Abstract
Background
Hemodialysis (HD) in infants is usually used when peritoneal dialysis (PD) has failed. We describe our experience with HD, outlining the morbidity, complications, and outcomes for infants weighing less than 10 kg managed with HD for more than 6 months over a 10-year period.
Methods
A retrospective review of the clinical notes was conducted to collect demographic information, anthropometric data, dietary history, site and form of vascular access, details of HD prescription, complications, and outcomes.
Results
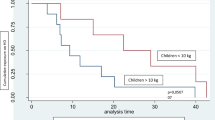
Nine patients weighing less than 10 kg were hemodialyzed for more than 6 months. Median age at commencement was 9 months. Median weight and height standard deviation score (SDS) at commencement of HD were −2.14 and −0.61, respectively, and at the end they were −1.56 and −1.61. Median energy intake was 96.6 kcal/kg/day and protein intake was 1.66 g/kg/day. Median number of line revisions was 0.32 line changes/patient year. Median central venous catheter (CVC) longevity was 13 months. Mean rate of line infection was 0.14/patient year. Median time on HD was 27 months. Median age at transplantation was 3.4 years.
Conclusions
This case series shows that chronic HD is a viable management option in children <10 kg. Access issues can be minimized with good line care to maximize line longevity and minimize line infection rates.
Similar content being viewed by others
References
Lewis MA, Shaw J, Sinha MD, Adalat S, Hussain F, Castledine C, van Schalkwyk D, Inward C (2010) UK Renal Registry 12th Annual Report (December 2009): chapter 14: demography of the UK paediatric renal replacement therapy population in 2008. Nephron Clin Pract 115(Suppl 1):c279–88
Mateer FM, Greenman L, Danowski TS (1955) Hemodialysis of the uremic child. AMA Am J Dis Child 89:645–655
Al-Hermi BE, Al-Saran K, Secker D, Geary DF (1999) Hemodialysis for end-stage renal disease in children weighing less than 10 kg. Pediatr Nephrol 13:401–403
Shroff R, Wright E, Ledermann S, Hutchinson C, Rees L (2003) Chronic hemodialysis in infants and children under 2 years of age. Pediatr Nephrol 18:378–383
Kovalski Y, Cleper R, Krause I, Davidovits M (2007) Hemodialysis in children weighing less than 15 kg: a single-center experience. Pediatr Nephrol 22:2105–2110
Cole TJ, Freeman JV, Preece MA (1998) British 1990 growth reference centiles for weight, height, body mass index and head circumference fitted by maximum penalized likelihood. Stat Med 17:407–429
Shaw V, Lawson M (2008) Clinical Paediatric Dietetics. Wiley-Blackwell, New York
KDOQI, National Kidney Foundation (2006) KDOQI Clinical Practice Guidelines and Clinical Practice Recommendations for Anemia in Chronic Kidney Disease. Am J Kidney Dis 47:S11–145
Carey WA, Talley LI, Sehring SA, Jaskula JM, Mathias RS (2007) Outcomes of dialysis initiated during the neonatal period for treatment of end-stage renal disease: a North American pediatric renal trials and collaborative studies special analysis. Pediatrics 119:e468–73
Goldstein SL (1997) Hemodialysis catheter survival and complications in children and adolescents. Pediatr Nephrol 11:74–77
Subcommittee RAS, London RCOPO, Society BT, Nephrologists BAOP. Treatment of adults and children with renal failure. Royal College of Physicians; 2002. www.renal.org
Hannah EL, Stevenson KB, Lowder CA, Adcox MJ, Davidson RL, Mallea MC, Narasimhan N, Wagnild JP (2002) Outbreak of hemodialysis vascular access site infections related to malfunctioning permanent tunneled catheters: making the case for active infection surveillance. Infect Control Hosp Epidemiol 23:538–541
Saxena AK, Panhotra BR, Al-Mulhim AS (2005) Vascular access-related infections in hemodialysis patients. Saudi J Kidney Dis Transpl 16:46–71
