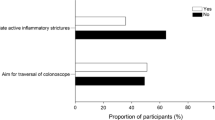
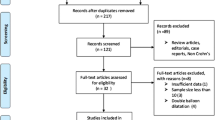
Abstract
Background
There is no information regarding the outcome of Crohn’s disease (CD) patients treated with endoscopic balloon dilation (EBD) in non-referral hospitals, nor on the efficacy of EBD in ulcerative colitis (UC). We report herein the results of the largest series published to date.
Aim
To assess the efficacy and safety of EBD for inflammatory bowel disease (IBD) stenosis performed in 19 hospitals with different levels of complexity and to determine factors related to therapeutic success.
Methods
We identified IBD patients undergoing EBD in the ENEIDA database. Efficacy of EBD was compared between CD and UC and between secondary and tertiary hospitals. Predictive factors of therapeutic success were assessed with multivariate analysis.
Results
Four-hundred dilations (41.2% anastomotic) were performed in 187 IBD patients (13 UC/Indeterminate colitis). Technical and therapeutic success per dilation was achieved in 79.5% and 55.3%, respectively. Therapeutic success per patient was achieved in 78.1% of cases (median follow-up: 40 months) with 49.7% requiring more than one dilation. No differences related to either diagnosis or hospital complexity was found. Technical success [OR 4.12 (95%CI 2.4–7.1)] and not receiving anti-TNF at the time of dilation [OR 1.7 (95% CI 1.1–2.6)] were independently related to therapeutic success per dilation. A stricture length ≤ 2 cm [HR 2.43 (95% CI 1.11–5.31)] was a predictive factor of long-term success per patient. The rate of major complications was 1.3%.
Conclusions
EBD can be performed with similar efficacy and safety in hospitals with differing levels of complexity and it might be a suitable treatment for UC with short stenosis. To achieve a technical success and the short length of the stenosis seem to be critical for long-term therapeutic success.


Similar content being viewed by others
Change history
18 June 2019
Javier P. Gisbert was listed incorrectly as Javier Pérez-Gisbert.
Abbreviations
- ASGE:
-
The American Society for Gastrointestinal Endoscopy
- CD:
-
Crohn’s disease
- COMBO:
-
Combined treatment (immunosuppressant plus biological treatment)
- EBD:
-
Endoscopic balloon dilation
- ENEIDA:
-
Estudio Nacional en Enfermedad Inflamatoria Intestinal sobre Determinantes genéticos y Ambientales (Spanish Study of Genetic and Environmental Determinants in Inflammatory Bowel Disease)
- GETECCU:
-
Grupo Español de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa (The Spanish Working Group in Crohn’s Disease and Colitis)
- IBD:
-
Inflammatory bowel disease
- IC:
-
Indeterminate colitis
- IFX:
-
Infliximab
- IMM:
-
Immunosuppressant’s alone
- IPAA:
-
Ileal pouch-anal anastomosis
- TNF:
-
Tumour Necrosis Factor
- UC:
-
Ulcerative colitis
References
Reimund JM, Wittersheim C, Dumont S, Muller CD, Kenney JS, Baumann R et al (1996) Increased production of tumour necrosis factor-alpha interleukin-1 beta, and interleukin-6 by morphologically normal intestinal biopsies from patients with Crohn’s disease. Gut 39(5):684–689
McKaig BC, Hughes K, Tighe PJ, Mahida YR (2002) Differential expression of TGF-beta isoforms by normal and inflammatory bowel disease intestinal myofibroblasts. Am J Physiol Cell Physiol 282(1):C172–C182
Rieder F, Lawrance IC, Leite A, Sans M (2011) Predictors of fibrostenotic Crohn’s disease. Inflamm Bowel Dis 17(9):2000–2007
Pucilowska JB, Williams KL, Lund PK (2010) Fibrogenesis IV. Fibrosis and inflammatory bowel disease: cellular mediators and animal models. Am J Physiol 279(4):653–659
Szabò H, Fiorino G, Spinelli A, Rovida S, Repici A, Malesci AC et al (2010) Review article: anti-fibrotic agents for the treatment of Crohn’s disease—lessons learnt from other diseases. Aliment Pharmacol Ther 31(2):189–201
