Abstract
Background
Hypercalciuria and hypocitraturia are considered the most important risk factors for urolithiasis. Citrate binds to urinary calcium to form a soluble complex which decreases the availability of ionized calcium (Ca2+) necessary for calcium oxalate formation and phosphate crystallization. The aims of this study were to assess the Ca2+ fraction in relation to total calciuria, citraturia and urinary pH and to determine whether urinary Ca2+ concentration is a helpful biomarker in metabolic evaluation of children with urolithiasis.
Methods
We collected 24-h urine samples from 123 stone-forming children and adolescents with hypocitraturia and from 424 healthy controls. Total calciuria (total calcium, Catotal), Ca2+, pH, citrate, oxalate and Bonn Risk Index (BRI) were assessed and compared between the two groups.
Results
Total calciuria and Ca2+ content were higher in stone-formers than in the healthy children. In both stone-formers and controls, Ca2+ content was inversely related to citraturia and urinary pH, whereas the Ca2+/Catotal ratio differed slightly between the groups. A large variability in Ca2+ level was found across individuals in both groups. The BRI increased with increasing calciuria and urine acidity.
Conclusions
Compared to controls, stone-formers with hypocitraturia demonstrated a higher urinary Ca2+ concentration, but this was proportional to calciuria. The large individual variability in urinary Ca2+ content limits its practical use in metabolic evaluation of children with urolithiasis. However, the Ca/Citrate ratio may be a useful clinical tool in evaluating children with urolithiasis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urinary calcium, one of the most important promoters of kidney stone formation, appears in urine in both bound and free states. A variety of small organic and inorganic negatively charged molecules can form soluble (e.g. with citrate) or insoluble (e.g. with oxalate or phosphate) complexes with calcium. A small amount of urinary calcium may also be bound to macromolecular amino acid residues of urinary proteins [1–3]. Only a certain, but individually variable, proportion of urinary calcium exists as ionized calcium (Ca2+). The more urinary calcium that is fixed in soluble and stable complexes the lower the probability of formation of insoluble calcium salts from the particular urine composition.
Although the urinary concentration of Ca2+ is regarded as one of the most important and pathophysiologically relevant parameters in the process of calcium stone formation, standard metabolic assessment in stone-forming patients includes only urinalysis of total calcium (calciuria, Catotal) and/or the calcium to creatinine ratio as opposed to serum samples in which the free calcium concentration is routinely assayed [3]. In contrast, low urinary citrate excretion has been considered a major risk factor for calcium stone formation. Citrate creates strong but soluble complexes with calcium, thereby effectively reducing the urinary concentration of Ca2+. Lowering the availability of Ca2+ reduces the nucleation and, as a consequence, the crystallization rate of both calcium oxalate (CaOx) and calcium phosphate [4, 5].
The urinary concentration of Ca2+ as well as renal citrate excretion are largely pH-dependent. Whereas urinary pH and Ca2+ are inversely related [6, 7], both systemic and urinary acidosis may inhibit renal citrate excretion if citrate reabsorption and metabolism in the proximal tubule become up-regulated in order to counteract imbalanced acid–base status [1, 8].
Despite the fact that the mechanisms of citrate action are quite well recognized, to date the statistical relationships between urinary Ca2+, total calciuria, citrate excretion and urinary pH have not been investigated. The aims of this study were, first, to investigate the above interrelationships in the context of the risk of CaOx crystallization and, secondly, to determine whether the measurement of urinary Ca2+ concentration is useful in the metabolic evaluation of children and adolescents with urolithiasis. We hypothesized that Ca2+ concentration in urine was a linear function of urinary citrate level.
