T, Carroll AG et al (2022) Neurological involvement in children with hemolytic uremic syndrome. Eur J Pediatr 181:501–512
Khalid M, Andreoli S (2019) Extrarenal manifestations of the hemolytic uremic syndrome associated with Shiga toxin-producing Escherichia coli (STEC HUS). Pediatr Nephrol 34:2495–2507
Brown CC, Garcia X, Bhakta RT, Sanders E, Prodhan P (2021) Severe acute neurologic involvement in children with hemolytic-uremic syndrome. Pediatrics 147:e2020013631
Walsh PR, Johnson S (2019) Eculizumab in the treatment of Shiga toxin haemolytic uraemic syndrome. Pediatr Nephrol 34:1485–1492
Rother RP, Rollins SA, Mojcik CF et al (2007) Discovery and development of the complement inhibitor eculizumab for the treatment of paroxysmal nocturnal hemoglobinuria. Nat Biotechnol 25:1256–1264
Keenswijk W, Raes A, Vande Walle J (2018) Is eculizumab efficacious in Shigatoxin-associated hemolytic uremic syndrome? A narrative review of current evidence. Eur J Pediatr 177:311–318
Lapeyraque AL, Malina M, Fremeaux-Bacchi V et al (2011) Eculizumab in severe Shiga-toxin-associated HUS. N Engl J Med 364:2561–2563
Loos S, Ahlenstiel T, Kranz B et al (2012) An outbreak of Shiga toxin-producing Escherichia coli O104:H4 hemolytic uremic syndrome in Germany: presentation and short-term outcome in children. Clin Infect Dis 55:753–759
Bauer A, Loos S, Wehrmann C et al (2014) Neurological involvement in children with E. coli O104:H4-induced hemolytic uremic syndrome. Pediatr Nephrol 29:1607–1615
Gitiaux C, Krug P, Grevent D et al (2013) Brain magnetic resonance imaging pattern and outcome in children with haemolytic-uraemic syndrome and neurological impairment treated with eculizumab. Dev Med Child Neurol 55:758–765
Ekinci Z, Kenan Bek K, Aytac MB, Karadenizli A, Hancer VS (2014) Renal outcome with eculizumab in two diarrhea-associated hemolytic ureamic syndrome cases with severe neurologic involvement. Hong Kong J Nephrol 16:46–49
Saini A, Emke A, Silva MC, Perlman SJ (2015) Response to eculizumab in Escherichia coli O157:H7 induced hemolytic uremic syndrome with severe neurological manifestations. Clin Pediatr (Phila) 54:387–389
Pape L, Hartmann H, Bange FC, Suerbaum S, Bueltmann E, Ahlenstiel-Grunow T (2015) Eculizumab in typical hemolytic uremic syndrome (HUS) With neurological involvement. Medicine (Baltimore) 94:e1000
Percheron L, Gramada R, Tellier S et al (2018) Eculizumab treatment in severe pediatric STEC-HUS: a multicenter retrospective study. Pediatr Nephrol 33:1385–1394
Monet-Didailler C, Chevallier A, Godron-Dubrasquet A et al (2020) Outcome of children with Shiga toxin-associated haemolytic uraemic syndrome treated with eculizumab: a matched cohort study. Nephrol Dial Transplant 35:2147–2153
Weber B, Chan D, Hammer S (2022) Eculizumab use in a temporarily dialysis-dependent patient with Shiga toxin–producing Escherichia coli hemolytic uremic syndrome with neurological complications. J Pediatr Pharmacol Ther 27:90–95
HSE Health Protection Surveillance Centre (2019) VTEC infection in Ireland, 2017. HSE HPSC, Dublin
Garnier A, Brochard K, Kwon T et al (2023) Efficacy and safety of eculizumab in pediatric patients affected by Shiga toxin-related hemolytic and uremic syndrome: a randomized, placebo-controlled trial. J Am Soc Nephrol. https://doi.org/10.1681/ASN.0000000000000182
Fiser DH, Tilford JM, Roberson PK (2000) Relationship of illness severity and length of stay to functional outcomes in the pediatric intensive care unit: a multi-institutional study. Crit Care Med 28:1173–1179
Acknowledgements
The authors would like to thank the other members of our research group who were involved with the care of these patients and in the creation of this article (Dr. Niamh Dolan, Prof. Bryan Lynch, Dr. Michael Riordan, and Dr. Maria Stack).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Prof. Awan previously served on the medical advisory board of Alexion. All authors have no conflicts, of a financial nature or otherwise, to declare.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wildes, D.M., Harvey, S., Costigan, C.S. et al. Eculizumab in STEC-HUS: a paradigm shift in the management of pediatric patients with neurological involvement. Pediatr Nephrol 39, 315–324 (2024). https://doi.org/10.1007/s00467-023-06102-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-06102-w