Abstract
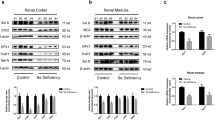
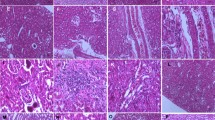
Selenium (Se) deficiency reduces glutathione peroxidase (GPx) activity, resulting in increased oxidative stress. We examined how Se deficiency induces renal injury via oxidative stress over time during the Se-deficient period. Seventy-two male Wistar rats were divided into two groups and fed either a control or Se-deficient diet. Rats were sacrificed on weeks 1, 2, 4, 6, 9, and 12. Blood and urine samples were collected, and the kidneys were removed. Urinalysis was performed, and creatinine clearance (Ccr) was calculated. Expressions of cellular GPx (cGPx) and phospholipid hydroperoxidase GPx (PHGPx) mRNA and GPx activity were measured. Histology was evaluated by light microscopy with immunohistochemistry for 4-hydroxy-2-nonenal (HNE) and vimentin. The Se-deficient diet caused significant decreases in GPx activity and cGPx mRNA expression but no change in PHGPx mRNA, together with significant proteinuria and glucosuria and slight decline in Ccr. The Se-deficient diet induced calcification in the kidney and increased the distribution of HNE and vimentin immunostaining in proximal tubuli, particularly around the outer medulla stripe. However, the histological damage did not progress after 6 weeks of deficiency. Se deficiency induces proteinuria and glucosuria with renal calcification, which may be primarily induced by injury of proximal tubuli via oxidative stress.






Similar content being viewed by others
References
Baliga R, Baliga M, Shah SV (1992) Effect of selenium-deficient diet in experimental glomerular disease. Am J Physiol 263:F56–F61
Nath KA, Salahudeen AK (1990) Induction of renal growth and injury in the intact rat kidney by dietary deficiency of antioxidants. J Clin Invest 86:1179–1192
Reddi AS, Bollineni JS (2001) Selenium-deficient diet induces renal oxidative stress and injury via TGF-β 1 in normal and diabetic rats. Kidney Int 59:1342–1353
Alftham G, Kumpulainen J (1982) Determination of selenium in small volume of blood plasma and serum by electrothermal atomic absorption spectrometry. Ann Chim Acta 140:221–227
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
L’Abbe MR, Trick KD (1994) Changes in pancreatic glutathione peroxidase and superoxide dismutase activities in the prediabetic diabetes-prone BB rat. Proc Soc Exp Biol Med 207:206–212
Imai H, Nakagawa Y (2003) Biological significance of phospholipids hydroperoxide glutathione peroxidase (PHGPx, GPX4) in mammalian cells. Free Radic Biol Med 34:145–169
Chen MM, Lam A, Abraham JA, Schreiner GF, Joly AH (2000) CTGF expression is induced by TGF-β in cardiac fibroblasts and cardiac myocytes: a potential role in heart fibrosis. J Mol Cell Cardiol 32:1805–1819
Bermano G, Nicol F, Dyer JA, Sunde RA, Beckett GJ, Arthur JR, Hesketh JE (1995) Tissue-specific regulation of selenoenzyme gene expression during selenium deficiency in rats. Biochem J 311:425–430
Nakane T, Asayama K, Kodera K, Hayashide H, Uchida N, Nakazawa S (1998) Effect of selenium deficiency on cellular and extracellular glutathione peroxidase: immunochemical detection and mRNA analysis in rat kidney and serum. Free Radial Biol Med 25:504–511
Pedraza-Chaverri J, Arevalo AE, Hernandez-Pando R, Larriva-Sahd J (1995) Effect of dietry antioxidants on puromycin aminonucleoside nephritic syndrome. Int J Biochem Cell Biol 27:683–691
Mylona-Karayanni C, Gourgiotis D, Bossios A, Kamper EF (2006) Oxidative stress and adhesion molecules in children with type 1 diabetes mellitus: a possible link. Pediatr Diabetes 7:51–59
Muthukumar A, Selvam R (1997) Renal injury mediated calcium oxalate nephrolithiasis: role of lipid peroxidation. Ren Fail 19:401–408