Methods
This observational cross-sectional study was undertaken in the Department of Pediatric Nephrology, University Children’s Hospital in Bialystok between 2002 and 2011. The study group consisted of 123 children and adolescents (66 boys, 57 girls) aged 3–18 (median 15.1) years with urinary calcareous stones of different composition, in whom hypocitraturia was diagnosed during the first metabolic evaluation of 24-h urine samples. Hypocitraturia was defined as urinary citrate excretion of less than 400 mg/g creatinine/24 h [9]. In all cases, urolithiasis was confirmed by means of ultrasonography, and in half of the cases, also by X-ray. In 36 patients (30 %), stone analysis confirmed the composition of urinary stones with a predominance of CaOx (29 patients) or calcium phosphate (7 patients) (DiaSys, Diagnostic Systems GmbH, Holzheim, Germany), whereas in the remaining patients stone analyses were unavailable.
The exclusion criteria included acute infection and inadequate 24-h urine collection assessed with urine creatinine excretion (normal 15–25 mg/kg/24 h).
The control group comprised 424 healthy children and adolescents (212 boys, 212 girls), aged 3–18 (median 14.6 years) years, with normal urinary excretion levels of citrate, calcium, oxalate, uric acid and cystine, and without abnormalities in dipstick urinalysis (Bayer Diagnostic, Bridgend, UK). These children were mainly volunteers with a past history of primary nocturnal enuresis or inguinal hernia who had been approached to participate while attending the academic center or the children of hospital staff. None of the controls reported a family history of urolithiasis, while their recent screening based on ultrasonography excluded urinary stones.
All study subjects were evaluated using the same standard protocol which also included the request to continue to follow their customary diet. During the 24-h urine collection, urine was stored in sterile, closed containers at 4 °C without the addition of preservatives, and all measurements were conducted within 4 h after the end of the collection period. Urinary citrate concentrations were determined by an enzymatic method using a commercial set (Boehringer Mannheim/R-Biopharm, Germany) and adjusted for creatinine. Urinary calcium and creatinine were assessed with the Cobas-Integra 800 analyzer and Roche reagents (Roche, Indianapolis, IN). Hypercalciuria was diagnosed for calcium excretions exceeding 4 mg/kg/24 h [9]. The urinary concentration of Ca2+ was measured using calcium ion selective electrodes (Rapidlab 855; Bayer, Leverkusen, Germany). Calibration and the quality assurance procedure, based on the calibration curves, were carried out every day. The corresponding value for Cabound was calculated as the difference between Catotal and Ca2+. Urinary oxalate was examined using standard enzymatic–spectrophotometric methodology (product nr. 591D; Trinity Biotech, Berkeley Heights, NJ). Hyperoxaluria was diagnosed when urinary oxalate excretion exceeded 0.5 mmol/1.73 m2/24 h [9]. Urinary pH was determined using a microcomputer pH meter (model CP-315M; Elmetron, Zabrze, Poland).
The Bonn Risk Index (BRI) is a measure of urinary CaOx stone formation risk and was studied according to the method of Laube [10], using a modified analytical system described elsewhere [11]. In brief, after determination of urinary Ca2+ content, the urine was titrated step-by-step with Ox2- by a computer-operated analytical system. At the moment crystallized particles of CaOx caused a 2 % decrease in light transmission, the computer application automatically stopped the titration process. The BRI value was then calculated as BRI = Ca2+:Ox2-.
Renal ultrasonography was carried out using high-resolution equipment (Toshiba SSH-140A; probe Convex 3.75 MHz; Toshiba, Japan) by a trained person.
Both parents and children older than 15 years of age gave informed consent for the study. The study was approved by the ethics committee of the Medical University of Bialystok.
Statistical analysis
Statistical analyses were performed using Statistica®, ver. 9.0 PL (StatSoft, Tulsa, OK). The Mann–Whitney U test was used for comparisons between two independent parameters, and the correlations were made with Spearman test. A p value of <0.05 was considered to be statistically significant.
Results
The characteristics of the urolithiasis patients with hypocitraturia and healthy controls are shown in Table 1. With respect to urinary pH, stone-formers and controls showed nearly identical median values of 6.31 and 6.33, respectively. The median values of Ca2+ and Catotal were higher in the patients than in the healthy controls, although the difference was significant only in relation to Ca2+ (p < 0.001 and p = 0.23, respectively). We found a significant difference in Ca2+/citrate quotient between the groups. Median Ca2+/citrate was fourfold higher in stone-formers than in controls (0.39 vs. 0.10; p < 0.001). The patients’ median BRI value was 1.02 l−1, which was more than twice as high as that of the controls (p < 0.001).
