Abstract
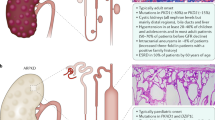
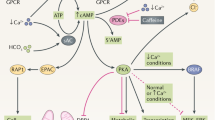
Autosomal dominant polycystic kidney disease (ADPKD) is the most common monogenic cause of kidney failure. It is a systemic disorder, not only affecting the kidneys, but also associated with cyst formation in other organs such as the liver, spleen, pancreas, and seminal vesicles. Other extra-renal symptoms may consist of intracranial arterial aneurysms, cardiac valvular defects, abdominal and inguinal hernias and colonic diverticulosis. Very little is known regarding bone involvement in ADPKD; however, recent evidence has revealed the potential role of fibroblast growth factor 23 (FGF23). FGF23 is an endocrine fibroblast growth factor acting in the kidney as a phosphaturic hormone and a suppressor of active vitamin D with key effects on the bone/kidney/parathyroid axis, and has been shown to increase in patients with ADPKD, even with normal renal function. The aim of this review is to provide an overview of bone and mineral abnormalities found in experimental models and in patients with ADPKD, and to discuss the possible role of FGF23 in this disease.
Similar content being viewed by others
Abbreviations
- ADPKD:
-
Autosomal dominant polycystic kidney disease
- [Ca2+]cyt :
-
Cytosolic Ca2+ concentration
- CKD:
-
Chronic kidney disease
- 1,25(OH)2D:
-
1,25-dihydroxyvitamin D
- FGF23:
-
Fibroblast growth factor 23
- GFR:
-
Glomerular filtration rate
- PC1:
-
Polycystin-1
- PC2:
-
Polycystin-2
- TmP/GFR:
-
Tubular maximum for phosphate corrected for the glomerular filtration rate
References
Gabow PA (1993) Autosomal dominant polycystic kidney disease. N Engl J Med 329:332–342
Harris PC, Torres VE (2009) Polycystic kidney disease. Annu Rev Med 60:321–337
Torres VE, Harris PC (2009) Autosomal dominant polycystic kidney disease: the last 3 years. Kidney Int 76:149–168
Wolf M (2012) Update on fibroblast growth factor 23 in chronic kidney disease. Kidney Int 82:737–747
Pavik I, Jaeger P, Kistler AD, Poster D, Krauer F, Cavelti-Weder C, Rentsch KM, Wuthrich RP, Serra AL (2011) Patients with autosomal dominant polycystic kidney disease have elevated fibroblast growth factor 23 levels and a renal leak of phosphate. Kidney Int 79:234–240
Pavik I, Jaeger P, Ebner L, Poster D, Krauer F, Kistler AD, Rentsch K, Andreisek G, Wagner CA, Devuyst O, Wuthrich RP, Schmid C, Serra AL (2012) Soluble klotho and autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol 7:248–257
Cheong B, Muthupillai R, Rubin MF, Flamm SD (2007) Normal values for renal length and volume as measured by magnetic resonance imaging. Clin J Am Soc Nephrol 2:38–45
Chapman AB, Schrier RW (1991) Pathogenesis of hypertension in autosomal dominant polycystic kidney disease. Semin Nephrol 11:653–660
Chapman AB, Johnson AM, Rainguet S, Hossack K, Gabow P, Schrier RW (1997) Left ventricular hypertrophy in autosomal dominant polycystic kidney disease. J Am Soc Nephrol 8:1292–1297
Sedman A, Bell P, Manco-Johnson M, Schrier R, Warady BA, Heard EO, Butler-Simon N, Gabow P (1987) Autosomal dominant polycystic kidney disease in childhood: a longitudinal study. Kidney Int 31:1000–1005
Schrier RW (2009) Renal volume, renin-angiotensin-aldosterone system, hypertension, and left ventricular hypertrophy in patients with autosomal dominant polycystic kidney disease. J Am Soc Nephrol 20:1888–1893
Gabow PA, Chapman AB, Johnson AM, Tangel DJ, Duley IT, Kaehny WD, Manco-Johnson M, Schrier RW (1990) Renal structure and hypertension in autosomal dominant polycystic kidney disease. Kidney Int 38:1177–1180
Kelleher CL, McFann KK, Johnson AM, Schrier RW (2004) Characteristics of hypertension in young adults with autosomal dominant polycystic kidney disease compared with the general U.S. population. Am J Hypertens 17:1029–1034
