Abstract
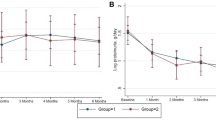
First line immunosuppressive treatment in steroid-resistant nephrotic syndrome in children is still open to discussion. We conducted a controlled multicentre randomized open label trial to test the efficacy and safety of cyclosporin A (CSA) versus cyclophosphamide pulses (CPH) in the initial therapy of children with newly diagnosed primary steroid-resistant nephrotic syndrome and histologically proven minimal change disease, focal segmental glomerulosclerosis or mesangial hypercellularity. Patients in the CSA group (n = 15) were initially treated with 150 mg/m2 CSA orally to achieve trough levels of 120–180 ng/ml, while patients in the CPH group (n = 17) received CPH pulses (500 mg/m2 per month intravenous). All patients were on alternate prednisone therapy. Patients with proteinuria >40 mg/m2 per hour at 12 weeks of therapy were allocated to a non-responder protocol with high-dose CSA therapy or methylprednisolone pulses. At week 12, nine of the 15 (60%) CSA patients showed at least partial remission, evidences by a reduction of proteinuria <40 mg/h per m2. In contrast, three of the 17 (17%) CPH patients responded (p < 0.05, intention-to-treat). Given these results, the study was stopped, in accordance with the protocol. After 24 weeks, complete remission was reached by two of the 15 (13%) CSA and one of the 17 (5%) CPH patients (p = n.s.). Partial remission was achieved by seven of the 15 (46%) CSA and two of the 15 (11%) CPH patients (p <0.05). Five patients in the CSA group and 14 patients in the CPH group were withdrawn from the study, most of them during the non-responder protocol. The number of adverse events was comparable between both groups. We conclude that CSA is more effective than CPH in inducing at least partial remission in steroid-resistant nephrotic syndrome in children.




Similar content being viewed by others
References
International Study of Kidney Disease in Children (1981) The primary nephrotic syndrome in children. Identification of patients with minimal change nephrotic syndrome from initial response to prednisone. A report of the International Study of Kidney Disease in Children. J Pediatr 98:561–564
Tarshish P, Tobin JN, Bernstein J, Edelmann CM Jr (1996) Cyclophosphamide does not benefit patients with focal segmental glomerulosclerosis. A report of the International Study of Kidney Disease in Children. Pediatr Nephrol 10:590–593
Abrantes MM, Cardoso LS, Lima EM, Penido Silva JM, Diniz JS, Bambirra EA, Oliveira EA (2006) Predictive factors of chronic kidney disease in primary focal segmental glomerulosclerosis. Pediatr Nephrol 21:1003–1012
Kim JS, Bellew CA, Silverstein DM, Aviles DH, Boineau FG, Vehaskari VM (2005) High incidence of initial and late steroid resistance in childhood nephrotic syndrome. Kidney Int 68:1275–1281
Chesney R (2004) The changing face of childhood nephrotic syndrome. Kidney Int 66:1294–1302
Tejani A, Suthanthiran M, Pomrantz A (1991) A randomized controlled trial of low-dose prednisone and ciclosporin versus high-dose prednisone in nephrotic syndrome of children. Nephron 59:96–99
Niaudet P, Fuchshuber A, Gagnadoux MF, Habib R, Broyer M (1997) Cyclosporine in the therapy of steroid-resistant idiopathic nephrotic syndrome. Kidney Int Suppl 58:S85–90
Gregory MJ, Smoyer WE, Sedman A, Kershaw DB, Valentini RP, Johnson K, Bunchman TE (1996) Long-term cyclosporine therapy for pediatric nephrotic syndrome: a clinical and histologic analysis. J Am Soc Nephrol 7:543–549
Singh A, Tejani C, Tejani A (1999) One-center experience with cyclosporine in refractory nephrotic syndrome in children. Pediatr Nephrol 13:26–32
El-Husseini A, El-Basuony F, Mahmoud I, Sheashaa H, Sabry A, Hassan R, Taha N, Hassan N, Sayed-Ahmad N, Sobh M (2005) Long-term effects of cyclosporine in children with idiopathic nephrotic syndrome: a single-centre experience. Nephrol Dial Transplant 20:2433–2438
Mahmoud I, Basuni F, Sabry A, El-Husseini A, Hassan N, Ahmad NS, Elbaz M, Moustafa F, Sobh M (2005) Single-centre experience with cyclosporin in 106 children with idiopathic focal segmental glomerulosclerosis. Nephrol Dial Transplant 20:735–742
