Abstract
Background
Minimally invasive surgery (MIS) may improve surgical recovery and reduce time to adjuvant systemic therapy after colon cancer resection. The objective of this study was to determine the effect of MIS on the initiation of adjuvant systemic therapy and survival in patients with stage III colon cancer.
Methods
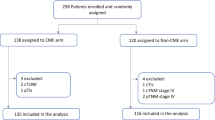
The 2010–2014 National Cancer Database was queried for patients with resected stage III colon adenocarcinoma, and divided into MIS, which included laparoscopic and robotic approaches, and open surgery. Propensity-score matching was used to balanced open and MIS groups. The main outcome measures were delayed initiation of adjuvant systemic therapy (defined as > 8 weeks after surgery) and 5-year overall survival (OS). Multiple Cox regression was performed to identify independent predictors for 5-year OS, including an interaction between delayed systemic therapy and MIS, and adjusted for clustering at the hospital level.
Results
There were 86,680 patients that were included in this study. Overall, 45% (38,713) underwent MIS colectomy, of which 93% underwent laparoscopic and 7% robotic surgery. After matching, 33,183 open patients were balanced to 33,183 MIS patients. Patient, tumor, and facility characteristics were similar in the matched cohort. More patients in the MIS group received adjuvant therapy within 8 weeks of surgery (49% vs. 42%, p < 0.001), and fewer MIS patients did not receive any systemic therapy (30% vs. 35%, p < 0.001). Delayed initiation of systemic therapy > 8 weeks was associated with worse 5-year OS (HR 1.27, 95%CI 1.19–1.36). MIS was independently associated with improved survival (HR 0.92, 95%CI 0.86–0.97). This relationship remained even if 90-day mortality was excluded.
Conclusions
MIS approaches are associated with less delay to the initiation of adjuvant systemic therapy and improved survival in patients with stage III colon adenocarcinoma. Surgeons should favor MIS approaches for the treatment of stage III colon adenocarcinoma whenever possible.



Similar content being viewed by others
References
Brenner H, Kloor M, Pox CP (2014) Colorectal cancer. Lancet 383:1490–1502
Boland GM, Chang GJ, Haynes AB, Chiang YJ, Chagpar R, Xing Y, Hu CY, Feig BW, You YN, Cormier JN (2013) Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer 119:1593–1601
Booth CM, Nanji S, Wei X, Peng Y, Biagi JJ, Hanna TP, Krzyzanowska MK, Mackillop WJ (2016) Use and effectiveness of adjuvant chemotherapy for stage III colon cancer: a population-based study. J Natl Compr Canc Netw 14:47–56
Biagi JJ, Raphael MJ, Mackillop WJ, Kong W, King WD, Booth CM (2011) Association between time to initiation of adjuvant chemotherapy and survival in colorectal cancer: a systematic review and meta-analysis. JAMA 305:2335–2342
Becerra AZ, Probst CP, Tejani MA, Aquina CT, Gonzalez MG, Hensley BJ, Noyes K, Monson JR, Fleming FJ (2015) Opportunity lost: adjuvant chemotherapy in patients with stage III colon cancer remains underused. Surgery 158:692–699
Lee L, Tran T, Mayo NE, Carli F, Feldman LS (2014) What does it really mean to “recover” from an operation? Surgery 155:211–216
Jin LX, Sanford DE, Squires MH, Moses LE, Yan Y, Poultsides GA, Votanopoulos KI, Weber SM, Bloomston M, Pawlik TM, Hawkins WG, Linehan DC, Schmidt C, Worhunsky DJ, Acher AW, Cardona K, Cho CS, Kooby DA, Levine EA, Winslow E, Saunders N, Spolverato G, Colditz GA, Maithel SK, Fields RC (2016) Interaction of postoperative morbidity and receipt of adjuvant therapy on long-term survival after resection for gastric adenocarcinoma: results from the U.S. Gastric Cancer Collaborative. Ann Surg Oncol 23:2398–2408
Schwenk W, Haase O, Neudecker J, Muller JM (2005) Short term benefits for laparoscopic colorectal resection. Cochrane Database Syst Rev 3::CD003145