Cosnes J, Gower-Rousseau C, Seksik P, Cortot A, Gower-Rousseau C, Seksik P et al (2011) Epidemiology and natural history of inflammatory bowel diseases. Gastroenterology 140(6):1785–1794
Solberg IC, Vatn MH, Høie O, Stray N, Sauar J, Jahnsen J et al (2007) Clinical course in Crohn’s disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol 5:1430–1438
Peyrin-Biroulet L, Loftus EV, Colombel J-F, Sandborn WJ (2010) The natural history of adult Crohn’s disease in population-based cohorts. Am J Gastroenterol 105(2):289–297
Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M (1990) Predictability of the postoperative course of Crohn’s disease. Gastroenterology 99:956–963
Wibmer AG, Kroesen AJ, Gröne J, Buhr H-J, Ritz J-P (2010) Comparison of strictureplasty and endoscopic balloon dilatation for stricturing Crohn’s disease–review of the literature. Int J Colorectal Dis 25(10):1149–1157
Hassan C, Zullo A, DeFrancesco V, Ierardi E, Giustini M, Pitidis A et al (2007) Systematic review: endoscopic dilatation in Crohn’s disease. Aliment Pharmacol Ther 26:1457–1464
Thienpont C, D’Hoore A, Vermeire S, Demedts I, Bisschops R, Coremans G et al (2010) Long-term outcome of endoscopic dilatation in patients with Crohn’s disease is not affected by disease activity or medical therapy. Gut 59(3):320–324
Gustavsson A, Magnuson A, Blomberg B, Andersson M, Halfvarson J, Tysk C (2012) Endoscopic dilation is an efficacious and safe treatment of intestinal strictures in Crohn’s disease. Aliment Pharmacol Ther 36(2):151–158
Bhalme M, Sarkar S, Lal S, Bodger K, Baker R, Willert RP (2014) Endoscopic balloon dilatation of Crohn’s disease strictures: results from a large United kingdom series. Inflamm Bowel Dis 20(2):265–270
Morar PS, Faiz O, Warusavitarne J, Brown S, Cohen R, Hind D et al (2015) Systematic review with meta-analysis: endoscopic balloon dilatation for Crohn’s disease strictures. Aliment Pharmacol Ther 42(10):1137–1148
Navaneethan U, Lourdusamy V, Njei B, Shen B (2016) Endoscopic balloon dilation in the management of strictures in Crohn???s disease: a systematic review and meta-analysis of non-randomized trials. Surg Endosc Other Interv Tech. 30(12):5434–5443
Atreja A, Aggarwal A, Dwivedi S, Rieder F, Lopez R, Lashner BA et al (2014) Safety and efficacy of endoscopic dilation for primary and anastomotic Crohn’s disease strictures. J Crohn’s Colitis. 8(5):392–400
Cotton PB, Eisen GM, Aabakken L, Baron TH, Hutter MM, Jacobson BC et al (2010) A lexicon for endoscopic adverse events: report of an ASGE workshop Gastrointest Endosc. Am Soc Gastrointest Endosc 71(3):446–454
Silverberg MS, Satsangi J, Ahmad T, Arnott IDR, Bernstein CN, Brant SR et al (2005) Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol 19(Suppl A):5A–36A
Dear KLE, Hunter JO (2001) Colonoscopic hydrostatic balloon dilatation of Crohn’ s strictures. J Clin Gastroenterol 33(4):315–318
Blomberg B, Rolny P, Järnerot G (1991) Endoscopic treatment of anastomotic strictures in Crohn’s disease. Endoscopy. 23(4):195–198
Ferlitsch A, Reinisch W, Püpök A, Dejaco C, Schillinger M, Schöfl R et al (2006) Safety and efficacy of endoscopic balloon dilation for treatment of Crohn’s disease strictures. Endoscopy 38(5):483–487
Thomas-Gibson S, Brooker JC, Hayward CMM, Shah SG, Williams CB, Saunders BP (2003) Colonoscopic balloon dilation of Crohn’s strictures: a review of long-term outcomes. Eur J Gastroenterol Hepatol 15(5):485–488
Kaplan GG, McCarthy EP, Ayanian JZ, Korzenik J, Hodin R, Sands BE (2008) Impact of hospital volume on postoperative morbidity and mortality following a colectomy for ulcerative colitis. Gastroenterology 134(3):680–687
Feldman M, Friedman LS, Brandt LJ (2016) Sleisenger and Fordtran’s gastrointestinal and liver disease, vol 2, 10th edn. Elsevier, Amsterdam, p 2616
