Abstract
Background
Minimally invasive colorectal resection (MICR) is associated with persistently elevated plasma VEGF levels that may stimulate angiogenesis in residual tumor foci. Placenta growth factor (PlGF) stimulates neovascularization in tumors by modulating VEGF’s effects. This study’s purpose was to determine the impact of MICR on blood PlGF levels in cancer patients (Study A) and to compare PreOp levels in patients with cancer and benign (BEN) disease (Study B).
Methods
Blood samples were collected preoperatively, on postoperative day (POD) 1, POD 3, and at various time points 2–4 weeks after surgery. Samples from 7-day periods after POD 6 were bundled to allow analysis. Plasma PlGF levels were determined via ELISA, results reported as mean ± SD, and data analyzed via t test. Significance was set at p < 0.008 after Bonferroni correction.
Results
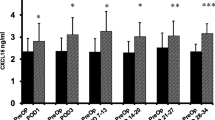
Study A: 76 colorectal cancer (CRC) patients had MICR (laparoscopic, 59%; hand-assisted, 41%). The mean length of stay was 5.8 ± 2.1 days. The mean PreOp PlGF level was 15.4 ± 4.3 pg/ml. Significantly increased levels were noted on POD 1 (25.8 ± 7.7 pg/ml, p < 0.001), POD 3 (22.9 ± 6.7, p < 0.001), POD 7–13 (19.2 ± 5.1, p < 0.001), and POD 14–20 (19.5 ± 6.7, p < 0.002). The mean POD 21–27 level was not significantly different from baseline. Study B included 126 CRC and 111 BEN patients. PreOp levels were higher in the CRC patients (15.6 ± 5.3 pg/ml) than in the BEN group (13.5 ± 5.5 pg/ml, p = 0.001).
Conclusions
PlGF levels are elevated for 3 weeks after MICR and PreOp plasma levels are higher in CRC patients than in BEN disease patients. The cause of the postoperative increase is unclear. The persistently higher blood levels of PlGF and VEGF after MICR may stimulate angiogenesis in residual tumor foci. Further studies regarding late blood protein alterations after surgery appear to be indicated.


Similar content being viewed by others
References
Kirman I, Cekic V, Poltaratskaia N, Asi Z, Bessler M, Huang EH, Forde KA, Whelan RL (2002) Plasma from patients undergoing major open surgery stimulates in vitro tumor growth: lower insulin-like growth factor binding protein 3 levels may, in part, account for this change. Surgery 132(2):186–192
Belizon A, Balik E, Horst P, Feingold D, Arnell T, Azarani T, Cekic V, Skitt R, Kumara S, Whelan RL (2008) Persistent elevation of plasma vascular endothelial growth factor levels during the first month after minimally invasive colorectal resection. Surg Endosc 22(2):287–297
Shantha Kumara HM, Feingold D, Kalady M, Dujovny N, Senagore A, Hyman N, Cekic V, Whelan RL (2009) Colorectal resection is associated with persistent proangiogenic plasma protein changes: postoperative plasma stimulates in vitro endothelial cell growth, migration, and invasion. Ann Surg 249(6):973–977
Wei SC, Tsao PN, Yu SC, Shun CT, Tsai-Wu JJ, Wu CH, Su YN, Hsieh FJ, Wong JM (2005) Placenta growth factor expression is correlated with survival of patients with colorectal cancer. Gut 54(5):666–672
Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M, Wu Y, Bono F, Devy L, Beck H, Scholz D, Acker T, DiPalma T, Dewerchin M, Noel A, Stalmans I, Barra A, Blacher S, Vandendriessche T, Ponten A, Eriksson U, Plate KH, Foidart JM, Schaper W, Charnock-Jones DS, Hicklin DJ, Herbert JM, Collen D, Persico MG (2001) Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 7(5):575–583
Fischer C, Jonckx B, Mazzone M, Zacchigna S, Loges S, Pattarini L, Chorianopoulos E, Liesenborghs L, Koch M, De Mol M, Autiero M, Wyns S, Plaisance S, Moons L, van Rooijen N, Giacca M, Stassen JM, Dewerchin M, Collen D, Carmeliet P (2007) Anti-PlGF inhibits growth of VEGF(R)-inhibitor-resistant tumors without affecting healthy vessels. Cell 131(3):463–475
Monsky WL, Fukumura D, Gohongi T, Ancukiewcz M, Weich HA, Torchilin VP, Yuan F, Jain RK (1999) Augmentation of transvascular transport of macromolecules and nanoparticles in tumors using vascular endothelial growth factor. Cancer Res 59(16):4129–4135
Park JE, Chen HH, Winer J, Houck KA, Ferrara N (1994) Placenta growth factor. Potentiation of vascular endothelial growth factor bioactivity, in vitro and in vivo, and high affinity binding to Flt-1 but not to Flk-1/KDR. J Biol Chem 269(41):25646–25654
Autiero M, Luttun A, Tjwa M, Carmeliet P (2003) Placental growth factor and its receptor, vascular endothelial growth factor receptor-1: novel targets for stimulation of ischemic tissue revascularization and inhibition of angiogenic and inflammatory disorders. J Thromb Haemost 1(7):1356–1370
Lee JC, Chow NH, Wang ST, Huang SM (2000) Prognostic value of vascular endothelial growth factor expression in colorectal cancer patients. Eur J Cancer 36(6):748–753
Wong MP, Cheung N, Yuen ST, Leung SY, Chung LP (1999) Vascular endothelial growth factor is up-regulated in the early pre-malignant stage of colorectal tumour progression. Int J Cancer 81(6):845–850
Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM (1995) Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res 55(18):3964–3968
Landriscina M, Cassano A, Ratto C, Longo R, Ippoliti M, Palazzotti B, Crucitti F, Barone C (1998) Quantitative analysis of basic fibroblast growth factor and vascular endothelial growth factor in human colorectal cancer. Br J Cancer 78(6):765–770
Adini A, Kornaga T, Firoozbakht F, Benjamin LE (2002) Placental growth factor is a survival factor for tumor endothelial cells and macrophages. Cancer Res 62(10):2749–2752
Takahashi A, Sasaki H, Kim SJ, Tobisu K, Kakizoe T, Tsukamoto T, Kumamoto Y, Sugimura T, Terada M (1994) Markedly increased amounts of messenger RNAs for vascular endothelial growth factor and placenta growth factor in renal cell carcinoma associated with angiogenesis. Cancer Res 54(15):4233–4237
Chen CN, Hsieh FJ, Cheng YM, Cheng WF, Su YN, Chang KJ, Lee PH (2004) The significance of placenta growth factor in angiogenesis and clinical outcome of human gastric cancer. Cancer Lett 213(1):73–82
Ho MC, Chen CN, Lee H, Hsieh FJ, Shun CT, Chang CL, Lai YT, Lee PH (2007) Placenta growth factor not vascular endothelial growth factor A or C can predict the early recurrence after radical resection of hepatocellular carcinoma. Cancer Lett 250(2):237–249
Matsumoto K, Suzuki K, Koike H, Okamura K, Tsuchiya K, Uchida T, Takezawa Y, Kobayashi M, Yamanaka H (2003) Prognostic significance of plasma placental growth factor levels in renal cell cancer: an association with clinical characteristics and vascular endothelial growth factor levels. Anticancer Res 23(6D):4953–4958
Parr C, Watkins G, Boulton M, Cai J, Jiang WG (2005) Placenta growth factor is over-expressed and has prognostic value in human breast cancer. Eur J Cancer 41(18):2819–2827
Kirman I, Jain S, Cekic V, Belizon A, Balik E, Sylla P, Arnell T, Forde KA, Whelan RL (2006) Altered plasma matrix metalloproteinase-9/tissue metalloproteinase-1 concentration during the early postoperative period in patients with colorectal cancer. Surg Endosc 20(3):482–486
Shantha Kumara HM, Hoffman A, Luchtefeld M, Kalady M, Hyman N, Feingold D, Baxter R, Whelan RL (2009) Minimally invasive colon resection is associated with a transient increase in plasma sVEGFR1 levels and a decrease in sVEGFR2 levels during the early postoperative period. Surg Endosc 23(4):694–699
Karayiannakis AJ, Makri GG, Mantzioka A, Karousos D, Karatzas G (1997) Systemic stress response after laparoscopic or open cholecystectomy: a randomized trial. Br J Surg 84(4):467–471
Schietroma M, Carlei F, Mownah A, Franchi L, Mazzotta C, Sozio A, Amicucci G (2004) Changes in the blood coagulation, fibrinolysis, and cytokine profile during laparoscopic and open cholecystectomy. Surg Endosc 18(7):1090–1096
Ordemann J, Jacobi CA, Schwenk W, Stosslein R, Muller JM (2001) Cellular and humoral inflammatory response after laparoscopic and conventional colorectal resections. Surg Endosc 15:600–608
Failla CM, Odorisio T, Cianfarani F, Schietroma C, Puddu P, Zambruno G (2000) Placenta growth factor is induced in human keratinocytes during wound healing. J Invest Dermatol 115(3):388–395
George DJ, Halabi S, Shepard TF, Vogelzang NJ, Hayes DF, Small EJ, Kantoff PW (2001) Prognostic significance of plasma vascular endothelial growth factor levels in patients with hormone-refractory prostate cancer treated on Cancer and Leukemia Group B 9480. Clin Cancer Res 7(7):1932–1936
Gorski DH, Leal AD, Goydos JS (2003) Differential expression of vascular endothelial growth factor-A isoforms at different stages of melanoma progression. J Am Coll Surg 197(3):408–418
Maeda K, Kang SM, Onoda N, Ogawa M, Kato Y, Sawada T, Chung KH (1999) Vascular endothelial growth factor expression in preoperative biopsy specimens correlates with disease recurrence in patients with early gastric carcinoma. Cancer 86(4):566–571
Escudero-Esparza A, Martin TA, Davies ML, Jiang WG (2009) PGF isoforms, PLGF1 and PGF2, in colorectal cancer and the prognostic significance. Cancer Genomics Proteonomics 6(4):239–246
Wei SC, Liang JT, Tsao PN, Hsieh FJ, Yu SC, Wong JM (2009) Preoperative serum placental growth factor level is a prognostic biomarker in colorectal cancer. Dis Colon Rectum 52(9):1630–1636
Ribatti D (2008) The discovery of the placental growth factor and its role in angiogenesis: a historical review. Angiogenesis 11(3):215–221
Matsumoto T, Claesson-Welsh L (2001) VEGF receptor signal transduction. Sci STKE (112):RE21
Disclosures
H. M. C. Shantha Kumara, Jenny C. Cabot, Xiaohong Yan, Sonali A. C. Herath, Martin Luchtefeld, Matthew F. Kalady, Daniel L. Feingold, Raymond Baxter, and Richard Whelan have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shantha Kumara, H.M.C., Cabot, J.C., Yan, X. et al. Minimally invasive colon resection is associated with a persistent increase in plasma PlGF levels following cancer resection. Surg Endosc 25, 2153–2158 (2011). https://doi.org/10.1007/s00464-010-1514-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-010-1514-z