Abstract
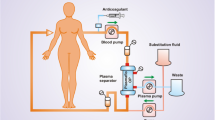
Regional citrate anticoagulation (RCA) has been considered to be a standard component of pediatric apheresis therapy for more than a decade. However, data on dosing recommendations and evaluations of the effectiveness and safety of anticoagulation are rarely found in published reports. The aim of this retrospective analysis was to present our single-center experience with RCA in pediatric apheresis therapy with the aim of developing an operating procedure. Five children aged 7–14 years underwent a total of 72 (range 3–44) therapeutic apheresis sessions with RCA in the form of immunoadsorption therapy (2 patients), low-density lipoprotein (LDL)-apheresis (1 patient), and plasmapheresis (two patients). A 3% citrate solution was used. Citrate flow was started at 4.0% of the blood flow velocity and was adapted to match post-filter ionized calcium levels ≤0.30 mmol/l. Once the patient’s ionized calcium fell to <1.05 mmol/l, an intravenous 10% calcium gluconate solution was administered. Twenty pediatric apheresis patients who received standard heparinization, matched for age, body surface area, processed plasma volume, and blood flow velocity, were enrolled in the study as a comparison group. No side effects were experienced in 72 apheresis session. The 3% citrate solution had to be reduced gradually during the apheresis session and was infused at a mean of 2.8–3.8% of the blood flow rate. Serum bicarbonate levels before and after the apheresis session with RCA [23.9 (range 18.9–30.1) vs. 26.3 (20.2–33.0) mmol/l, respectively] were significantly different (p = 0.013). All patients required intravenous calcium substitution to maintain serum calcium levels within the physiological range. Due to the administration of the 3% citrate solution and calcium, all patients significantly gained weight during the procedure, with a median weight gain of 2.5% (p < 0.001). The extra fluid load caused problems in patients with kidney failure. Our regimen with RCA is safe, feasible, and effective in pediatric therapeutic apheresis therapy. For RCA in apheresis, we recommend (1) a citrate (3%) flow of 3.3% of the blood flow, (2) prophylactic intravenous calcium substitution from the beginning, and (3) a more highly concentrated citrate solution in the case of oliguric patients.
Similar content being viewed by others
Abbreviations
- ACD-A:
-
Anticoagulant citrate dextrose Solution A
- ACT:
-
Activated coagulation time
- HIT:
-
Heparin-induced thrombocytopenia
- LDL:
-
Low-density lipoprotein cholesterol
- RCA:
-
Regional citrate anticoagulation
References
Witt V, Stegmayr B, Ptak J, Wikström B, Berlin G, Axelsson CG, Griskevicius A, Centoni PE, Liumbruno GM, Molfettini P, Audzijoniene J, Mokvist K, Sojka BN, Norda R, Ramlow W, Blaha M, Evergren M, Tomaz J (2008) World apheresis registry data from 2003 to 2007, the pediatric and adolescent side of the registry. Transfus Apher Sci 39:255–260
Bunchman T (2002) Plasmapheresis and renal replacement therapy in children. Curr Opin Pediatr 14:310–314
Gorlin JB (1999) Therapeutic plasma exchange and cytapheresis in pediatric patients. Transfus Sci 21:21–39
Linnenberger ML, Price TH (2005) Use of cellular and plasma apheresis in the critically Ill patient: part II: clinical indications and applications. J Intensive Care Med 20:88–103
Davenport A (2003) Anticoagulation options for pediatric hemodialysis. Hemodial Int 7:168–176
Kreuzer M, Ehrich JH, Pape L (2010) Haemorrhagic complications in paediatric dialysis-dependent acute kidney injury: Incidence and impact on outcome. Nephrol Dial Transplant 25:1140–1146
Janssen MJ (1996) The bleeding risk in chronic haemodialysis. Preventive strategies in high-risk patients. Neth J Med 48:198–207
Bock KR (2005) Renal replacement therapy in pediatric critical care medicine. Curr Opin Pediatr 17:368–371
Bolan CD, Cecco SA, Wesley RA, Horne M, Yau YY, Remaley AT, Childs RW, Barrett AJ, Rehak NN, Leitman SF (2002) Controlled study of citrate effects and response to i.v. calcium administration during allogeneic peripheral blood progenitor cell donation. Transfusion 42:935–946
Bolan CD, Yau YY, Cullis HC, Horwitz ME, Mackall CL, Barrett AJ, Malech HL, Rehak NN, Wayne AS, Leitman SF (2004) Pediatric large-volume leukapheresis: a single institution experience with heparin versus citrate-based anticoagulant regimens. Transfusion 44:229–238
De Silvestro G, Tison T, Vicarioto M, Bagatella P, Stefanutti C, Marson P (2009) The Italian registry of pediatric therapeutic apheresis: a report on activity during 2005. J Clin Apher 24:1–5
Nuthall G, Skippen P, Daoust C, Al-Jofan F, Seear M (2002) Citrate anticoagulation in a piglet model of pediatric continuous renal replacement therapy. Crit Care Med 30:900–903
Kreuzer M, Ahlenstiel T, Kanzelmeyer T, Ehrich JH, Pape L (2010) Management of regional citrate anticoagulation in pediatric high-flux dialysis: Activated coagulation time versus post-filter ionized calcium. Pediatr Nephrol 25:1305–1310
van de Wetering J, Westendorp RG, van der Hoeven JG, Stolk B, Feuth JD, Chang PC (1996) Heparin use in continuous renal replacement procedures: the struggle between filter coagulation and patient hemorrhage. J Am Soc Nephrol 7:45–50
Dzik WH, Kirkley SA (1988) Citrate toxicity during massive blood transfusion. Transfus Med Rev 2:76–94
Dhondt A, Vanholder R, Tielemans C, Glorieux G, Waterloos MA, De Smet R, Lameire N (2000) Effect of regional citrate anticoagulation on leukopenia, complement activation, and expression of leukocyte surface molecules during hemodialysis with unmodified cellulose membranes. Nephron 85:334–342
Kim HC (2000) Therapeutic pediatric apheresis. J Clin Apher 15:129–157
Witt V (2010) Training courses for pediatric apheresis on site; how apheresis technology transfer can be performed. Transfus Apher Sci. doi:10.1016/j.transci.2010.07.024
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kreuzer, M., Ahlenstiel, T., Kanzelmeyer, N. et al. Regional citrate anticoagulation—a safe and effective procedure in pediatric apheresis therapy. Pediatr Nephrol 26, 127–132 (2011). https://doi.org/10.1007/s00467-010-1658-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-010-1658-z