Abstract
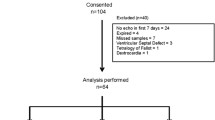
Patent ductus arteriosus (PDA) is the most common cardiovascular abnormality of the preterm infant usually treated with ibuprofen (IBU). PDA is strictly related to oxidative stress (OS) in neonates. This study tests the hypothesis that OS occurs in neonates with PDA and that IBU treatment reduces OS. Forty-three preterm babies with gestational age (GA) <33 weeks were studied prospectively. Three urine samples were collected: at time 0 (before starting treatment), time 1 (after pharmacological PDA closure), and time 2 (7 days after the end of treatment) in all patients. OS was studied by measuring urinary isoprostane (IPs) levels. The results showed significant changes in urinary IP levels from time 0 to time 2 (Kruskal–Wallis, p = 0.047). Time trend showed a significant decrease in IPs from time 0 to time 1 after IBU therapy (p = 0.0067). This decrease was followed by an increase in IPs levels 7 days after treatment. IBU therapy for PDA closure reduced the risk of OS related to free-radical (FR) generation. This antioxidant effect of IBU may be beneficial in preterm babies with PDA who are at high risk for OS.


Similar content being viewed by others
References
Van Overmeire B, Chemtob S (2005) The pharmacologic closure of the patent ductus arteriosus. Semin Fetal Neonatal Med 10:177–184
Brook MM, Heymann MA (1995) Patent ductus arteriosus. In: Emmanouilides GC, Riemenschneider TA, Allen HD, Gutgesell HP (eds) Moss and Adams' heart disease in infants, children, and adolescents: including the fetus and young adult, 5th edn. Williams & Wilkins, Baltimore, pp 746–764
Gersony WM (1986) Patent ductus arteriosus in the neonate. Pediatr Clin North Am 33:545–560
Patel J, Roberts I, Azzopardi D, Hamilton P, Edwards AD (2000) Randomized double-blind controlled trial comparing the effects of ibuprofen with indomethacin on cerebral hemodynamics in preterm infants with patent ductus arteriosus. Pediatr Res 47:36–42
Buonocore G, Perrone S, Longini M, Vezzosi P, Marzocchi B, Paffetti P, Bracci R (2002) Oxidative stress in preterm neonates at birth and on the seventh day of life. Pediatr Res 52:46–49
Basu S (2008) F2-isoprostanes in human health and diseases: from molecular mechanisms to clinical implications. Antioxid Redox Signal 10:1405–1434
Buonocore G, Perrone S, Longini M, Paffetti P, Vezzosi P, Gatti MG, Bracci R (2003) Non protein bound iron as early predictive marker of neonatal brain damage. Brain 126:1224–1230
Buonocore G, Groenendaal F (2007) Anti-oxidant strategies. Semin Fetal Neonatal Med 12:287–295
Palmer C, Menzies SL, Roberts RL, Pavlick G, Connor JR (1999) Changes in iron histochemistry after hypoxic-ischemic brain injury in the neonatal rat. J Neurosci Res 56:60–71
Comporti M, Signorini C, Leoncini S, Buonocore G, Rossi V, Ciccoli L (2004) Plasma F2-isoprostanes are elevated in newborns and inversely correlated to gestational age. Free Radic Biol Med 37:724–732
Longini M, Perrone S, Kenanidis A, Vezzosi P, Marzocchi B, Petraglia F, Centini G, Buonocore G (2005) Isoprostanes in amniotic fluid: a predictive marker for fetal growth restriction in pregnancy. Free Radic Biol Med 38:1537–1541
Longini M, Perrone S, Vezzosi P, Marzocchi B, Kenanidis A, Centini G, Rosignoli L, Buonocore G (2007) Association between oxidative stress in pregnancy and preterm premature rupture of membranes. Clin Biochem 40:793–797
Hamburger SA, McCay PB (1990) Spin trapping of ibuprofen radicals: evidence that ibuprofen is a hydroxyl radical scavenger. Free Radic Res Commun 9:337–342
Saugstad OD (2005) Oxidative stress in the newborn – a 30-year perspective. Biol Neonate 88:228–236
Ohlsson A, Walia R, Shah S (2008) Ibuprofen for the treatment of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database Syst Rev CD003481
Fell JM, Thakkar H, Newman DJ, Price CP (1997) Measurement of albumin and low molecular weight proteins in the urine of newborn infants using a cotton wool ball collection method. Acta Paediatr 86:518–522
Milne GL, Yin H, Brooks JD, Sanchez S, Jackson Roberts L, Morrow JD 2nd (2007) Quantification of F2-isoprostanes in biological fluids and tissues as a measure of oxidant stress. Methods Enzymol 433:113–126
Roberts LJ, Morrow JD (2000) Measurement of F(2)-isoprostanes as an index of oxidative stress in vivo. Free Radic Biol Med 28:505–513
Basu S (2004) Isoprostanes: novel bioactive products of lipid peroxidation. Free Radic Res 38:105–122
Pratico D (1999) F2-Isoprostanes: Sensitive and specific non-invasive indices of lipid peroxidation in vivo. Atherosclerosis 147:1–10
Helmersson J, Basu S (2001) F2-Isoprostanes and prostaglandin F2α metabolite excretion rate and day to day variation in healthy humans. Prostaglandins Leukot Essent Fatty Acids 65:99–102
Davies NM (1998) Clinical pharmacokinetics of ibuprofen. The first 30 years. Clin Pharmacokinet 34:101–154
Milatovic D, Zaja-Milatovic S, Montine KS, Shie FS, Montine TJ (2004) Neuronal oxidative damage and dendritic degeneration following activation of CD14-dependent innate immune response in vivo. J Neuroinflammation 1:20
Ascenzi P, di Masi A, De Sanctis G, Coletta M, Fasano M (2009) Ibuprofen modulates allosterically NO dissociation from ferrous nitrosylated human serum heme-albumin by binding to three sites. Biochem Biophys Res Commun 387:83–86
Klein T, Reutter F, Schweer H, Seyberth HW, Nüsing RM (1997) Generation of the isoprostane 8-epi-prostaglandin F2α in vitro and in vivo via the cyclooxygenases. J Pharmacol Exp Ther 282:1658–1665
Acknowledgment
We thank EURAIBI FONDAZIONE ONLUS for support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Longini, M., Perrone, S., Vezzosi, P. et al. Isoprostane levels in urine of preterm newborns treated with ibuprofen for patent ductus arteriosus closure. Pediatr Nephrol 26, 105–109 (2011). https://doi.org/10.1007/s00467-010-1651-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-010-1651-6