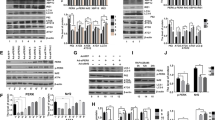
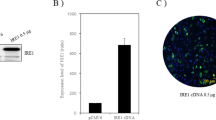
Abstract
The goal of the study is to examine the relationship between the sensor molecules, Hypoxia Inducible Factor-1 (HIF-1), AMP activated Protein Kinase (AMPK) and mammalian Target of Rapamycin (mTOR) in chondrocyte survival and autophagy. We showed that chondrocytes expressed the energy sensor AMPK-1 and that activation increased with maturation. In addition, we showed that thapsigargin treatment activated AMPK and autophagy in a HIF-1-dependent manner. Using serum-starved AMPK-silenced cells, we demonstrated that AMPK was required for the induction of the autophagic response. We also noted a change in chondrocyte sensitivity to apoptogens, due to activation of caspase-8 and cleavage and activation of the pro-apoptotic protein, BID. To test the hypothesis that AMPK signaling directly promoted autophagy, we inhibited AMPK activity in mTOR silenced cells and showed that while mTOR suppression induced autophagy, AMPK inhibition did not block this activity. Based on these findings, it is concluded that because of the micro-environmental changes experienced by the chondrocyte, autophagy is activated by AMPK in a HIF-1-dependent manner.






Similar content being viewed by others
References
Rajpurohit R, Koch CJ, Tao Z, Teixeira CM, Shapiro IM (1996) Adaptation of chondrocytes to low oxygen tension: relationship between hypoxia and cellular metabolism. J Cell Physiol 168:424–432
Schipani E, Ryan HE, Didrickson S, Kobayashi T, Knight M, Johnson RS (2001) Hypoxia in cartilage: HIF-1alpha is essential for chondrocyte growth arrest and survival. Genes Dev 15:2865–2876
Bohensky J, Shapiro IM, Leshinsky S, Terkhorn SP, Adams CS, Srinivas V (2007) HIF-1 regulation of chondrocyte apoptosis: induction of the autophagic pathway. Autophagy 3:207–214
Zhang H, Bosch-Marce M, Shimoda LA, Tan YS, Baek JH, Wesley JB, Gonzalez FJ, Semenza GL (2008) Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J Biol Chem 283:10892–10903
Srinivas V, Bohensky J, Zahm AM, Shapiro IM (2009) Autophagy in mineralizing tissues: microenvironmental perspectives. Cell Cycle 8:391–393
Bohensky J, Terkhorn SP, Freeman TA, Adams CS, Garcia JA, Shapiro IM, Srinivas V (2009) Regulation of autophagy in cartilage: HIF-2 suppresses chondrocyte autophagy. Arthritis Rheum 60:1406–1415
Barnes K, Ingram JC, Porras OH, Barros LF, Hudson ER, Fryer LG, Foufelle F, Carling D, Hardie DG, Baldwin S (2002) Activation of GLUT1 by metabolic and osmotic stress: potential involvement of AMP-activated protein kinase (AMPK). J Cell Sci 115:2433–2442
Liang J, Shao SH, Xu ZX, Hennessy B, Ding Z, Larrea M, Kondo S, Dumont DJ, Gutterman JU, Walker CL, Slingerland JM, Mills GB (2007) The energy sensing LKB1-AMPK pathway regulates p27 (kip1) phosphorylation mediating the decision to enter autophagy or apoptosis. Nat Cell Biol 9:218–224
Davies SP, Helps NR, Cohen PT, Hardie DG (1995) 5′-AMP inhibits dephosphorylation, as well as promoting phosphorylation, of the AMP-activated protein kinase. Studies using bacterially expressed human protein phosphatase-2C alpha and native bovine protein phosphatase-2AC. FEBS Lett 377:421–425
Levine B (2006) Unraveling the role of autophagy in cancer. Autophagy 2:65–66
Klionsky DJ (2005) Autophagy. Curr Biol 15:R282–R283
Sabatini DM (2006) mTOR and cancer: insights into a complex relationship. Nat Rev Cancer 6:729–734
Nobukuni T, Joaquin M, Roccio M, Dann SG, Kim SY, Gulati P, Byfield MP, Backer JM, Natt F, Bos JL, Zwartkruis FJ, Thomas G (2005) Amino acids mediate mTOR/raptor signaling through activation of class 3 phosphatidylinositol 3OH-kinase. Proc Natl Acad Sci U S A 102:14238–14243
Nobukuni T, Kozma SC, Thomas G (2007) hvps34, an ancient player, enters a growing game: mTOR Complex1/S6K1 signaling. Curr Opin Cell Biol 19:135–141
Lum JJ, DeBerardinis RJ, Thompson CB (2005) Autophagy in metazoans: cell survival in the land of plenty. Nat Rev Mol Cell Biol 6:439–448
Cota D, Proulx K, Smith KA, Kozma SC, Thomas G, Woods SC, Seeley RJ (2006) Hypothalamic mTOR signaling regulates food intake. Science 312:927–930
Bohensky J, Shapiro IM, Leshinsky S, Watanabe H, Srinivas V (2007) PIM-2 is an independent regulator of chondrocyte survival and autophagy in the epiphyseal growth plate. J Cell Physiol 213:246–251
Matsumoto H, Debolt K, Shapiro IM (1988) Adenine, guanine, and inosine nucleotides of chick growth cartilage: relationship between energy status and the mineralization process. J Bone Miner Res 3:347–352
Meley D, Bauvy C, Houben-Weerts JH, Dubbelhuis PF, Helmond MT, Codogno P, Meijer AJ (2006) AMP-activated protein kinase and the regulation of autophagic proteolysis. J Biol Chem 281:34870–34879
Erlich S, Alexandrovich A, Shohami E, Pinkas-Kramarski R (2007) Rapamycin is a neuroprotective treatment for traumatic brain injury. Neurobiol Dis 26:86–93
Gordon PB, Holen I, Fosse M, Røtnes JS, Seglen PO (1993) Dependence of hepatocytic autophagy on intracellularly sequestered calcium. J Biol Chem 268:26107–26112
Høyer-Hansen M, Bastholm L, Szyniarowski P, Campanella M, Szabadkai G, Farkas T, Bianchi K, Fehrenbacher N, Elling F, Rizzuto R, Mathiasen IS, Jäättelä M (2007) Control of macroautophagy by calcium, calmodulin-dependent kinase kinase-beta, and Bcl-2. Mol Cell 25:193–205
Terkhorn SP, Bohensky J, Shapiro IM, Koyama E, Srinivas V (2007) Expression of HIF prolyl hydroxylase isozymes in growth plate chondrocytes: Relationship between maturation and apoptotic sensitivity. J Cell Physiol 210:257–265
Acknowledgements
This work was supported by NIH grants DE 015694 and DE 016383 (to VS) and DE 010875 and DE 013319 (to IMS).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Bohensky, J., Leshinsky, S., Srinivas, V. et al. Chondrocyte autophagy is stimulated by HIF-1 dependent AMPK activation and mTOR suppression. Pediatr Nephrol 25, 633–642 (2010). https://doi.org/10.1007/s00467-009-1310-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-009-1310-y