Abstract
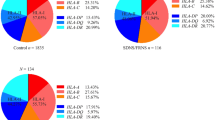
The study was aimed at investigating the association between MDR-1 genetic polymorphisms [C1236T, G2677T(A), C3435T] and parameters describing the clinical course and treatment response of childhood steroid-responsive nephrotic syndrome (SRNS). Three MDR-1 genetic markers were analyzed in 108 children diagnosed with SRNS and in 135 healthy controls with neither allergic nor renal disease. All subjects were genotyped by PCR-restriction fragment length polymorphism (RFLP) analysis, and an EM algorithm-based analysis was utilized to estimate haplotype frequencies. As expected, there was no difference in genotypic and allelic distribution between and among SRNS patients and healthy children. However, all individual polymorphisms were strongly associated with time to response to initial prednisone therapy. The frequencies of the mutated alleles were higher in late responders (time to remission: >7 days) to oral prednisone (0.53, 0.52,0.66) than in early responders (time to remission: <7 days; 0.24, 0.19, 0.32), with all p values <0.001 for positions 1236, 2677 and 3435, respectively). Odds ratios (ORs) reflecting the strength of the associations were as follows: 6.79 (95% CI:1.96– 23.54) for 1236 T/T, 13.7 (95% CI:2.78–67) for 2677 T/T and 9.92 (95% CI: 3.01–32.71) for 3435 T/T as compared to the respective-wild type homozygotes. The TTT haplotype was similarly found to be significantly associated with late oral steroid response (0.49 vs. 0.19, p=0.0003). Variants 1236T, 2677TA and 3435T identify patients that respond slower to oral prednisone. Although the functional properties of the substitutions investigated here are still to be determined, our findings may be a small step toward the optimization of immunosuppressive therapy in SRNS children.

Similar content being viewed by others
References
Hogg RJ, Portman RJ, Milliner D, Lemley KV, Eddy A, Ingelfinger J (2000) Evaluation and management of proteinuria and nephrotic syndrome in children: recommendations from a pediatric nephrology panel established at the National Kidney Foundation conference on proteinuria, albuminuria, risk, assessment, detection, and elimination (PARADE). Pediatrics 105:1242–1249
Ahmad H, Tejani A (2000) Predictive value of repeat renal biopsies in children with nephrotic syndrome. Nephron 84:342–346
Lande MB, Gullion C, Hogg RJ, Gauthier B, Shah B, Leonard MB, Bonilla-Felix M, Nash M, Roy S 3rd, Strife CF, Arbus G (2003) Long versus standard initial steroid therapy for children with the nephrotic syndrome. A report from the Southwest Pediatric Nephrology Study Group. Pediatr Nephrol 18:342–634
Sakaeda, T, Nakamura T, Okumura K (2004) Pharmacogenetics of drug transporters and its impact on the pharmacotherapy. Curr Top Med Chem 4:1385–1398
Dilger K, Schwab M, Fromm MF (2004) Identification of budesonide and prednisone as substrates of the intestinal drug efflux pump P-glycoprotein. Inflamm Bowel Dis 10:578–583
Farrell RJ, Menconi MJ, Keates AC, Kelly CP (2002) P-glycoprotein-170 inhibition significantly reduces cortisol and cyclosporin efflux from human intestinal epithelial cells and T lymphocytes. Aliment Pharmacol Ther 16:1021–1031
Klimecki WT, Futscher BW, Grogan TM, Dalton WS (1994) P-glycoprotein expression and function in circulating blood cells from normal volunteers. Blood 83:2451–2458
Stachowski J, Zanker CB, Runowski D, Zaniew M, Peszko A, Medynska A, Zwolinska D, Rogowska-Kalisz A, Hyla-Klekot L, Szprygner K, Weglarska J, Sieniawska M, Musial W, Maciejewski J, Baldamus CA (2000) Resistance to therapy in primary nephrotic syndrome: effect of MDR1 gene activity. Pol Merkuriusz Lek 8:218–221
Richaud-Patin Y, Soto-Vega E, Jakez-Ocampo J, Llorente L (2004) P-glycoprotein in autoimmune diseases. Autoimmun Rev 3:188–192
Tsujimura S, Saito K, Nakayamada S, Nakano K, Tsukada J, Kohno K, Tanaka Y (2004) Transcriptional regulation of multidrug resistance-1 gene by interleukin-2 in lymphocytes. Genes Cells 9:1265–1273
Raghu G, Park SW, Roninson IB, Mechetner EB (1996) Monoclonal antibodies against P-glycoprotein, an MDR1 gene product, inhibit interleukin-2 release from PHA-activated lymphocytes. Exp Hematol 24:1258–1264
Gollapud S, Gupta S (2001) Anti-P-glycoprotein antibody-induced apoptosis of activated peripheral blood lymphocytes: a possible role of P-glycoprotein in lymphocyte survival. J Clin Immunol 21:420–340
Ieiri I, Takane H, Otsubo K (2004) The MDR1 (ABCB1) gene polymorphism and its clinical implications. Clin Pharmacokinet 43:553–576
Woodahl EL, Ho RJ (2004) The role of MDR1 genetic polymorphisms in interindividual variability in P-glycoprotein expression and function. Curr Drug Metab 5:11–19
Soranzo N, Cavalleri GL, Weale ME, Wood NW, Depondt C, Marguerie R, Sisodiya SM, Goldstein DB (2004) Identifying candidate causal variants responsible for altered activity of the ABCB1 multidrug resistance gene. Genome Res 14:1333–1344
