Abstract
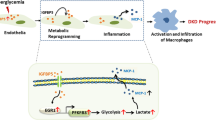
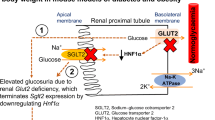
Changes in glucose transporter expression in glomerular cells occur early in diabetes. These changes, especially the GLUT1 increase in mesangial cells, appear to play a pathogenic role in the development of ECM expansion and perhaps other features of diabetic nephropathy. In addition, it appears that at least some diabetic patients may be predisposed to nephropathy because of polymorphisms in their GLUT1 genes. GLUT1 overexpression leads to increased glucose metabolic flux which in turn triggers the polyol pathway and activation of PKCα and Β1. Activation of these PKC isoforms can lead directly to AP-1 induced increases in fibronectin expression and ECM accumulation. Other, more novel effects of GLUT1 on cellular hypertrophy and injury could also promote changes of diabetic nephropathy. Strategies to prevent GLUT1 overexpression could ameliorate or prevent the progression of diabetic nephropathy.

Similar content being viewed by others
References
The DCCT Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329:977–986
Devaskar S, Mueckler M (1992) The mammalian glucose transporters. Pediatr Res 31:1–13
Kahn B, Flier J (1990) Regulation of glucose transporter gene regulation in vitro and in vivo. Diabetes Care 13:548–564
Klip A, Tsakiridis T, Marette A, Ortiz PA (1994) Regulation of expression of glucose transporters by glucose: a review of studies in vivo and in cell cultures. FASEB J 8:43–53
Mauer SM, Steffes MW, Ellis EN, Sutherland DE, Brown DM, Goetz FC (1984) Structural-functional relationships in diabetic nephropathy. J Clin Invest 74:1143–1155
Meyer TW, Bennett PH, Nelson RG (1999) Podocyte number predicts long-term urinary albumin excretion in Pima Indians with type II diabetes and microalbuminuria. Diabetologia 42:1341–1344
Pagtalunan ME, Miller PL, Jumping-Eagle S, Nelson RG, Myers BD, Rennke HG, Coplon NS, Sun L, Meyer TW (1997) Podocyte loss and progressive glomerular injury in type II diabetes. J Clin Invest 99:342–348
Brosius FC 3rd, Briggs JP, Marcus RG, Barac-Nieto M, Charron MJ (1992) Insulin-responsive glucose transporter expression in renal microvessels and glomeruli. Kidney Int 42:1086–1092
Heilig C, Zaloga C, Lee M, Zhao X, Riser B, Brosius F, Cortes P (1995) Immunogold localization of high affinity GLUT isoforms in normal rat kidney. Lab Invest 73:674–684
Gregoire F (1975) Oxidative metabolism of the normal rat glomerulus. Kidney Int 7:86–93
Coward RJM, Welsh GI, Holman GD, Kerjaschki D, Tavare MJ, Mathieson PW, Saleem MA (2003) Human podocytes rapidly utilize glucose by both GLUT1 and GLUT4 in response to insulin, with significant differences in glucose transporter levels occurring in diabetic nephropathy. J Am Soc Nephrol 14:388A (abstract)
Schiffer M, Ranalletta M, Susztak K, Charron JM, Bottinger EP (2003) Localization of the GLUT8 glucose transporter in the kidney and regulation in vivo in non-diabetic and diabetic conditions. J Am Soc Nephrol 14:308A
Wakisaka M, He Q, Spiro M, Spiro R (1995) Glucose entry into mesangial cells is mediated by both Na+-coupled and facilitative glucose transporters. Diabetalogia 38:291–297
Marcus RG, England R, Nguyen K, Charron MJ, Briggs JP, Brosius FC 3rd (1994) Altered renal expression of the insulin-responsive glucose transporter GLUT4 in experimental diabetes mellitus. Am J Physiol 267:F816–824
