Abstract
Background
Duodenal gastrointestinal stromal tumors (GISTs) are rare, and reports on duodenal GIST bleeding are few. We analyzed the risk factors and clinical outcomes of hemorrhagic duodenal GISTs and compared them with those of gastric GISTs.
Methods
Primary duodenal GISTs surgically diagnosed between January 1998 and December 2017 were retrospectively reviewed. Furthermore, patients with duodenal GIST were compared with those with primary gastric GIST histopathologically diagnosed between January 1998 and May 2015 using previously published data.
Results
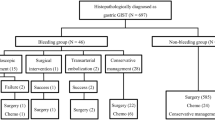
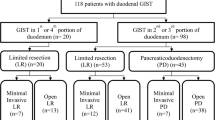
Of the 170 total patients with duodenal GISTs, 48 (28.2%) exhibited tumor bleeding. Endoscopic intervention, embolization, and non-interventional conservative treatment were performed for initial hemostasis in 17, 1, and 30 patients, respectively. The 5-year survival rate was 81.9% in the bleeding group and 89.4% in the non-bleeding group (P = 0.495). Multivariate analysis showed that p53 positivity was a significant risk factor for duodenal GIST bleeding (hazard ratio [HR] 2.781, P = 0.012), and age ≥ 60 years (HR 3.163, P = 0.027), a large maximum diameter (comparing four groups: < 2, 2–5, 5–10, and ≥ 10 cm), and mitotic count ≥ 5/high-power field (HPF) (HR 3.265, P = 0.032) were risk factors for overall survival. The incidence of bleeding was significantly higher in duodenal GISTs than in gastric GISTs (28.2% vs. 6.6%, P < 0.001), and the re-bleeding rate after endoscopic hemostasis was also higher in duodenal GISTs than in gastric GISTs (41.2% vs. 13.3%, P = 0.118).
Conclusion
In patients with duodenal GIST with old age, large tumor diameter, and mitotic count ≥ 5/HPF, a treatment plan should be established in consideration of the poor prognosis, although tumor bleeding does not adversely affect the prognosis. Duodenal GISTs have a higher incidence of tumor bleeding and re-bleeding rate after endoscopic hemostasis than gastric GISTs.



Similar content being viewed by others
References
Nilsson B, Bumming P, Meis-Kindblom JM, Oden A, Dortok A, Gustavsson B, Sablinska K, Kindblom LG (2005) Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era—a population-based study in western Sweden. Cancer 103(4):821–829. https://doi.org/10.1002/cncr.20862
Tran T, Davila JA, El-Serag HB (2005) The epidemiology of malignant gastrointestinal stromal tumors: an analysis of 1,458 cases from 1992 to 2000. Am J Gastroenterol 100(1):162–168. https://doi.org/10.1111/j.1572-0241.2005.40709.x
Schottenfeld D, Beebe-Dimmer JL, Vigneau FD (2009) The epidemiology and pathogenesis of neoplasia in the small intestine. Ann Epidemiol 19(1):58–69. https://doi.org/10.1016/j.annepidem.2008.10.004
Buchs NC, Bucher P, Gervaz P, Ostermann S, Pugin F, Morel P (2010) Segmental duodenectomy for gastrointestinal stromal tumor of the duodenum. World J Gastroenterol 16(22):2788–2792. https://doi.org/10.3748/wjg.v16.i22.2788
Miettinen M, Lasota J (2001) Gastrointestinal stromal tumors–definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis. Virchows Arch 438(1):1–12. https://doi.org/10.1007/s004280000338
Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, Miettinen M, O'Leary TJ, Remotti H, Rubin BP, Shmookler B, Sobin LH, Weiss SW (2002) Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol 33(5):459–465. https://doi.org/10.1053/hupa.2002.123545
Joensuu H, Fletcher C, Dimitrijevic S, Silberman S, Roberts P, Demetri G (2002) Management of malignant gastrointestinal stromal tumours. Lancet Oncol 3(11):655–664. https://doi.org/10.1016/S1470-2045(02)00899-9
Miettinen M, Sobin LH, Lasota J (2005) Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol 29(1):52–68. https://doi.org/10.1097/01.pas.0000146010.92933.de