Figure 1a–d shows scatter plots of urinary concentrations and excretions of Ca2+, Catotal, citrate and oxalate in various combinations, which compare the data of hypocitraturic stone-formers (citrate <400 mg/g creatinine) and normocitraturic controls. The influence of pH on data distribution was investigated by separately plotting the data of urine samples below and above the median urinary pH of each group.
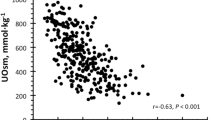
Scatter plots of urinary concentration and excretion of ionized (Ca 2+), total calcium ( calciuria; Ca total ), citrate and oxalate in various combinations. a Ca2+ vs. citrate excretion in urine, b Ca2+to Catotal ratio vs. citrate excretion in urine, c oxalate vs. Ca2+/citrate excretion ratio in urine, d Bonn Risk Index (BRI) vs. citrate excretion in urine. The influence of pH on the data distribution is investigated by separately plotting the data of urine samples below and above the median urinary pH of each group. Dotted lines show limit values for hypocitraturia (a, b, d), hyperoxaluria (c) and BRI (d)
Figure 1a shows a plot of the 24-h urinary concentration of Ca2+ versus citrate excretion. The Ca2+ concentration in urine showed a declining trend with both increasing urinary citrate and alkalinization of urine. In stone-formers, the parameters were citrate 273.4 mg/g creatinine and Ca2+ 0.51 mmol/l versus citrate 288.8 mg/g creatinine and Ca2+ 0.36 mmol/l at low and high pH, respectively. In the controls, the parameters were citrate 559.5 mg/g creatinine and Ca2+ 0.35 mmol/l versus citrate 631.9 mg/g creatinine and Ca2+ 0.28 mmol/l at low and high pH, respectively.
Figure 1b presents a plot of Ca2+/Catotal versus citraturia. Interestingly, citrate influenced the Ca2+/Catotal ratio only to a small extent (stone-formers 0.20 vs. controls 0.14; p = 0.15). In both groups, the Ca2+/Catotal ratio slightly increased with increasing acidity of the urine, showing high individual variability. The intra-group influence of pH on Ca2+/Catotal was 0.18 versus 0.14 at low and high pH, respectively, in stone-formers and 0.16 versus 0.14 at low and pH, respectively, in the controls.
Figure 1c shows the plot of oxaluria versus the Ca2+/citrate excretion ratio. Compared to the controls, the data for stone-formers showed wide scattering. Hyperoxaluria was only observed in stone-formers. The median Ca2+/citrate excretion ratio was fourfold higher in stone-formers than in controls (0.39 vs. 0.10; p < 0.001), due to the higher urinary Ca2+ and lower urinary citrate concentrations in stone-formers. The intra-group influence of urinary pH on the Ca2+/citrate ratio was greater in stone-formers than in controls (low vs. high pH: stone-formers, 0.37 vs. 0.25; controls, 0.12 vs. 0.09).
The relationship between BRI values and urinary citrate levels are shown in Fig. 1d. Both groups demonstrated median BRI values below the cut-off value for children of 1.93 l−1 [12]. BRI values of >1.93 l−1 were observed only in the stone-former group. The intra-group influence of urinary pH on BRI was remarkable in stone-formers but smaller in controls (low vs. high pH: stone-formers, BRI 1.36 vs. BRI 0.49; controls, BRI 0.52 vs. BRI 0.39). However, there were some stone-formers who had a low BRI.
Discussion
Hypocitraturia in children is usually defined as 24-h citrate excretion of <400 mg/g creatinine [9]. Other definitions are more restrictive by setting the urine citrate level at <180 mg/g creatinine regardless of gender [13] or <125 mg/g creatinine in boys and <300 mg/g creatinine in girls [14].