Beathard GA (2003) Catheter management protocol for catheter-related bacteremia prophylaxis. Semin Dial 16:403–405
Stefanidis CJ (2009) Prevention of catheter-related bacteremia in children on hemodialysis: time for action. Pediatr Nephrol 24:2087–2095
Karlberg J, Schaefer F, Hennicke M, Wingen AM, Rigden S, Mehls O (1996) Early age-dependent growth impairment in chronic renal failure. European study group for nutritional treatment of chronic renal failure in childhood. Pediatr Nephrol 10:283–287
Ulinski T, Cochat P (2006) Longitudinal growth in children following kidney transplantation: from conservative to pharmacological strategies. Pediatr Nephrol 21:903–909
Fischbach M, Terzic J, Menouer S, Dheu C, Seuge L, Zalosczic A (2010) Daily on line haemodiafiltration promotes catch-up growth in children on chronic dialysis. Nephrol Dial Transplant 25:867–873
Fischbach M, Terzic J, Menouer S, Dheu C, Soskin S, Helmstetter A, Burger M-C (2006) Intensified and daily hemodialysis in children might improve statural growth. Pediatr Nephrol 21:1746–1752
Fine RN, Martz K, Stablein D (2010) What have 20 years of data from the North American Pediatric Renal Transplant Cooperative Study taught us about growth following renal transplantation in infants, children, and adolescents with end-stage renal disease? Pediatr Nephrol 25:739–746
Kari JA, Gonzalez C, Ledermann SE, Shaw V, Rees L (2000) Outcome and growth of infants with severe chronic renal failure. Kidney Int 57:1681–1687
KDOQI Work Group (2009) KDOQI Clinical Practice Guideline for Nutrition in Children with CKD: 2008 update. Executive summary. Am J Kidney Dis 53:S11–104
Schaefer F, Warady B, Investigators IPPDN. International Pediatric Peritoneal Dialysis Network [Internet]. pedpd.org; Available from: http://www.pedpd.org
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Appendix 1
CVC care
All our CVCs are placed by experienced pediatric surgeons and we have a small team of trained specialized nurses who perform CVC care within a strict protocol as outlined below.
Prophylactic antibiotics are given immediately prior to CVC placement (vancomycin 10 mg/kg). The dressing is left untouched for 5 days and then changed three times a week by the HD nursing team, when skin is cleaned with 0.5 % chlorhexidine and the dressing is changed with strict adherence to a sterile non-touch technique. Sterile gloves are worn and the child is positioned as necessary to avoid any accidental contact with the line. Neither staff nor patient wears a mask.
If the exit site appears erythematous at any stage, a skin swab is taken and the site is treated with a topical antibiotic cream. Treatment is discontinued if the swab is negative. If positive, then treatment is continued according to sensitivities until a subsequent swab is negative. Each child is given a full body screen (swabs from nose, throat, axilla, groin, CVC site, and any other devices in situ) 3-monthly including culture for MRSA, ESBL, and VRE. Only trained HD nurses from our unit are allowed to access the CVCs. Parents are advised to wash children in shallow baths only, no showers, and lines are secured far from the nappy area where applicable.
Bacteremia is suspected if the child becomes pyrexic or unwell at any stage. Blood is sent for culture, full blood count and CRP prior to the commencement of IV vancomycin. The antibiotics are discontinued if the culture is negative after 48 h, and if it is positive, then treatment is continued according to sensitivities and in discussion with the consultant microbiologist and nephrologist.
Rights and permissions
About this article
Cite this article
Quinlan, C., Bates, M., Sheils, A. et al. Chronic hemodialysis in children weighing less than 10 kg. Pediatr Nephrol 28, 803–809 (2013). https://doi.org/10.1007/s00467-012-2373-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-012-2373-8