Caterino S, Lorenzon L, Petrucciani N, Iannicelli E, Pilozzi E, Romiti A, Cavallini M, Ziparo V (2011) Gastrointestinal stromal tumors: correlation between symptoms at presentation, tumor location and prognostic factors in 47 consecutive patients. World J Surg Oncol 9:13. https://doi.org/10.1186/1477-7819-9-13
Park KS, Huh KC, Hwang I, Kwon JH, Kwon JG, Kim GH, Shin JE, Kim ES, Cho KB (2013) Multi-center study regarding the risk factors for bleeding in gastrointestinal stromal tumor. Hepatogastroenterology 60(122):235–239. https://doi.org/10.5754/hge12622
Liu Q, Li Y, Dong M, Kong F, Dong Q (2017) Gastrointestinal bleeding is an independent risk factor for poor prognosis in GIST patients. Biomed Res Int 2017:7152406. https://doi.org/10.1155/2017/7152406
Pih GY, Jeon SJ, Ahn JY, Na HK, Lee JH, Jung KW, Kim DH, Choi KD, Song HJ, Lee GH, Jung HY, Kim SO (2019) Clinical outcomes of upper gastrointestinal bleeding in patients with gastric gastrointestinal stromal tumor. Surg Endosc. https://doi.org/10.1007/s00464-019-06816-9
Miettinen M, Lasota J (2006) Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol 23(2):70–83. https://doi.org/10.1053/j.semdp.2006.09.001
Wasag B, Debiec-Rychter M, Pauwels P, Stul M, Vranckx H, Oosterom AV, Hagemeijer A, Sciot R (2004) Differential expression of KIT/PDGFRA mutant isoforms in epithelioid and mixed variants of gastrointestinal stromal tumors depends predominantly on the tumor site. Mod Pathol 17(8):889–894. https://doi.org/10.1038/modpathol.3800136
Watson RR, Binmoeller KF, Hamerski CM, Shergill AK, Shaw RE, Jaffee IM, Stewart L, Shah JN (2011) Yield and performance characteristics of endoscopic ultrasound-guided fine needle aspiration for diagnosing upper GI tract stromal tumors. Dig Dis Sci 56(6):1757–1762. https://doi.org/10.1007/s10620-011-1646-6
Rutkowski P, Nowecki ZI, Michej W, Debiec-Rychter M, Wozniak A, Limon J, Siedlecki J, Grzesiakowska U, Kakol M, Osuch C, Polkowski M, Gluszek S, Zurawski Z, Ruka W (2007) Risk criteria and prognostic factors for predicting recurrences after resection of primary gastrointestinal stromal tumor. Ann Surg Oncol 14(7):2018–2027. https://doi.org/10.1245/s10434-007-9377-9
Wozniak A, Rutkowski P, Piskorz A, Ciwoniuk M, Osuch C, Bylina E, Sygut J, Chosia M, Rys J, Urbanczyk K, Kruszewski W, Sowa P, Siedlecki J, Debiec-Rychter M, Limon J, GIST Polish Clinical Registry (2012) Prognostic value of KIT/PDGFRA mutations in gastrointestinal stromal tumours (GIST): Polish Clinical GIST Registry experience. Ann Oncol 23(2):353–360. https://doi.org/10.1093/annonc/mdr127
Mercadante S, Fusco F, Valle A, Fulfaro F, Casuccio A, Silvestro S, Donelli E (2004) Factors involved in gastrointestinal bleeding in advanced cancer patients followed at home. Support Care Cancer 12(2):95–98. https://doi.org/10.1007/s00520-003-0519-8
Kim YI, Choi IJ, Cho SJ, Lee JY, Kim CG, Kim MJ, Ryu KW, Kim YW, Park YI (2013) Outcome of endoscopic therapy for cancer bleeding in patients with unresectable gastric cancer. J Gastroenterol Hepatol 28(9):1489–1495. https://doi.org/10.1111/jgh.12262
Sheibani S, Kim JJ, Chen B, Park S, Saberi B, Keyashian K, Buxbaum J, Laine L (2013) Natural history of acute upper GI bleeding due to tumours: short-term success and long-term recurrence with or without endoscopic therapy. Aliment Pharmacol Ther 38(2):144–150. https://doi.org/10.1111/apt.12347
Seya T, Tanaka N, Yokoi K, Shinji S, Oaki Y, Tajiri T (2008) Life-threatening bleeding from gastrointestinal stromal tumor of the stomach. J Nippon Med Sch 75(5):306–311. https://doi.org/10.1272/jnms.75.306
Aboutaleb E, Kothari M, Damrah O, Canelo R (2009) c-KIT positive gastrointestinal stromal tumor presenting with acute bleeding in a patient with neurofibromatosis type 1: a case report. Int Semin Surg Oncol 6:17. https://doi.org/10.1186/1477-7800-6-17