Hodson EM, Habashy D, Craig JC (2006) Interventions for idiopathic steroid-resistant nephrotic syndrome in children. Cochrane Database Syst Rev:CD003594
Lieberman KV, Tejani A (1996) A randomized double-blind placebo-controlled trial of cyclosporine in steroid-resistant idiopathic focal segmental glomerulosclerosis in children. J Am Soc Nephrol 7:56–63
International study of Kidney Disease in Children (1974) Prospective, controlled trial of cyclophosphamide therapy in children with nephrotic syndrome. Report of the International study of Kidney Disease in Children. Lancet 2:423–427
Balow JE, Boumpas DT, Fessler BJ, Austin HA 3rd (1996) Management of lupus nephritis. Kidney Int Suppl 53:S88–92
Elhence R, Gulati S, Kher V, Gupta A, Sharma RK (1994) Intravenous pulse cyclophosphamide—a new regime for steroid-resistant minimal change nephrotic syndrome. Pediatr Nephrol 8:1–3
Mantan M, Bagga A, Siriam C, Hari P (2004) Efficacy of IV pulse cyclophosphamide (CP) vs iv steroids & oral CP for steroid resistant nephrotic syndrome. Pediatr Nephrol 19:C73
Tune BM, Kirpekar R, Sibley RK, Reznik VM, Griswold WR, Mendoza SA (1995) Intravenous methylprednisolone and oral alkylating agent therapy of prednisone-resistant pediatric focal segmental glomerulosclerosis: a long-term follow-up. Clin Nephrol 43:84–88
Tune BM, Lieberman E, Mendoza SA (1996) Steroid-resistant nephrotic focal segmental glomerulosclerosis: a treatable disease. Pediatr Nephrol 10:772–778
Waldo FB, Benfield MR, Kohaut EC (1992) Methylprednisolone treatment of patients with steroid-resistant nephrotic syndrome. Pediatr Nephrol 6:503–505
Waldo FB, Benfield MR, Kohaut EC (1998) Therapy of focal and segmental glomerulosclerosis with methylprednisolone, cyclosporine A, and prednisone. Pediatr Nephrol 12:397–400
Niaudet P (2000) Treatment of lupus nephritis in children. Pediatr Nephrol 14:158–166
Ehrich JH, Brodehl J (1993) Long versus standard prednisone therapy for initial treatment of idiopathic nephrotic syndrome in children. Arbeitsgemeinschaft für Pädiatrische Nephrologie. Eur J Pediatr 152:357–361
International Study of Kidney Disease in Children (1981) The primary nephrotic syndrome in children. Identification of patients with minimal change nephrotic syndrome from initial response to prednisone. J Pediatr 98:561–564
Karle SM, Uetz B, Ronner V, Glaeser L, Hildebrandt F, Fuchshuber A (2002) Novel mutations in NPHS2 detected in both familial and sporadic steroid-resistant nephrotic syndrome. J Am Soc Nephrol 13:388–393
Ruf RG, Schultheiss M, Lichtenberger A, Karle SM, Zalewski I, Mucha B, Everding AS, Neuhaus T, Patzer L, Plank C, Haas JP, Ozaltin F, Imm A, Fuchshuber A, Bakkaloglu A, Hildebrandt F, APN Study Group (2004) Prevalence of WT1 mutations in a large cohort of patients with steroid-resistant and steroid-sensitive nephrotic syndrome. Kidney Int 66:564–570
Ruf RG, Lichtenberger A, Karle SM, Haas JP, Anacleto FE, Schultheiss M, Zalewski I, Imm A, Ruf EM, Mucha B, Bagga A, Neuhaus T, Fuchshuber A, Bakkaloglu A, Hildebrandt F, Arbeitsgemeinschaft Für Pädiatrische Nephrologie Study Group (2004) Patients with mutations in NPHS2 (podocin) do not respond to standard steroid treatment of nephrotic syndrome. J Am Soc Nephrol 15:722–732
Mucha B, Ozaltin F, Hinkes BG, Hasselbacher K, Ruf RG, Schultheiss M, Hangan D, Hoskins BE, Everding AS, Bogdanovic R, Seeman T, Hoppe B, Hildebrandt F, Members of the APN Study Group (2006) Mutations in the Wilms’ tumor 1 gene cause isolated steroid resistant nephrotic syndrome and occur in exons 8 and 9. Pediatr Res 59:325–331
Ponticelli C, Rizzoni G, Edefonti A, Altieri P, Rivolta E, Rinaldi S, Ghio L, Lusvarghi E, Gusmano R, Locatelli F, Pasquali S, Castellani A, Della Casa-Alberighi O (1993) A randomized trial of cyclosporine in steroid-resistant idiopathic nephrotic syndrome. Kidney Int 43:1377–1384
Bajpai A, Bagga A, Hari P, Dinda A, Srivastava RN (2003) Intravenous cyclophosphamide in steroid-resistant nephrotic syndrome. Pediatr Nephrol 18:351–356
Eddy AA, Symons JM (2003) Nephrotic syndrome in childhood. Lancet 362:629–639
Cattran DC, Appel GB, Hebert LA, Hunsicker LG, Pohl MA, Hoy WE, Maxwell DR, Kunis CL (1999) A randomized trial of cyclosporine in patients with steroid-resistant focal segmental glomerulosclerosis. North America Nephrotic Syndrome Study Group. Kidney Int 56:2220–2226