Heilig CW, Liu Y, England RL, Freytag SO, Gilbert JD, Heilig KO, Zhu M, Concepcion LA, Brosius FC 3rd (1997) d -Glucose stimulates mesangial cell GLUT1 expression and basal and IGF-I-sensitive glucose uptake in rat mesangial cells: implications for diabetic nephropathy. Diabetes 46:1030–1039
D’Agord Schaan B, Lacchini S, Bertoluci MC, Irigoyen MC, Machado UF, Schmid H (2001) Increased renal GLUT1 abundance and urinary TGF-beta 1 in streptozotocin-induced diabetic rats: implications for the development of nephropathy complicating diabetes. Horm Metab Res 33:664–669
Chen S, Heilig K, Brosius F III, Heilig C (2003) Diabetes increases glomerular GLUT1 and antisense-GLUT1 protects against diabetic glomerulosclerosis. J Am Soc Nephrol 14:46A
Heilig CW, Concepcion LA, Riser BL, Freytag SO, Zhu M, Cortes P (1995) Overexpression of glucose transporters in rat mesangial cells cultured in a normal glucose milieu mimics the diabetic phenotype. J Clin Invest 96:1802–1814
Heilig CW, Kreisbergh JI, Freytag S, Murakami T, Ebina Y, Guo L, Heilig K, Jin Y, Henry D, Brosius FC III (2001) Antisense-GLUT1 protects mesangial cells from d -glucose-induced transporter and fibronectin expression. Am J Physiol 280:F657–666
Gunning P, Leavitt J, Muscat G, Ng SY, Kedes L (1987) A human beta-actin expression vector system directs high-level accumulation of antisense transcripts. Proc Natl Acad Sci U S A 84:4831–4835
Oberley TD, Coursin DB, Cihla HP, Oberley LW, el-Sayyad N, Ho YS (1993) Immunolocalization of manganese superoxide dismutase in normal and transgenic mice expressing the human enzyme. Histochem J 25:267–279
Heilig KO, Chen S, Xiang M, Brosius FC, Heilig CW (2004) Transgenic overexpression of GLUT1 in glomeruli produces features of diabetic nephropathy in mice. J Am Soc Nephrol (abstract)
Hodgkinson AD, Millward BA, Demaine AG (2001) Polymorphisms of the glucose transporter (GLUT1) gene are associated with diabetic nephropathy. Kidney Int 59:985–989
Ng DP, Canani L, Araki S, Smiles A, Moczulski D, Warram JH, Krolewski AS (2002) Minor effect of GLUT1 polymorphisms on susceptibility to diabetic nephropathy in type 1 diabetes. Diabetes 51:2264–2269
Vaulont S, Kahn A (1994) Transcriptional control of metabolic regulation genes by carbohydrates. FASEB J 8:28–35
Hsu CC, Kao WH, Steffes M, Brancati F, Heilig C, Shuldiner A, Boerwinkle E, Coresh J (2004) Genetic variation of glucose transporter-1 (GLUT1) and albuminuria in the atherosclerosis risk in communities study. Diabetes 53:A200 (abstract)
Liu ZH, Guan TJ, Chen ZH, Li LS (1999) Glucose transporter (GLUT1) allele (XbaI-) associated with nephropathy in non-insulin-dependent diabetes mellitus. Kidney Int 55:1843–1848
Grzeszczak W, Moczulski DK, Zychma M, Zukowska-Szczechowska E, Trautsolt W, Szydlowska I (2001) Role of GLUT1 gene in susceptibility to diabetic nephropathy in type 2 diabetes. Kidney Int 59:631–636
Tarnow L, Grarup N, Hansen T, Parving HH, Pedersen O (2001) Diabetic microvascular complications are not associated with two polymorphisms in the GLUT-1 and PC-1 genes regulating glucose metabolism in Caucasian type 1 diabetic patients. Nephrol Dial Transplant 16:1653–1656
Henry DN, Buski JV, Concepcion LA, Brosius FC III, Heilig CW (1999) Glucose transporters control the expression of aldose reductase, PKCα, and GLUT1 genes in mesangial cells in vitro. Am J Physiol 277: F97–F104