Bianchi M, De Pascalis B, Koch M (2010) Jejunal GIST: an unusual cause of gastrointestinal bleeding. Dig Liver Dis 42(10):744. https://doi.org/10.1016/j.dld.2009.06.008
Anjiki H, Kamisawa T, Sanaka M, Ishii T, Kuyama Y (2010) Endoscopic hemostasis techniques for upper gastrointestinal hemorrhage: a review. World J Gastrointest Endosc 2(2):54–60. https://doi.org/10.4253/wjge.v2.i2.54
Hall PA, McCluggage WG (2006) Assessing p53 in clinical contexts: unlearned lessons and new perspectives. J Pathol 208(1):1–6. https://doi.org/10.1002/path.1913
Wong NA, Young R, Malcomson RD, Nayar AG, Jamieson LA, Save VE, Carey FA, Brewster DH, Han C, Al-Nafussi A (2003) Prognostic indicators for gastrointestinal stromal tumours: a clinicopathological and immunohistochemical study of 108 resected cases of the stomach. Histopathology 43(2):118–126. https://doi.org/10.1046/j.1365-2559.2003.01665.x
Hillemanns M, Pasold S, Bottcher K, Hofler H (1998) Prognostic factors of gastrointestinal stromal tumors of the stomach. Verh Dtsch Ges Pathol 82:261–266
Cunningham RE, Abbondanzo SL, Chu WS, Emory TS, Sobin LH, O'Leary TJ (2001) Apoptosis, bcl-2 expression, and p53 expression in gastrointestinal stromal/smooth muscle tumors. Appl Immunohistochem Mol Morphol 9(1):19–23. https://doi.org/10.1097/00129039-200103000-00005
Chang MS, Choe G, Kim WH, Kim YI (1998) Small intestinal stromal tumors: a clinicopathologic study of 31 tumors. Pathol Int 48(5):341–347. https://doi.org/10.1111/j.1440-1827.1998.tb03916.x
Giuliano K, Nagarajan N, Canner J, Najafian A, Wolfgang C, Schneider E, Meyer C, Lennon AM, Johnston FM, Ahuja N (2017) Gastric and small intestine gastrointestinal stromal tumors: do outcomes differ? J Surg Oncol 115(3):351–357. https://doi.org/10.1002/jso.24514
Liu Z, Zheng G, Liu J, Liu S, Xu G, Wang Q, Guo M, Lian X, Zhang H, Feng F (2018) Clinicopathological features, surgical strategy and prognosis of duodenal gastrointestinal stromal tumors: a series of 300 patients. BMC Cancer 18(1):563. https://doi.org/10.1186/s12885-018-4485-4
Iorio N, Sawaya RA, Friedenberg FK (2014) Review article: the biology, diagnosis and management of gastrointestinal stromal tumours. Aliment Pharmacol Ther 39(12):1376–1386. https://doi.org/10.1111/apt.12761
Miettinen M, Wang ZF, Lasota J (2009) DOG1 antibody in the differential diagnosis of gastrointestinal stromal tumors: a study of 1840 cases. Am J Surg Pathol 33(9):1401–1408. https://doi.org/10.1097/PAS.0b013e3181a90e1a
Valsangkar N, Sehdev A, Misra S, Zimmers TA, O'Neil BH, Koniaris LG (2015) Current management of gastrointestinal stromal tumors: surgery, current biomarkers, mutations, and therapy. Surgery 158(5):1149–1164. https://doi.org/10.1016/j.surg.2015.06.027
Author information
Authors and Affiliations
Contributions
GYP: analysis and interpretation of data, and drafting of the article. JYA: study conception and design, critical revision of the article for important intellectual content, and final approval of the article. HKN: final approval of the article. JHL: final approval of the article. KWJ: final approval of the article. DHK.: final approval of the article. KDC: final approval of the article. HJS.: final approval of the article. G.H.L.: final approval of the article. HYJ.: final approval of the article.
Corresponding author
Ethics declarations
Disclosures
Gyu Young Pih, Ji Yong Ahn, Ji Young Choi, Hee Kyong Na, Jeong Hoon Lee, Kee Wook Jung, Do Hoon Kim, Kee Don Choi, Ho June Song, Gin Hyug Lee, and Hwoon-Yong Jung have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pih, G.Y., Ahn, J.Y., Choi, J.Y. et al. Clinical outcomes of tumor bleeding in duodenal gastrointestinal stromal tumors: a 20-year single-center experience. Surg Endosc 35, 1190–1201 (2021). https://doi.org/10.1007/s00464-020-07486-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07486-8