Fick GM, Johnson AM, Hammond WS, Gabow PA (1995) Causes of death in autosomal dominant polycystic kidney disease. J Am Soc Nephrol 5:2048–2056
Torres VE, Donovan KA, Scicli G, Holley KE, Thibodeau SN, Carretero OA, Inagami T, McAteer JA, Johnson CM (1992) Synthesis of renin by tubulocystic epithelium in autosomal-dominant polycystic kidney disease. Kidney Int 42:364–373
Loghman-Adham M, Soto CE, Inagami T, Cassis L (2004) The intrarenal renin-angiotensin system in autosomal dominant polycystic kidney disease. Am J Physiol Ren Physiol 287:F775–F788
Gabow PA (1990) Autosomal dominant polycystic kidney disease–more than a renal disease. Am J Kidney Dis 16:403–413
Bae KT, Zhu F, Chapman AB, Torres VE, Grantham JJ, Guay-Woodford LM, Baumgarten DA, King BF Jr, Wetzel LH, Kenney PJ, Brummer ME, Bennett WM, Klahr S, Meyers CM, Zhang X, Thompson PA, Miller JP (2006) Magnetic resonance imaging evaluation of hepatic cysts in early autosomal-dominant polycystic kidney disease: the Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease cohort. Clin J Am Soc Nephrol 1:64–69
Grantham JJ (2008) Clinical practice. Autosomal dominant polycystic kidney disease. N Engl J Med 359:1477–1485
Brun M, Maugey-Laulom B, Eurin D, Didier F, Avni EF (2004) Prenatal sonographic patterns in autosomal dominant polycystic kidney disease: a multicenter study. Ultrasound Obstet Gynecol 24:55–61
Fick-Brosnahan G, Johnson AM, Strain JD, Gabow PA (1999) Renal asymmetry in children with autosomal dominant polycystic kidney disease. Am J Kidney Dis 34:639–645
Schrier RW, Johnson AM, McFann K, Chapman AB (2003) The role of parental hypertension in the frequency and age of diagnosis of hypertension in offspring with autosomal-dominant polycystic kidney disease. Kidney Int 64:1792–1799
Rizk D, Chapman A (2008) Treatment of autosomal dominant polycystic kidney disease (ADPKD): the new horizon for children with ADPKD. Pediatr Nephrol 23:1029–1036
Sharp C, Johnson A, Gabow P (1998) Factors relating to urinary protein excretion in children with autosomal dominant polycystic kidney disease. J Am Soc Nephrol 9:1908–1914
Mekahli D, Woolf AS, Bockenhauer D (2010) Similar renal outcomes in children with ADPKD diagnosed by screening or presenting with symptoms. Pediatr Nephrol 25:2275–2282
Selistre L, de Souza V, Ranchin B, Hadj-Aissa A, Cochat P, Dubourg L (2012) Early renal abnormalities in children with postnatally diagnosed autosomal dominant polycystic kidney disease. Pediatr Nephrol 27:1589–1593
Bergmann C, Bruchle NO, Frank V, Rehder H, Zerres K (2008) Perinatal deaths in a family with autosomal dominant polycystic kidney disease and a PKD2 mutation. N Engl J Med 359:318–319
Michaud EJ, Yoder BK (2006) The primary cilium in cell signaling and cancer. Cancer Res 66:6463–6467
Praetorius HA, Spring KR (2005) A physiological view of the primary cilium. Annu Rev Physiol 67:515–529
Ou G, Blacque OE, Snow JJ, Leroux MR, Scholey JM (2005) Functional coordination of intraflagellar transport motors. Nature 436:583–587
Qin H, Burnette DT, Bae YK, Forscher P, Barr MM, Rosenbaum JL (2005) Intraflagellar transport is required for the vectorial movement of TRPV channels in the ciliary membrane. Curr Biol 15:1695–1699
Rohatgi R, Snell WJ (2010) The ciliary membrane. Curr Opin Cell Biol 22:541–546
Hu Q, Milenkovic L, Jin H, Scott MP, Nachury MV, Spiliotis ET, Nelson WJ (2010) A septin diffusion barrier at the base of the primary cilium maintains ciliary membrane protein distribution. Science 329:436–439
Goetz SC, Anderson KV (2010) The primary cilium: a signalling centre during vertebrate development. Nat Rev Genet 11:331–344
Wang X, Ward CJ, Harris PC, Torres VE (2010) Cyclic nucleotide signaling in polycystic kidney disease. Kidney Int 77:129–140
Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J (2003) Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet 33:129–137
Praetorius HA, Spring KR (2003) The renal cell primary cilium functions as a flow sensor. Curr Opin Nephrol Hypertens 12:517–520
Praetorius HA, Spring KR (2001) Bending the MDCK cell primary cilium increases intracellular calcium. J Membr Biol 184:71–79
Xiao ZS, Quarles LD (2010) Role of the polycytin-primary cilia complex in bone development and mechanosensing. Ann N Y Acad Sci 1192:410–421
AbouAlaiwi WA, Takahashi M, Mell BR, Jones TJ, Ratnam S, Kolb RJ, Nauli SM (2009) Ciliary polycystin-2 is a mechanosensitive calcium channel involved in nitric oxide signaling cascades. Circ Res 104:860–869
Nauli SM, Kawanabe Y, Kaminski JJ, Pearce WJ, Ingber DE, Zhou J (2008) Endothelial cilia are fluid shear sensors that regulate calcium signaling and nitric oxide production through polycystin-1. Circulation 117:1161–1171
Boulter C, Mulroy S, Webb S, Fleming S, Brindle K, Sandford R (2001) Cardiovascular, skeletal, and renal defects in mice with a targeted disruption of the Pkd1 gene. Proc Natl Acad Sci U S A 98:12174–12179
Voets T, Nilius B (2009) TRPCs, GPCRs and the Bayliss effect. EMBO J 28:4–5
Brayden JE, Earley S, Nelson MT, Reading S (2008) Transient receptor potential (TRP) channels, vascular tone and autoregulation of cerebral blood flow. Clin Exp Pharmacol Physiol 35:1116–1120
Sharif-Naeini R, Folgering JH, Bichet D, Duprat F, Lauritzen I, Arhatte M, Jodar M, Dedman A, Chatelain FC, Schulte U, Retailleau K, Loufrani L, Patel A, Sachs F, Delmas P, Peters DJ, Honore E (2009) Polycystin-1 and −2 dosage regulates pressure sensing. Cell 139:587–596
Nilius B (2009) Polycystins under pressure. Cell 139:466–467
Xiao Z, Zhang S, Mahlios J, Zhou G, Magenheimer BS, Guo D, Dallas SL, Maser R, Calvet JP, Bonewald L, Quarles LD (2006) Cilia-like structures and polycystin-1 in osteoblasts/osteocytes and associated abnormalities in skeletogenesis and Runx2 expression. J Biol Chem 281:30884–30895
Xiao Z, Zhang S, Magenheimer BS, Luo J, Quarles LD (2008) Polycystin-1 regulates skeletogenesis through stimulation of the osteoblast-specific transcription factor RUNX2-II. J Biol Chem 283:12624–12634
Xiao Z, Dallas M, Qiu N, Nicolella D, Cao L, Johnson M, Bonewald L, Quarles LD (2011) Conditional deletion of Pkd1 in osteocytes disrupts skeletal mechanosensing in mice. FASEB J 25:2418–2432
Lu W, Shen X, Pavlova A, Lakkis M, Ward CJ, Pritchard L, Harris PC, Genest DR, Perez-Atayde AR, Zhou J (2001) Comparison of Pkd1-targeted mutants reveals that loss of polycystin-1 causes cystogenesis and bone defects. Hum Mol Genet 10:2385–2396
Watnick TJ, Torres VE, Gandolph MA, Qian F, Onuchic LF, Klinger KW, Landes G, Germino GG (1998) Somatic mutation in individual liver cysts supports a two-hit model of cystogenesis in autosomal dominant polycystic kidney disease. Mol Cell 2:247–251
Cameron JR (1961) Bilateral “hereditary” polycystic disease of the kidneys associated with bilateral teratodactyly of the feet. Br J Urol 33:473–477
Pretorius DH, Lee ME, Manco-Johnson ML, Weingast GR, Sedman AB, Gabow PA (1987) Diagnosis of autosomal dominant polycystic kidney disease in utero and in the young infant. J Ultrasound Med 6:249–255
Turco AE, Padovani EM, Chiaffoni GP, Peissel B, Rossetti S, Marcolongo A, Gammaro L, Maschio G, Pignatti PF (1993) Molecular genetic diagnosis of autosomal dominant polycystic kidney disease in a newborn with bilateral cystic kidneys detected prenatally and multiple skeletal malformations. J Med Genet 30:419–422
Ubara Y, Higa Y, Tagami T, Suwabe T, Nomura K, Kadoguchi K, Hoshino J, Sawa N, Katori H, Takemoto F, Kitajima I, Hara S, Takaichi K (2005) Pelvic insufficiency fracture related to autosomal dominant polycystic kidney disease. Am J Kidney Dis 46:e103–e111