Hypocitraturia can be a concomitant effect to compensate for metabolic acidosis due to a number of possible causes, such as complete and incomplete distal renal tubular acidosis, hypokalemia, severe bowel dysfunction, high protein diet, urinary tract infection and many other factors. However, the majority of hypocitraturia cases are thought to be idiopathic [15]. None of the patients studied here had secondary causes of hypocitraturia, such as hypokalemia, chronic diarrhea, inflammatory bowel diseases or a complete renal tubular acidosis. However, the occurrence of partial renal tubular acidosis as a potential cause of hypocitraturia could not be definitely excluded; unfortunately, the clinical picture of partial renal tubular acidosis is less evident [16], and a screening for differences in the urine–blood (U–B) carbon dioxide partial pressure (PCO2) could not be conducted for all participants [17]. One of the possible causes of hypocitraturia in our patients, particularly among the adolescents, could have been a high intake of animal protein [18] which is customary in Poland. However, a causative link could not be proven. Thus, we assume that the observed hypocitraturia was idiopathic in most cases.
Several earlier studies that investigated pediatric kidney stone disease reported that hypocitraturia was the most common accompanying metabolic abnormality, found in up to 63 % of patients [19, 20]. This was particularly true when the high “cut-off” value of hypocitraturia (400 mg/g creatinine) was used. This value was recently disputed by Srivastava et al. [13] who demonstrated 180 mg/g as a cut-off point for differentiation of hypocitraturia. However, for the purpose our study, we did not think that this difference in cut-off values would play a role.
In our study, the magnitude of calciuria was not significantly different in the urolithiasis group when compared to controls, but the Ca2+ level was significantly higher in stone-formers, presumably due to hypocitraturia. However, the gradient of the regression line in the plot of urinary Ca2+/Catotal versus citraturia was very low, and the median values of the ratio did not differ significantly between the groups [0.20 (patients) vs. 0.14 (healthy controls), p = 0.15] (Table 1, Fig. 1b). In other words, the binding capacity of Ca2+ was almost proportional to the calciuria rate in both groups. Simultaneously, the Ca2+ concentration showed a high variability between individuals, regardless of the respective urinary citrate level, with a 5–95 % range of 0.12–1.20 mmol/l in urolithiasis patients and 0.14–0.69 mmol/l in controls (Table 1). This variability is at least partly caused by similar variations in calciuria range (Table 1) and differences in urinary pH, with higher values of Ca2+ in the subgroups associated with more acidic urines (Fig. 1a). Therefore, we can infer that the absolute values of urinary Ca2+ mainly depend on Catotal and on urinary pH.
Our observation of the effect of urine pH on urinary Ca2+ concentration is supported by the results of other researchers who found an increase in urinary pH to be the main determining factor of therapeutic action of potassium citrate in low urinary citrate urolithiasis [2, 21–23]. However, alkalinizing the urine too much may increase the risk of calcium phosphate crystal formation [24]. Additionally, at high urinary pH, the fraction of triple-negatively charged citrate increases, thereby increasing the overall inhibitory effect of citrate as more calcium ions can be bound [25].
The extent of urinary citrate excretion also depends on the balance of the body’s acid–base status. Systemic, intracellular and urinary acidosis decrease renal citrate excretion, mainly by up-regulation of the citrate reabsorption process within the proximal tubule [1, 8, 15]. Thus, we could theoretically expect a lower urine pH in the hypocitraturic participants of the study. On the other hand, other studies have revealed that the urine of stone-formers tends to show a higher mean pH value compared to that of healthy controls [21, 22]. In our study, the median urinary pH of healthy controls and hypocitraturic stone-formers was nearly identical. Whether this paradoxical association was the cause or the effect of urinary citrate level needs further elucidation.