Sonnenberg A, Genta RM (2015) Epithelial Dysplasia and Cancer in IBD Strictures. J Crohns Colitis. 9(9):769–775
Fumery M, Pineton de Chambrun G, Stefanescu C, Buisson A, Bressenot A, Beaugerie L, Amiot A, Altwegg R, Savoye G, Abitbol V, Bouguen G, Simon M, Duffas JP, Hébuterne X, Nancey S, Roblin X, Leteurtre E, Bommelaer G, Lefevre JH, Brunetti F, Guillon F, Bouhn P-BL (2015) Detection of dysplasia or cancer in 3.5% patients with inflammatory bowel disease and colonic strictures. Clin Gastroenterol Hepatol 13(10):1770–1775
Khan F, Shen B (2019) Inflammation and neoplasia of the pouch in inflammatory bowel disease. Curr Gastroenterol Rep 21(4):1–7
Bemelman WA, Warusavitarne J, Sampietro GM, Serclova Z, Zmora O, Luglio G et al (2018) ECCO-ESCP consensus on surgery for Crohn’s disease. J Crohn’s Colitis 12(1):1–16
East JE, Brooker JC, Rutter MD, Saunders BP (2007) A pilot study of intrastricture steroid versus placebo injection after balloon dilatation of crohn’s strictures. Clin Gastroenterol Hepatol 5(9):1065–1069
Loras C, Pérez-Roldan F, Gornals JB, Barrio J, Igea F, González-Huix F et al (2012) Endoscopic treatment with self-expanding metal stents for Crohn’s disease strictures. Aliment Pharmacol Ther 36(9):833–839
Acknowledgements
On the behalf of the ENEIDA registry of GETECCU (in addition to the authors): Julià Panés, MD PhD (Hospital Clínic de Barcelona, Barcelona), Míguel Mínguez, MD PhD (Hospital Clínico de Valencia, Valencia), Valle García Sánchez, MD PhD (Hospital Reina Sofía, Córdoba), Javier Pérez-Gisbert, MD PhD (Hospital La Princesa, Madrid), Fernando Gomollón, MD PhD (Hospital Lozano Blesa, Zaragoza), Marta Piqueras, MD (Consorci Sanitari de Terrassa, Barcelona), José Luís Cabriada, MD PhD (Hospital de Galdakao, Bizkaia), Montserrat Andreu, MD PhD (Hospital del Mar, Barcelona).
Author information
Authors and Affiliations
Consortia
Contributions
CL: design of the study, study coordination, acquisition of data, statistical analysis and interpretation of data, manuscript writing, critical revision of the manuscript, and approval of the final draft submitted. XA: study coordination, acquisition of data, statistical analysis and interpretation of data, manuscript writing, critical revision of the manuscript, and approval of the final draft submitted. ME: design of the study, study coordination, critical revision of the manuscript, and approval of the final draft submitted. FFB, YZ: statistical analysis and interpretation of data, critical revision of the manuscript, and approval of the final draft submitted. BG, MS, VS, MB, ED, MC, ER, BS, XC, JB, JG, EI, JPG, MJC, YB, DM, ALS, IR, LB, MA, LM, MDM: acquisition of data, approval of the final draft submitted.
Corresponding author
Ethics declarations
Disclosures
Xavier Andújar, Carme Loras, Begoña González, Milena Socarras, Vicente, Maia Boscà, Eugeni Domenech, Margalida Calafat, Esther Rodríguez, Beatriz Sicilia, Xavier Calvet, Jesús Barrio, Jordi Guardiola, Eva Iglesias, María José Casanova, Yolanda Ber, David Monfort, Antonio López-Sanromán, Iago Rodríguez, Luís Bujanda, Lucía Márquez, María Dolores Martín-Arranz, Yamile Zabana, Fernando Fernández-Bañares and María Esteve have no conflict of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The members of the ENEIDA registry of GETECCU are listed in the “Acknowledgement” section.
Rights and permissions
About this article
Cite this article
Andújar, X., Loras, C., González, B. et al. Efficacy and safety of endoscopic balloon dilation in inflammatory bowel disease: results of the large multicenter study of the ENEIDA registry. Surg Endosc 34, 1112–1122 (2020). https://doi.org/10.1007/s00464-019-06858-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-019-06858-z