Feather SA, Winyard PJ, Dodd S, Woolf AS (1997) Oral-facial-digital syndrome type 1 is another dominant polycystic kidney disease: clinical, radiological and histopathological features of a new kindred. Nephrol Dial Transplant 12:1354–1361
Feather SA, Woolf AS, Donnai D, Malcolm S, Winter RM (1997) The oral-facial-digital syndrome type 1 (OFD1), a cause of polycystic kidney disease and associated malformations, maps to Xp22.2–Xp22.3. Hum Mol Genet 6:1163–1167
Ramos FJ, Kaplan BS, Bellah RD, Zackai EH, Kaplan P (1998) Further evidence that the Hajdu-Cheney syndrome and the “serpentine fibula-polycystic kidney syndrome” are a single entity. Am J Med Genet 78:474–481
Isidor B, Le Merrer M, Exner GU, Pichon O, Thierry G, Guiochon-Mantel A, David A, Cormier-Daire V, Le Caignec C (2011) Serpentine fibula-polycystic kidney syndrome caused by truncating mutations in NOTCH2. Hum Mutat 32:1239–1242
Gray MJ, Kim CA, Bertola DR, Arantes PR, Stewart H, Simpson MA, Irving MD, Robertson SP (2012) Serpentine fibula polycystic kidney syndrome is part of the phenotypic spectrum of Hajdu-Cheney syndrome. Eur J Hum Genet 20:122–124
Anoop UR, Verma K, Narayanan K (2011) Primary cilia in the pathogenesis of dentigerous cyst: a new hypothesis based on role of primary cilia in autosomal dominant polycystic kidney disease. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 111:608–617
Liu S, Quarles LD (2007) How fibroblast growth factor 23 works. J Am Soc Nephrol 18:1637–1647
Yamazaki Y, Tamada T, Kasai N, Urakawa I, Aono Y, Hasegawa H, Fujita T, Kuroki R, Yamashita T, Fukumoto S, Shimada T (2008) Anti-FGF23 neutralizing antibodies show the physiological role and structural features of FGF23. J Bone Miner Res 23:1509–1518
Yoshiko Y, Wang H, Minamizaki T, Ijuin C, Yamamoto R, Suemune S, Kozai K, Tanne K, Aubin JE, Maeda N (2007) Mineralized tissue cells are a principal source of FGF23. Bone 40:1565–1573
Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, Fujita T, Nakahara K, Fukumoto S, Yamashita T (2004) FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res 19:429–435
Razzaque MS (2009) Does FGF23 toxicity influence the outcome of chronic kidney disease? Nephrol Dial Transplant 24:4–7
Krajisnik T, Bjorklund P, Marsell R, Ljunggren O, Akerstrom G, Jonsson KB, Westin G, Larsson TE (2007) Fibroblast growth factor-23 regulates parathyroid hormone and 1alpha-hydroxylase expression in cultured bovine parathyroid cells. J Endocrinol 195:125–131
Ben-Dov IZ, Galitzer H, Lavi-Moshayoff V, Goetz R, Kuro-o M, Mohammadi M, Sirkis R, Naveh-Many T, Silver J (2007) The parathyroid is a target organ for FGF23 in rats. J Clin Invest 117:4003–4008
Bacchetta J, Sea JL, Chun RF, Lisse TS, Wesseling-Perry K, Gales B, Adams JS, Salusky IB, Hewison M (2012) FGF23 inhibits extra-renal synthesis of 1,25-dihydroxyvitamin D in human monocytes. J Bone Miner Res 28:46–55
Kurosu H, Kuro OM (2009) The Klotho gene family as a regulator of endocrine fibroblast growth factors. Mol Cell Endocrinol 299:72–78
Farrow EG, Davis SI, Summers LJ, White KE (2009) Initial FGF23-mediated signaling occurs in the distal convoluted tubule. J Am Soc Nephrol 20:955–960
Hu MC, Shi M, Zhang J, Pastor J, Nakatani T, Lanske B, Razzaque MS, Rosenblatt KP, Baum MG, Kuro-o M, Moe OW (2010) Klotho: a novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J 24:3438–3450
Chang Q, Hoefs S, van der Kemp AW, Topala CN, Bindels RJ, Hoenderop JG (2005) The beta-glucuronidase klotho hydrolyzes and activates the TRPV5 channel. Science 310:490–493
Juppner H, Wolf M, Salusky IB (2010) FGF-23: more than a regulator of renal phosphate handling? J Bone Miner Res 25:2091–2097
Prie D, Urena Torres P, Friedlander G (2009) Latest findings in phosphate homeostasis. Kidney Int 75:882–889