We found a significant difference in the Ca2+/citrate quotient between the groups. The median Ca2+/citrate value was fourfold higher in stone-formers than in controls (0.39 vs. 0.10, respectively; p < 0.001). This may have resulted from higher urinary Ca2+ as well as from lower urinary citrate levels in the stone-formers. There was also an explicitly pronounced difference in total the calcium/citrate ratio between the groups. Our findings support previous reports that acknowledged this ratio as a practical risk factor for stone formation and a good discriminating biomarker between stone-formers and healthy individuals [13, 26].
We provide evidence of a high influence of urinary pH on the risk of CaOx crystallization measured with BRI (Fig. 1d). The BRI was about threefold greater in the subgroup of stone-formers with urinary pH below the median than in the subgroup with pH above median (1.36 vs. 0.49 l−1, respectively). This was partly a consequence of the higher Ca2+ in the ‘low’ pH conditions (0.51 vs. 0.36 mmol/l) (Fig. 1a). The dependence of BRI on urinary pH was not so pronounced in the controls. To our surprise, there were quite a large number of stone-formers with low BRI. Presumably, CaOx precipitation in vivo could have occurred in these individuals and thereby could have lowered the post-renal concentration of both Ca2+ and the other stone-forming constituents [27]. These pre-analytical factors are unavoidable and might lead to a lowering of BRI values.
We are aware of several limitations to our study. The relationships between Ca2+, calciuria and citrate excretion were studied in only one sample of urine within a certain range of urinary pH (5–95 % pH range: 5.63–7.08), and the results cannot be generalized beyond this range. Additionally, there are molecules other than citrate that form complexes with calcium and possibly reduce urinary Ca2+; however, these were not investigated within the framework of our study. Furthermore, there was a slight difference in age between the groups. However, this did not detract from the main findings of the study.
In summary, stone-formers with hypocitraturia have a higher Ca2+ concentration than healthy children. The results of our study support the hypothesis that Ca2+ concentration in the urine is inversely correlated with citraturia; however, this relationship appears, at least partly, to be dependent on total calciuria. The BRI, which reflects the risk of CaOx crystallization, increases with increased calciuria and urine acidification. The large variability in the proportions of urinary Ca2+ and bound calcium calls into question the importance of standard Ca2+ measurements in clinical practice at our current stage of knowledge. On the other hand, the Ca/citrate ratio may be a useful clinical tool in evaluating children with urolithiasis.
References
Simpson DP (1983) Citrate excretion: a window on renal metabolism. Am J Physiol 1244:F223–F234
Rodgers A, Allie-Hamdulay S, Jackson G (2006) Therapeutic action of citrate in urolithiasis explained by chemical speciation: increase in pH is the determinant factor. Nephrol Dial Transplant 21:361–369
Baird GS (2011) Ionized calcium. Clin Chim Acta 12:696–701
Pak CY (1994) Citrate and renal calculi: an update. Miner Electrolyte Metab 20:371–377
Nicar MJ, Hill K, Pak CYC (1987) Inhibition by citrate of spontaneous precipitation of calcium oxalate in vitro. J Bone Miner Res 2:215–220
Langley SEM, Fry CH (1997) The influence of pH on urinary ionized [Ca2+]: differences between urinary stone formers and normal subjects. Br J Urol 79:8–14
Halperin ML, Cheema Dhadli S, Kamel KS (2006) Physiology of acid–base balance: links with kidney stone prevention. Semin Nephrol 26:441–446
Moe YW, Preisig PA (2006) Dual role of citrate in mammalian urine. Curr Opin Nephrol Hypertens 15:419–424
