Abstract
Background and aims
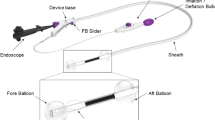
Colonic endoscopic submucosal dissection (ESD) is time-consuming and bears a high risk of perforation. The aim of the present study was to compare the safety and efficacy between novel articulating devices and conventional ESD in live porcine colon models.
Methods
Thirty ESDs in ten pigs were carried out at three different locations (15, 25, and 35 cm from the anus) by the conventional method (n = 15) and by the new method (n = 15). Procedure times, adverse events (perforation, bleeding), and damage to the muscular layer were recorded, and the ESD time per unit area of the specimens was calculated.
Results
The perforation rate using the conventional method was 6.7% (1/15), whereas that using the new method was 0.0%. The number of sites of muscular damage was significantly lower in the new than conventional method (6 vs. 37, respectively; P = 0.024). The mean procedure time was significantly shorter in the new than conventional method (4.6 ± 2.0 vs. 7.0 ± 4.1 min/cm2, respectively; P = 0.042).
Conclusions
Use of the new ESD method allows for reduced adverse events and a shortened resection time.







Similar content being viewed by others
Abbreviations
- ESD:
-
Endoscopic submucosal dissection
References
Saito Y, Uraoka T, Yamaguchi Y et al (2010) A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections. Gastrointest Endosc 72:1217–1725
Gotoda T, Kondo H, Ono H et al (1999) A new endoscopic mucosal resection procedure using an insulation-tipped electrosurgical knife for rectal flat lesions: report of two cases. Gastrointest Endosc 50:560–563
Maple JT, Abu Dayyeh BK, Chauhan SS et al (2015) Endoscopic submucosal dissection. Gastrointest Endosc 81:1311–1325
Bhatt A, Abe S, Kumaravel A et al (2015) Indications and techniques for endoscopic submucosal dissection. Am J Gastroenterol 110:784–791
Fujiya M, Tanaka K, Dokoshi T et al (2015) Efficacy and adverse events of EMR and endoscopic submucosal dissection for the treatment of colon neoplasms: a meta-analysis of studies comparing EMR and endoscopic submucosal dissection. Gastrointest Endosc 81:583–595
Hotta K, Oyama T, Shinohara T et al (2010) Learning curve for endoscopic submucosal dissection of large colorectal tumors. Dig Endosc 22:302–306
Patel N, Patel K, Ashrafian H et al (2016) Colorectal endoscopic submucosal dissection: a systematic review of mid-term clinical outcomes. Dig Endosc 28:405–416
Fukami N (2013) What we want for ESD is a second hand! Traction method. Gastrointest Endosc 78:274–276
Kume K (2014) Endoscopic therapy for early gastric cancer: standard techniques and recent advances in ESD. World J Gastroenterol 20:6425–6432
Ikeda K, Sumiyama K, Tajiri H et al (2011) Evaluation of a new multitasking platform for endoscopic full-thickness resection. Gastrointest Endosc 73:117–122
Diana M, Chung H, Liu KH et al (2013) Endoluminal surgical triangulation: overcoming challenges of colonic endoscopic submucosal dissections using a novel flexible endoscopic surgical platform: feasibility study in a porcine model. Surg Endosc 27:4130–4135
Chiu PW, Phee SJ, Bhandari P et al (2015) Enhancing proficiency in performing endoscopic submucosal dissection (ESD) by using a prototype robotic endoscope. Endosc Int Open 03:E439–E442
Zizer E, Roppenecker D, Helmes F et al (2016) A new 3D-printed overtube system for endoscopic submucosal dissection: first results of a randomized study in a poricine model. Endoscopy 48:762–765
Nakadate R, Kenmotsu H, Nakamura S et al (2014) 2.6 mm articulating endoscopic submucosal dissection device insertable into standard endoscope. Int J CARS 9(Suppl 1):S181–S182
Nakadate R, Nakamura S, Moriyama T et al (2015) Gastric endoscopic submucosal dissection using novel 2.6-mm articulating devices: an ex vivo comparative and in vivo feasibility study. Endoscopy 47:820–824
Acknowledgements
We thank Angela Morben, DVM, ELS, from Edanz Group (http://www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
YO: contributed to the study concept and design, the acquisition, analysis and interpretation of the data, the statistical analysis, and the initial drafting of the manuscript; RN: contributed to the study concept and design, the acquisition, the analysis and interpretation of the data, the drafting of the manuscript, and the critical revision of the manuscript for important intellectual content; SN, JA, SO, TM, ME, TI, KO, TA, TI: contributed to data collection and interpretation, and critically reviewed the manuscript; TK and MH: contributed to the critical revision of the manuscript for important intellectual content and approved the final draft for submission. All authors approved the final version of the manuscript, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Disclosures
Drs. Okamoto, Nakadate, Nakamura, Arata, Ohuchida, and Hashizume have received research funding from Hogy Medical Co. Ltd., Japan. Drs. Oguri, Moriyama, Esaki, Iwasa, Akahoshi, Ikeda and Kitazono have no conflicts of interest or financial ties to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Video 1 The first part of the video shows the dexterous movement, quick response, and small bending radius of the novel articulating devices. The second part of the video shows the endoscopic camera view during an in vivo porcine colorectal ESD. (MPG 45344 KB)
Rights and permissions
About this article
Cite this article
Okamoto, Y., Nakadate, R., Nakamura, S. et al. Colorectal endoscopic submucosal dissection using novel articulating devices: a comparative study in a live porcine model. Surg Endosc 33, 651–657 (2019). https://doi.org/10.1007/s00464-018-6408-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-018-6408-5