Dowson HM, Cowie AS, Ballard K, Gage H, Rockall TA (2008) Systematic review of quality of life following laparoscopic and open colorectal surgery. Colorectal Dis 10:757–768
Jung YB, Kang J, Park EJ, Baik SH, Lee KY (2016) Time to initiation of adjuvant chemotherapy in colon cancer: comparison of open, laparoscopic, and robotic surgery. J Laparoendosc Adv Surg Tech A 26:799–805
Malietzis G, Mughal A, Currie AC, Anyamene N, Kennedy RH, Athanasiou T, Jenkins JT (2015) Factors implicated for delay of adjuvant chemotherapy in colorectal cancer: a meta-analysis of observational studies. Ann Surg Oncol 22:3793–3802
Bilimoria KY, Stewart AK, Winchester DP, Ko CY (2008) The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol 15:683–690
Deyo RA, Cherkin DC, Ciol MA (1992) Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol 45:613–619
Bos AC, van Erning FN, van Gestel YR, Creemers GJ, Punt CJ, van Oijen MG, Lemmens VE (2015) Timing of adjuvant chemotherapy and its relation to survival among patients with stage III colon cancer. Eur J Cancer 51:2553–2561
Sun Z, Adam MA, Kim J, Nussbaum DP, Benrashid E, Mantyh CR, Migaly J (2016) Determining the optimal timing for initiation of adjuvant chemotherapy after resection for stage II and III colon cancer. Dis Colon Rectum 59:87–93
Des Guetz G, Nicolas P, Perret GY, Morere JF, Uzzan B (2010) Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur J Cancer 46:1049–1055
In H, Bilimoria KY, Stewart AK, Wroblewski KE, Posner MC, Talamonti MS, Winchester DP (2014) Cancer recurrence: an important but missing variable in national cancer registries. Ann Surg Oncol 21:1520–1529
Dehejia RH, S W (1999) Causal effects in nonexperimental studies: reevaluating the evaluation of training programs. J Am Stat Assoc 94:1053–1062
Austin PC (2009) Some methods of propensity-score matching had superior performance to others: results of an empirical investigation and Monte Carlo simulations. Biom J Biom Z 51:171–184
Flury BK, Reidwyl H (1986) Standard distance in univariate and multivariate analysis. Am Stat 40:249–251
Austin PC (2009) Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun Stat Simul Comput 38:1228–1234
Linden A (2017) Improving causal inference with a doubly robust estimator that combines propensity score stratification and weighting. J Eval Clin Pract 23:697–702
Kim YW, Choi EH, Kim BR, Ko WA, Do YM, Kim IY (2017) The impact of delayed commencement of adjuvant chemotherapy (eight or more weeks) on survival in stage II and III colon cancer: a national population-based cohort study. Oncotarget 8:80061–80072
Becerra AZ, Aquina CT, Mohile SG, Tejani MA, Schymura MJ, Boscoe FP, Xu Z, Justiniano CF, Boodry CI, Swanger AA, Noyes K, Monson JR, Fleming FJ (2017) Variation in delayed time to adjuvant chemotherapy and disease-specific survival in stage III colon cancer patients. Ann Surg Oncol 24:1610–1617
Veenhof AA, Vlug MS, van der Pas MH, Sietses C, van der Peet DL, de Lange-de Klerk ES, Bonjer HJ, Bemelman WA, Cuesta MA (2012) Surgical stress response and postoperative immune function after laparoscopy or open surgery with fast track or standard perioperative care: a randomized trial. Ann Surg 255:216–221
Xu D, Li J, Song Y, Zhou J, Sun F, Wang J, Duan Y, Hu Y, Liu Y, Wang X, Sun L, Wu L, Ding K (2015) Laparoscopic surgery contributes more to nutritional and immunologic recovery than fast-track care in colorectal cancer. World J Surg Oncol 13::18
King PM, Blazeby JM, Ewings P, Kennedy RH (2008) Detailed evaluation of functional recovery following laparoscopic or open surgery for colorectal cancer within an enhanced recovery programme. Int J Colorectal Dis 23:795–800
Pecorelli N, Fiore JF Jr, Gillis C, Awasthi R, Mappin-Kasirer B, Niculiseanu P, Fried GM, Carli F, Feldman LS (2016) The six-minute walk test as a measure of postoperative recovery after colorectal resection: further examination of its measurement properties. Surg Endosc 30:2199–2206