Hollis S, Campbell F (1999) What is meant by intention to treat analysis? Survey of published randomised controlled trials. Br Med J 319:670–674
Garin EH, Orak JK, Hiott KL, Sutherland SE (1988) Cyclosporine therapy for steroid-resistant nephrotic syndrome. A controlled study. Am J Dis Child 142:985–988
Buyukcelik M, Cengiz N, Dursun H, Soran M, Bayazit AK, Noyan A, Anarat A (2006) Intravenous pulse cyclophosphamide therapy in focal segmental glomerulosclerosis. Clin Nephrol 65:7–12
Al Salloum AA (2004) Pulse cyclophosphamide therapy for steroid-resistant focal segmental glomerulosclerosis in children. Ann Saudi Med 24:27–30
Cattran DC, Rao P (1998) Long-term outcome in children and adults with classic focal segmental glomerulosclerosis. Am J Kidney Dis 32:72–79
Raafat RH, Kalia A, Travis LB, Diven SC (2004) High-dose oral cyclosporin therapy for recurrent focal segmental glomerulosclerosis in children. Am J Kidney Dis 44:50–56
Ingulli E, Singh A, Baqi N, Ahmad H, Moazami S, Tejani A (1995) Aggressive, long-term cyclosporine therapy for steroid-resistant focal segmental glomerulosclerosis. J Am Soc Nephrol 5:1820–1825
Mendoza SA, Tune BM (1992) Treatment of childhood nephrotic syndrome. J Am Soc Nephrol 3:889–894
Kirpekar R, Yorgin PD, Tune BM, Kim MK, Sibley RK (2002) Clinicopathologic correlates predict the outcome in children with steroid-resistant idiopathic nephrotic syndrome treated with pulse methylprednisolone therapy. Am J Kidney Dis 39:1143–1152
Yorgin PD, Krasher J, Al-Uzri AY (2001) Pulse methylprednisolone treatment of idiopathic steroid-resistant nephrotic syndrome. Pediatr Nephrol 16:245–250
Weber S, Gribouval O, Esquivel EL, Morinière V, Tête MJ, Legendre C, Niaudet P, Antignac C (2004) NPHS2 mutation analysis shows genetic heterogeneity of steroid-resistant nephrotic syndrome and low post-transplant recurrence. Kidney Int 66:571–579
Acknowledgements
We thank all the members of the Arbeitsgemeinschaft für Pädiatrische Nephrologie for their help in the design and realization of this project. For their help in coordinating the study, we thank K. Benz, K.-D. Nüsken, B. Geißler and K. Escherich. We very much appreciate the willingness of our patients and their families to participate in this trial. This project was supported in part by a grant of Novartis Pharma, Nuremberg, Germany.
Conflict of interest statement
None to declare.
Members of Protocol committee
W. Rascher, C. Plank (Erlangen, Germany); C. Aufricht (Vienna, Austria); J.H. Ehrich (Hannover, Germany); A. Fuchshuber (Freiburg, Germany); P. Hoyer (Essen); M. Kemper (Hamburg); O. Mehls, K. Schärer, B. Tönshoff (Heidelberg, Germany); U. Querfeld (Berlin, Germany); M. Schwab (Stuttgart, Germany).
List of contributing investigators
Austria: C. Aufricht; K. Arbeiter; T. Müller; A. Grillenberg; M. Mayrhofer; R. Vargha (Vienna, Linz)Germany: U. Querfeld; J. Gellermann; D. Haffner; M. Zimmering; (Berlin), H. Bachmann; N. Matthes (Bremen); G. Kalvoda; B. Mayer; S. Nolte; I. Wülke (Dresden); F. Wegner (Düren); W. Rascher; K. Benz; K. Dittrich; J. Dötsch; K-D. Nüsken; C. Plank (Erlangen); M. Pohl; S. Jacobi; C. von Schnakenburg (Freiburg); J.D. Schwarke; T. Henne (Hamburg); D.E. Müller-Wiefel; M. Kemper; A. Link; K. Zepf; (Hamburg); J.H. Ehrich; D. Franke; J. Strehlau (Hannover); B. Tönshoff; R. Feneberg; R. Himmele (Heidelberg); P. Eggert; D. Roessner (Kiel); R. Beetz (Mainz); G. Klaus; M. Konrad (Marburg); H. Fehrenbach; D. Kleinert (Memmingen); K.G. Pistor; K. Timmermann (Moers); M. Wigger; E. Drückler; I. Franke; J. Muscheites; G. Warncke (Rostock); Mutation Analysis: F. Hildebrandt, B. Hinkes; B. Mucha; R. Ruf (Ann Arbor, USA).
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Plank, C., Kalb, V., Hinkes, B. et al. Cyclosporin A is superior to cyclophosphamide in children with steroid-resistant nephrotic syndrome—a randomized controlled multicentre trial by the Arbeitsgemeinschaft für Pädiatrische Nephrologie. Pediatr Nephrol 23, 1483–1493 (2008). https://doi.org/10.1007/s00467-008-0794-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-008-0794-1