Kabuki N, Okugawa T, Hayakawa H, Tomizawa S, Kasahara T, Uchiyama M (1998) Influence of age at onset on the outcome of steroid-sensitive nephrotic syndrome. Pediatr Nephrol 12:467–470
Constantinescu AR, Shah HB, Foote EF, Weiss LS (2000) Predicting first-year relapses in children with nephrotic syndrome. Pediatrics 105:492–495
Takeda A, Takimoto H, Mizusawa Y, Simoda M (2001) Prediction of subsequent relapse in children with steroid-sensitive nephrotic syndrome. Pediatr Nephrol 16:888–893
Nasi M. Aedelmann CM, Berstein J, Barnett HL (1992) The nephrotic syndrome. In: Edelmann CM, Berstein J, Meadow SR, Spitzer A, Travis LB (eds) Pediatric kidney disease, vol 2. Little, Brown and Company, Boston, pp 1247–1266
International Study of Kidney Disease in Children (1982) Early identification of frequent relapsers among children with minimal change nephrotic syndrome. A report of the International Study of Kidney Disease in Children. J Pediatr 101:514–518
Report of Arbeitsgemeinschaft für Pädiatrische Nephrologie (1987) Cyclophosphamide treatment of steroid dependent nephrotic syndrome: comparison of eight week with 12 week course. Report of Arbeitsgemeinschaft für Pädiatrische Nephrologie. Arch Dis Child 62:1102–1106
Cascorbi I, Gerloff T, Johne A, Meisel C, Hoffmeyer S, Schwab M, Schaeffeler E, Eichelbaum M, Brinkmann U, Roots I (2001) Frequency of single nucleotide polymorphisms in the P-glycoprotein drug transporter MDR1 gene in white subjects. Clin Pharmacol Ther 69:169–174
Imani S, Jusko WJ, Steiner R (1999) Diltiazem retards the metabolism of oral prednisone with effects on T-cell markers. Pediatr Transplant 3:126–130
Rostin M, Barthe P, Houin G, Alvinerie M, Bouissou F (1990) Pharmacokinetics of prednisolone in children with the nephrotic syndrome. Pediatr Nephrol 4:470–473
Gatti G, Perucca E, Frigo GM, Notarangelo LD, Barberis L, Martini A (1984) Pharmacokinetics of prednisone and its metabolite prednisolone in children with nephrotic syndrome during the active phase and in remission. Br J Clin Pharmacol 17:423–431
Corrigan CJ, Brown PH, Barnes NC, Szefler SJ, Tsai JJ, Frew AJ, Kay AB (1991) Glucocorticoid resistance in chronic asthma. Glucocorticoid pharmacokinetics, glucocorticoid receptor characteristics, and inhibition of peripheral blood T cell proliferation by glucocorticoids in vitro. Am Rev Respir Dis 144:1016–1025
Magee MH, Blum RA, Lates CD, Jusko WJ (2002) Pharmacokinetic /pharmacodynamic model for prednisolone inhibition of whole blood lymphocyte proliferation. Br J Clin Pharmacol 53:474–484
Tsujimura S, Saito K, Nakayamada S, Nakano K, Tanaka Y (2005) Clinical relevance of the expression of P-glycoprotein on peripheral blood lymphocytes to steroid resistance in patients with systemic lupus erythematosus. Arthritis Rheum 52:1676–1683
Zheng H, Webber S, Zeevi A, Schuetz E, Zhang J, Lamba J, Bowman P, Burckart GJ (2002) The MDR1 polymorphisms at exons 21 and 26 predict steroid weaning in pediatric heart transplant patients. Hum Immunol 63:765–770
Asano T, Takahashi KA, Fujioka M, Inoue S, Okamoto M, Sugioka N, Nishino H, Tanaka T, Hirota Y, Kubo T (2003) ABCB1 C3435T and G2677T/A polymorphism decreased the risk for steroid-induced osteonecrosis of the femoral head after kidney transplantation. Pharmacogenetics 13:675–682
Sakaeda T (2005) MDR1 genotype-related pharmacokinetics: fact or fiction? Drug Metab Pharmacokinet 20:391–414
Lamba J, Strom S, Venkataramanan R, Thummel KE, Lin YS, Liu W, Cheng C, Lamba V, Watkins PB, Schuetz E (2006) MDR1 genotype is associated with hepatic cytochrome P450 3A4 basal and induction phenotype. Clin Pharmacol Ther 79: 325–338
Kurzawski M, Pawlik A, Gornik W, Drozdzik M (2006) Frequency of common MDR1 gene variants in a Polish population. Pharmacol Rep 58:35–40
Kim RB, Leake BF, Choo EF, Dresser GK, Kubba SV, Schwarz UI, Taylor A, Xie HG, McKinsey J, Zhou S, Lan LB, Schuetz JD, Schuetz EG, Wilkinson GR (2001) Identification of functionally variant MDR1 alleles among European Americans and African Americans. Clin Pharmacol Ther 70:189–199
Kroetz DL, Pauli-Magnus C, Hodges LM, Huang CC, Kawamoto M, Johns SJ, Stryke D, Ferrin TE, DeYoung J, Taylor T, Carlson EJ, Herskowitz I, Giacomini KM, Clark AG (2003) Pharmacogenetics of Membrane Transporters Investigators. Sequence diversity and haplotype structure in the human ABCB1 (MDR1, multidrug resistance transporter) gene. Pharmacogenetics 13:481–494
Morita N, Yasumori T, Nakayama K (2003) Human MDR1 polymorphism: G2677T/A and C3435T have no effect on MDR1 transport activities. Biochem Pharmacol 65:1843–1852
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wasilewska, A., Zalewski, G., Chyczewski, L. et al. MDR-1 gene polymorphisms and clinical course of steroid-responsive nephrotic syndrome in children. Pediatr Nephrol 22, 44–51 (2007). https://doi.org/10.1007/s00467-006-0275-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0275-3