Aloia TA, Zimmitti G, Conrad C, Gottumukalla V, Kopetz S, Vauthey JN (2014) Return to intended oncologic treatment (RIOT): a novel metric for evaluating the quality of oncosurgical therapy for malignancy. J Surg Oncol 110:107–114
Kim BJ, Caudle AS, Gottumukkala V, Aloia TA (2016) The impact of postoperative complications on a timely return to intended oncologic therapy (RIOT): the role of enhanced recovery in the cancer journey. Int Anesthesiol Clin 54:e33–e46
Cools-Lartigue J, Spicer J, McDonald B, Gowing S, Chow S, Giannias B, Bourdeau F, Kubes P, Ferri L (2013) Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J Clin Invest. https://doi.org/10.1172/JCI67484
Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH (2015) Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg 261:497–505
McSorley ST, Horgan PG, McMillan DC (2016) The impact of the type and severity of postoperative complications on long-term outcomes following surgery for colorectal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol 97:168–177
Di B, Li Y, Wei K, Xiao X, Shi J, Zhang Y, Yang X, Gao P, Zhang K, Yuan Y, Zhang D, Wei X, Liu S, Wang J, Wang X, Zhang Y, Cai H (2013) Laparoscopic versus open surgery for colon cancer: a meta-analysis of 5-year follow-up outcomes. Surg Oncol 22:e39–e43
Kuhry E, Schwenk WF, Gaupset R, Romild U, Bonjer HJ (2008) Long-term results of laparoscopic colorectal cancer resection. Cochrane Database Syst Rev 2::CD003432
Brooks JM, Ohsfeldt RL (2013) Squeezing the balloon: propensity scores and unmeasured covariate balance. Health Serv Res 48:1487–1507
Martel G, Crawford A, Barkun JS, Boushey RP, Ramsay CR, Fergusson DA (2012) Expert opinion on laparoscopic surgery for colorectal cancer parallels evidence from a cumulative meta-analysis of randomized controlled trials. PLoS ONE 7:e35292
Akmal Y, Bailey C, Baek JH, Metchikian M, Pigazzi A (2011) Oncological outcomes of laparoscopic colon resection for cancer after implementation of a full-time preceptorship. Surg Endosc 25:2967–2971
Park EJ, Kim MS, Kim G, Kim CH, Hur H, Min BS, Baik SH, Lee KY, Kim NK (2015) Long-term oncologic outcomes of laparoscopic right hemicolectomy during the learning curve period: comparative study with cases after the learning curve period. Surg Laparosc Endosc Percutan Tech 25:52–58
Panchal JM, Lairson DR, Chan W, Du XL (2016) Geographic variation in oxaliplatin chemotherapy and survival in patients with colon cancer. Am J Ther 23:e720–e729
Morris M, Platell C, Fritschi L, Iacopetta B (2007) Failure to complete adjuvant chemotherapy is associated with adverse survival in stage III colon cancer patients. Br J Cancer 96:701–707
Bailey MB, Davenport DL, Vargas HD, Evers BM, McKenzie SP (2014) Longer operative time: deterioration of clinical outcomes of laparoscopic colectomy versus open colectomy. Dis Colon Rectum 57:616–622
Curtis NJ, Taylor M, Fraser L, Salib E, Noble E, Hipkiss R, Allison AS, Dalton R, Ockrim JB, Francis NK (2017) Can the combination of laparoscopy and enhanced recovery improve long-term survival after elective colorectal cancer surgery? Int J Colorectal Dis 33(2):231–234
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Dr. Albert reports consultant’s fees from Applied Medical, Stryker, and Conmed, and stock options from Applied Medical. Drs. Lee, Wong-Chong Kelly, Nassif, and Monson have no conflicts of interest or financial ties to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, L., Wong-Chong, N., Kelly, J.J. et al. Minimally invasive surgery for stage III colon adenocarcinoma is associated with less delay to initiation of adjuvant systemic therapy and improved survival. Surg Endosc 33, 460–470 (2019). https://doi.org/10.1007/s00464-018-6319-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-018-6319-5