Khan SR, Hackett RL (1993) Hyperoxaluria, enzymuria and nephrolithiasis. Contrib Nephrol 101:190–193
Fukuda A, Osawa T, Hitomi K, Uchida K (1996) 4-hydroxy-2-nonenal cytotoxicity in renal proximal tubular cells. Protein modification and redox alteration. Arch Biochem Biophys 333:419–426
Bartsch H, Nair J, Velic I (1997) Etheno-DNA base adducts as tools in human cancer aetiology and chemoprevention. Eur J Cancer Prev 6:529–534
Toyokuni S, Miyake N, Hirai H, Hagiwara M, Kawakishi S, Osawa T, Uchida K (1995) The monoclonal antibody specific for the 4-hydroxy-2-nonenal histidine adducts. FEBS Lett 359:189–191
Tolstrong GV, Shoeman RL, Traub U, Traub P (2001) Role of intermediate filament protein vimentin in delaying senescence and in the spontaneous immortalization of mouse embryo fibroblasts. DNA Cell Biol 20:509–529
Belichenko I, Morishima N, Separovic D (2001) Caspase-resistant vimentin suppresses apoptosis after photodynamic treatment with a silicon phthalocyanine in Jurkat cells. Arch Biochem Biophys 390:57–63
Frei B, Winterhalter KH, Richter C (1985) Mechanism of alloxan-induced calcium release from rat liver mitochondria. J Biol Chem 260:7394–7401
Isaka Y, Fujiwara Y, Ueda N, Kanea Y, Kamada T, Imai E (1993) Glomerulosclerosis induced by in vitro transfection of transforming growth factor-β or platelet-derived growth factor gene into the rat kidney. J Clin Invest 92:2597–2601
Kopp JB, Factor VM, Mozes M, Nagy P, Sanderson N, Bottinger EP, Klotman PE, Thorgeirsson SS (1996) Transgenic mice with increased plasma levels of TGF-β 1 develop progressive renal disease. Lab Invest 74:991–1003
Roberts AB, McCune BK, Sporn MB (1992) TGF-β: regulation of extracellular matrix. Kidney Int 41:557–559
Basile DP (1999) The transforming growth factor beta system in kidney disease and repair: recent progress and future directions. Curr Opin Nephrol Hypertens 8:21–30
Maiorino M, Thomas JP, Girotti AW, Ursini F (1991) Reactivity of phospholipid hydroperoxide glutathione peroxidase with membrane and lipoprotein lipid hydroperoxides. Free Radic Res Commun 12–13:131–135
Thomas JP, Maiorino M, Ursini F, Girotti AW (1990) Protective action of phospholipids hydroperoxide glutathione peroxidase against membrane-damaging lipid peroxidation. In situ reduction of phospholipids and cholesterol hydroperoxides. J Biol Chem 265:454–461
Alissa EM, Bahijri SM, Ferns GA (2003) The controversy surrounding selenium and cardiovascular disease: a review of the evidence. Med Sci Monit 9:9–18
Becker DJ, Reul B, Ozcelikay AT, Buchet JP, Henquin J-C, Brichard SM (1996) Oral selenate improves glucose homeostasis and partly reverse abnormal expression of liver glycolytic and gluconeogenic enzymes in diabetic rats. Diabetologia 39:3–11
Battell ML, Delgatty HLM, McNeill JH (1998) Sodium selenate corrects glucose tolerance and heart function in STZ diabetic rats. Mol Cell Biochem 179:27–34
Danda RS, Habiba NM, Rincon-Choles H, Bhandari BK, Barnes JL, Abboud HE, Pergola PE (2005) Kidney involvement in a nongenetic rat model of type 2 diabetes. Kidney Int 68:2562–2571
Nath KA, Grande J, Croatt A, Haugen J, Kim Y, Rusenberg ME (1998) Redox regulation of renal DNA synthesis, transforming growth factor-β 1 and collagen gene expression. Kidney Int 53:367–381
Deguchi T, Takamoto M, Uehara N, Lidup WE, Suenaga A, Otagiri M (2005) Renal clearance of endogenous hippurate correlates with expression levels of renal organic anion transporters in uremic rats. J Pharmacol Exp Ther 314:932–938
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fujieda, M., Naruse, K., Hamauzu, T. et al. Effect of selenium-deficient diet on tubular epithelium in normal rats. Pediatr Nephrol 22, 192–201 (2007). https://doi.org/10.1007/s00467-006-0266-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0266-4