Isakova T, Xie H, Yang W, Xie D, Anderson AH, Scialla J, Wahl P, Gutierrez OM, Steigerwalt S, He J, Schwartz S, Lo J, Ojo A, Sondheimer J, Hsu CY, Lash J, Leonard M, Kusek JW, Feldman HI, Wolf M (2011) Fibroblast growth factor 23 and risks of mortality and end-stage renal disease in patients with chronic kidney disease. JAMA 305:2432–2439
Mirza MA, Hansen T, Johansson L, Ahlstrom H, Larsson A, Lind L, Larsson TE (2009) Relationship between circulating FGF23 and total body atherosclerosis in the community. Nephrol Dial Transplant 24:3125–3131
Mirza MA, Larsson A, Lind L, Larsson TE (2009) Circulating fibroblast growth factor-23 is associated with vascular dysfunction in the community. Atherosclerosis 205:385–390
Fukagawa M, Nii-Kono T, Kazama JJ (2005) Role of fibroblast growth factor 23 in health and in chronic kidney disease. Curr Opin Nephrol Hypertens 14:325–329
Gutierrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, Shah A, Smith K, Lee H, Thadhani R, Juppner H, Wolf M (2008) Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med 359:584–592
Nakanishi S, Kazama JJ, Nii-Kono T, Omori K, Yamashita T, Fukumoto S, Gejyo F, Shigematsu T, Fukagawa M (2005) Serum fibroblast growth factor-23 levels predict the future refractory hyperparathyroidism in dialysis patients. Kidney Int 67:1171–1178
Srivaths PR, Goldstein SL, Silverstein DM, Krishnamurthy R, Brewer ED (2011) Elevated FGF 23 and phosphorus are associated with coronary calcification in hemodialysis patients. Pediatr Nephrol 26:945–951
Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, Gutierrez OM, Aguillon-Prada R, Lincoln J, Hare JM, Mundel P, Morales A, Scialla J, Fischer M, Soliman EZ, Chen J, Go AS, Rosas SE, Nessel L, Townsend RR, Feldman HI, St John Sutton M, Ojo A, Gadegbeku C, Di Marco GS, Reuter S, Kentrup D, Tiemann K, Brand M, Hill JA, Moe OW, Kuro OM, Kusek JW, Keane MG, Wolf M (2011) FGF23 induces left ventricular hypertrophy. J Clin Invest 121:4393–4408
Marsell R, Krajisnik T, Goransson H, Ohlsson C, Ljunggren O, Larsson TE, Jonsson KB (2008) Gene expression analysis of kidneys from transgenic mice expressing fibroblast growth factor-23. Nephrol Dial Transplant 23:827–833
Bacchetta J, Dubourg L, Harambat J, Ranchin B, Abou-Jaoude P, Arnaud S, Carlier MC, Richard M, Cochat P (2010) The influence of glomerular filtration rate and age on fibroblast growth factor 23 serum levels in pediatric chronic kidney disease. J Clin Endocrinol Metab 95:1741–1748
Yamazaki Y, Imura A, Urakawa I, Shimada T, Murakami J, Aono Y, Hasegawa H, Yamashita T, Nakatani K, Saito Y, Okamoto N, Kurumatani N, Namba N, Kitaoka T, Ozono K, Sakai T, Hataya H, Ichikawa S, Imel EA, Econs MJ, Nabeshima Y (2010) Establishment of sandwich ELISA for soluble alpha-Klotho measurement: age-dependent change of soluble alpha-Klotho levels in healthy subjects. Biochem Biophys Res Commun 398:513–518
Torres VE, Chapman AB, Devuyst O, Gansevoort RT, Grantham JJ, Higashihara E, Perrone RD, Krasa HB, Ouyang J, Czerwiec FS (2012) Tolvaptan in patients with autosomal dominant polycystic kidney disease. N Engl J Med 367:2407–2418
Acknowledgements
This work was supported in part by an educational grant from the Research Foundation Flanders (FWO), Belgium. The authors would like to thank Pierre Cochat and Bruno Ranchin (University of Lyon, France), Elena Levtchenko and Pieter Evenepoel (University Hospital of Leuven) and Humbert De Smedt (KU Leuven) for their helpful thoughts and comments.
Disclosure of interests
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mekahli, D., Bacchetta, J. From bone abnormalities to mineral metabolism dysregulation in autosomal dominant polycystic kidney disease. Pediatr Nephrol 28, 2089–2096 (2013). https://doi.org/10.1007/s00467-012-2384-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-012-2384-5