Stapleton FB, Kroovand RL (1996) Stones in childhood. In: Coe FL, Favus MJ, Pak CYC, Parks JH, Preminger GM (eds) Kidney stones: medical and surgical management. Lippincott-Raven, Philadelphia, pp 1065–1080
Laube N, Schneider A, Hesse A (2000) A new approach to calculate the risk of calcium oxalate crystallization from unprepared native urine. Urol Res 28:274–280
Porowski T, Mrozek P, Sidun J, Zoch-Zwierz W, Konstantynowicz J, Kirejczyk JK, Motkowski R, Laube N (2010) Bonn Risk Index based micromethod for assessing risk of urinary calcium oxalate stone formation. J Urol 183:1157–1162
Porowski T, Zoch-Zwierz W, Wasilewska A, Spotyk A, Konstantynowicz J (2007) Normative data on the Bonn Risk Index for calcium oxalate crystallization in healthy children. Pediatr Nephrol 22:514–520
Srivastava T, Winston MJ, Auron A, Alon US (2009) Urine calcium/citrate ratio in children with hypercalciuric stones. Pediatr Res 66:85–90
Norman ME, Feldman NI, Cohn RM, Roth KS, McCurdy DK (1978) Urinary citrate excretion in the diagnosis of distal renal tubular acidosis. J Pediatr 92:394–400
Zuckerman JM, Assimos DG (2009) Hypocitraturia: pathophysiology and medical management. Rev Urol 11:134–144
Hess B, Michel R, Takkinen R, Ackermann D, Jaeger P (1994) Risk factors for low urinary citrate in calcium nephrolithiasis: low vegetable fibre intake and low urine volume to be added to the list. Nephrol Dial Transplant 9:642–649
Alon U, Hellerstein S, Warady BA (1991) Oral acetazolamide in the assessment of (urine-blood) PCO2. Pediatr Nephrol 5:307–311
Reddy ST, Wang CY, Sakhaee K, Brinkley L, Pak CY (2002) Effect of low-carbohydrate high-protein diets on acid–base balance, stone-forming propensity, and calcium metabolism. Am J Kidney Dis 40:265–274
Tefekli A, Esen T, Ziylan O, Erol B, Armagan A, Ander H, Akinci M (2003) Metabolic risk factors in pediatric and adult calcium oxalate stone formers: is there any differences? Urol Int 70:273–277
VanDervoort K, Wiesen J, Frank R, Vento S, Crosby V, Chandra M, Trachtman H (2007) Urolithiasis in pediatric patients: a single center study of incidence, clinical presentation and outcome. J Urol 177:2300–2305
Langley SE, Fry CH (1995) Differences in the free Ca2+ in undiluted urine from stone formers and normal subjects using a new generation of ion-selective electrodes. Br J Urol 75:288–295
Thode J, Holgersen RB, Gerstenberg T (1993) Variability of the calcium ion activity with pH in stone-forming and non-stone-forming urine. Scand J Clin Lab Invest 214[Suppl]:121–126
Kang DE, Maloney MM, Haleblian GE, Springhart WP, Honeycutt EF, Eisenstein EL, Marguet CG, Preminger GM (2007) Effect of medical management on recurrent stone formation following percutaneous nephrolithotomy. J Urol 177:1785–1788
Cameron MA, Sakhaee K, Moe OW (2005) Nephrolithiasis in children. Pediatr Nephrol 20:1587–1592
Laube N, Jansen B, Hesse A (2002) Citric acid or citrates in urine: which should we focus on in the prevention of calcium oxalate crystals and stones? Urol Res 30:336–341
DeFoor W, Minevich E, Jackson E, Reddy P, Clark C, Sheldon C, Asplin J (2008) Urinary metabolic evaluation in solitary and recurrent stone forming children. J Urol 179:2369–2372
Laube N, Pullmann M, Hergarten S, Hesse A (2003) Influence of urinary stones on the composition of a 24-hour urine sample. Clin Chem 49:281–285
Acknowledgments
This study was supported by a research grant from the Medical University of Bialystok (No. 113-41702L).
Author information
Authors and Affiliations
Corresponding author
Additional information
Tadeusz Porowski and Jan K. Kirejczyk contributed equally to this manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Porowski, T., Kirejczyk, J.K., Konstantynowicz, J. et al. Correspondence between Ca2+ and calciuria, citrate level and pH of urine in pediatric urolithiasis. Pediatr Nephrol 28, 1079–1084 (2013). https://doi.org/10.1007/s00467-013-2420-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2420-0