Abstract
Background
Suture dehiscence is one of the most feared postoperative complications. Correct intestinal vascularization is essential for its prevention. Indocyanine green (ICG) is one of the methods used to assess vascularization, but this assessment is usually subjective. Our group designed the SERGREEN program to obtain an objective measurement of the degree of vascularization. We do not know how long after ICG administration the fluorescence of the tissues should be evaluated, or how far away the measurement should be performed. The aim of this study is to establish the optimal moment and distance for analyzing the fluorescence saturation of ICG.
Methods
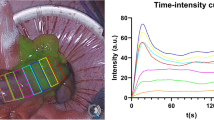
Prospective observational study in patients undergoing elective laparoscopic colorectal surgery. The optimal time for ICG analysis was tested in a sample of 20 patients (10 right colon and 10 left colon), and the optimal distance in a sample of ten patients. ICG was administered intravenously, and colon vascularization was quantified using SERGREEN; RGB (Red, Green, Blue) encoding was used. The intensity curve of the ICG was analyzed for ten minutes after its administration. Distances of 1, 3, and 5 cm were tested.
Results
The intensity of fluorescence increased until 1.5 min after ICG administration (reaching figures of 112.49 in the right colon and 93.95 in the left). It then remained fairly stable until 3.5 min (98.49 in the right and 83.35 in the left), at which point it began to decrease gradually.
ICG saturation was inversely proportional to the distance between the camera and the tissue. The best distance was 5 cm, where the confidence interval was narrower [CI 86.66–87.53].
Conclusion
The optimal time for determining ICG in the colon is between 1.5 and 3.5 min, in both right and left colon. The optimal distance is 5 cm. This information will help to establish parameters of comparison in normal and pathological situations.


Similar content being viewed by others
References
Kingham TP, Pachter HL (2009) Colonic anastomotic leak: risk factors, diagnosis, and treatment. J Am Coll Surg. 208(2):269–78. https://doi.org/10.1016/j.jamcollsurg.2008.10.015
Boni L, David G, Dionigi G, Rausei S, Cassinotti E, Fingerhut A (2016) Indocyanine green-enhanced fluorescence to assess bowel perfusion during laparoscopic colorectal resection. Surg Endosc 30(7):2736–2742. https://doi.org/10.1007/s00464-015-4540-z
Boyle NH, Manifold D, Jordan MH, Mason RC (2000) Intraoperative assessment of colonic perfusion using scanning laser doppler flowmetry during colonic resection. J Am Coll Surg 191(5):504–510
Vignali A, Gianotti L, Braga M, Radaelli G, Malvezzi L, Di Carlo V (1998) Altered microperfusion at the rectal stump is predictive for rectal anastomotic leak. Dis Colon Rectum 43(1):76–82
Diana M, Agnus V, Halvax P, Liu Y-Y, Dallemagne B, Schlagowski A-I, Geny B, Diemunsch P, Lindner V, Marescaux J (2015) Intraoperative fluorescence-based enhanced reality laparoscopic real-time imaging to assess bowel perfusion at the anastomotic site in an experimental model. Br J Surg 102(2):169–176
Protyniak B, Dinallo AM, Boyan WP Jr, Dressner RM, Arvanitis ML (2015) Intraoperative indocyanine green fluorescence angiography—An objective evaluation of anastomotic perfusion in colorectal surgery. Am Surg 81(6):580–584
Sherwinter DA, Gallagher J, Donkar T (2012) Intra-operative transanal near infrared imaging of colorectal anastomotic perfusion: a feasibility study. Colorectal Dis 15:91–96
Serra-Aracil X, Garcia-Nalda A, Serra-Gomez B, Serra-Gomez A, Mora-Lopez L, Pallisera-Lloveras A, Lucas-Guerrero V, Navarro-Soto S (2021) Experimental study of the quantification of indocyanine green fluorescence in ischemic and non-ischemnic anastomoses, using the SERGREEN software program. Res Sq. https://doi.org/10.21203/rs.3.rs-880156/v1
McCulloch P, Altman DG, Campbell WB, Flum DR, Glasziou P, Marshall JC, Nicholl J, Collaboration B, Aronson JK, Barkun JS, Blazeby JM, Boutron IC, Campbell WB, Clavien PA, Cook JA, Ergina PL, Feldman LS, Flum DR, Maddern GJ, Nicholl J, Reeves BC, Seiler CM, Strasberg SM, Meakins JL, Ashby D, Black N, Bunker J, Burton M, Campbell M, Chalkidou K, Chalmers I, de Leval M, Deeks J, Ergina PL, Grant A, Gray M, Greenhalgh R, Jenicek M, Kehoe S, Lilford R, Littlejohns P, Loke Y, Madhock R, McPherson K, Meakins J, Rothwell P, Summerskill B, Taggart D, Tekkis P, Thompson M, Treasure T, Trohler U, Vandenbroucke J (2009) No surgical innovation without evaluation: the IDEAL recommendations. Lancet 374:1105–1112
Hirst A, Philippou Y, Blazeby J, Campbell B, Campbell M, Feinberg J, Rovers M, Blencowe N, Pennell C, Quinn T, Rogers W, Cook J, Kolias AG, Agha R, Dahm P, Sedrakyan A, McCulloch P (2019) No surgical innovation without evaluation: evolution and further development of the IDEAL framework and recommendations. Ann Surg 269:211–220
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, Initiative STROBE (2007) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 147:573–577
Urbanavičius L, Pattyn P, Van de Putte D, Venskutonis D (2011) How to assess intestinal viability during surgery: a review of techniques. World J Gastrointest Surg 3(5):59–69
Lütken CD, Achiam MP, Svendsen MB, Boni L, Nerup N (2020) Optimizing quantitative fluorescence angiography for visceral perfusion assessment. Surg Endoscopy 34:5223–5233
Lütken CD, Achiam MP, Osterkamp J, Svendsen MB, Nerup N (2021) Quantification of fluorescence angiography: toward a reliable intraoperative assessment of tissue perfusion—A narrative review. Langenbeck’s Arch Surg 406:251–259
Acknowledgements
We thank all the members of the Coloproctology Unit of the Department of General Surgery, Borja Serra-Gomez and Alvaro Serra-Gómez for their technical support with the SERGREEN program, and Michael Maudsley for his help with the English.
Author information
Authors and Affiliations
Contributions
All the authors agree to the submission of the paper. All the authors contributed to the design and writing of the paper.
Corresponding author
Ethics declarations
Disclosures
Xavier Serra-Aracil, Victoria Lucas-Guerrero, Albert Garcia-Nalda, Laura Mora-López, Anna Pallisera-Lloveras, Anna Serracant, and Salvador Navarro have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Serra-Aracil, X., Lucas-Guerrero, V., Garcia-Nalda, A. et al. When should indocyanine green be assessed in colorectal surgery, and at what distance from the tissue? Quantitative measurement using the SERGREEN program. Surg Endosc 36, 8943–8949 (2022). https://doi.org/10.1007/s00464-022-09343-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-022-09343-2