Weigert C, Brodbeck K, Brosius FC III, Huber M, Lehmann R, Friess U, Aulwurm S, Häring H, Schleicher ED, Heilig CW (2003) Evidence for a novel TGF-β1-independent mechanism of fibrosis in mesangial cells overexpressing glucose transporters. Diabetes 52:527–535
Whiteside CI, Dlugosz JA (2002) Mesangial cell protein kinase C isozyme activation in the diabetic milieu. Am J Physiol Renal Physiol 282:F975–980
Inoki K, Haneda M, Maeda S, Koya D, Kikkawa R (1999) TGF-beta 1 stimulates glucose uptake by enhancing GLUT1 expression in mesangial cells. Kidney Int 55:1704–1712
Nose A, Mori Y, Uchiyama-Tanaka Y, Kishimoto N, Maruyama K, Matsubara H, Iwasaka T (2003) Regulation of glucose transporter (GLUT1) gene expression by angiotensin II in mesangial cells: involvement of HB-EGF and EGF receptor transactivation. Hypertens Res 26:67–73
Morissette MR, Howes AL, Zhang T, Brown JH (2003) Upregulation of GLUT1 expression is necessary for hypertrophy and survival in neonatal rat cardiomyocytes. J Mol Cell Cardiol 35:1217–1227
Giorgino F, de Robertis O, Laviola L, Montrone C, Perrini S, McCowen KC, Smith RJ (2000) The sentrin-conjugating enzyme mUbc9 interacts with GLUT4 and GLUT1 glucose transporters and regulates transporter levels in skeletal muscle cells. Proc Natl Acad Sci U S A 97:1125–1130
Bandyopadhyay G, Sajan MP, Kanoh Y, Standaert ML, Burke TR Jr, Quon MJ, Reed BC, Dikic I, Noel LE, Newgard CB, Farese R (2000) Glucose activates mitogen-activated protein kinase (extracellular signal-regulated kinase) through proline-rich tyrosine kinase-2 and the Glut1 glucose transporter. J Biol Chem 275:40817–40826
Bunn RC, Jensen MA, Reed BC (1999) Protein interactions with the glucose transporter binding protein GLUT1CBP that provide a link between GLUT1 and the cytoskeleton. Mol Biol Cell 10:819–832
Zhang JZ, Hayashi H, Ebina Y, Prohaska R, Ismail-Beigi F (1999) Association of stomatin (band 7.2b) with Glut1 glucose transporter. Arch Biochem Biophys 372:173–178
Plas DR, Talapatra S, Edinger AL, Rathmell JC, Thompson CB (2001) Akt and Bcl-xL promote growth factor-independent survival through distinct effects on mitochondrial physiology. J Biol Chem 276:12041–12048
Gottlob K, Majewski N, Kennedy S, Kandel E, Robey RB, Hay N (2001) Inhibition of early apoptotic events by Akt/PKB is dependent on the first committed step of glycolysis and hexokinase. Genes Dev 15:1406–1418
Sakyo T, Kitagawa T (2002) Differential localization of glucose transporter isoforms in non-polarized mammalian cells: distribution of GLUT1 but not GLUT3 to detergent-resistant membrane domains. Biochim Biophys Acta 1567:165–175
Acknowledgements
These studies were supported by a grant from the Juvenile Diabetes Research Foundation Center for Excellence at the University of Michigan (Award N001280, Project 7), a Juvenile Diabetes Research Foundation Innovative Grant (5-2000-892), and the Animal Models of Diabetic Complications Consortium (National Institutes of Health, UO1 DK60994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brosius, F.C., Heilig, C.W. Glucose transporters in diabetic nephropathy. Pediatr Nephrol 20, 447–451 (2005). https://doi.org/10.1007/s00467-004-1748-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-004-1748-x