Abstract
Introduction
Bariatric surgery has evolved into multiple forms in the last decades, combining food restriction and malabsorption. The aim of this study was to develop a new technique based on food restriction and early stimulation of the distal gut, thus maintaining the alimentary tract continuity.
Methods
Thirty-two Yorkshire pigs, weight 22.2 ± 5.4 kg (mean ± SD) were randomly assigned to four laparoscopic procedures: ileal transposition (IT, n = 8); sleeve gastrectomy with ileal transposition (SGIT, n = 8); Roux-en-Y gastric bypass (GBP, n = 8); sham operation (SHAM, n = 8). Firing 45-mm linear staplers over a 60-F bougie, resecting the greater curvature and fundus, constituted a sleeve gastrectomy. Ileal transposition was performed by isolating a 100-cm ileal segment proximal to the ileocecal valve and by dividing the proximal jejunum 15 cm distal to the ligament of Treitz and performing re-anastomosis. Gastric bypass consisted of creating a proximal gastric pouch and a 300 cm alimentary limb. Sham operation was performed by bowel transections and re-anastomosis in the ileum and proximal jejunum together with gastrotomy and closure. Animals were evaluated weekly for weight increase and food intake. We performed a logistic regression analysis to compare weight progression curves, and analysis of variance (ANOVA) and Bonferroni (Dunn) tests to detect differences in weight and food intake.
Results
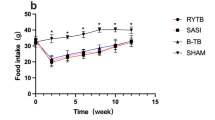
We observed significant differences in mean weight after 18 weeks between SGIT (30.9 ± 13.4 kg) and SHAM (72.5 ± 10.7 kg) (p = 0.0002), and GBP (28.6 ± 2.5 kg) and SHAM (p = 0.0001), and IT (56.1 ± 13.4 kg) and SHAM (p = 0.0081). No differences were observed between RYGB and SGIT. We also observed significant differences in food intake (grams per day) in the third month between SGIT (1668 ± 677 g) versus SHAM (3252 ± 476 g) (p = 0.0006), and GBP (2011 ± 565 g) versus SHAM (p = 0.039). No differences were observed in food intake between SGIT and GBP.
Conclusion
SGIT proved to be as effective in the short term as GBP on weight progression with no bypass of the proximal gut.



Similar content being viewed by others
References
Mun EC, Blackburn GL, Matthews JB (2001) Current status of medical and surgical therapy for obesity. Gastroenterology 120(3):669–81
Naslund E, Hellstrom PM, Kral JG (2001) The gut and food intake: an update for surgeons. J Gastrointest Surg 5(5):556–67
Wynne K, Stanley S, Bloom S (2004) The gut and regulation of body weight. J Clin Endocrinol Metab 89(6):2576–82
Hung CC, Pirie F, Luan J, et al. (2004) Studies of the peptide YY and neuropeptide Y2 receptor genes in relation to human obesity and obesity-related traits. Diabetes 53(9):2461–6
Canbeyli RS, Koopmans HS (1984) The role of jejunal signals in the long-term regulation of food intake in the rat. Physiol Behav 33(6):945–50
Wren AM, Small CJ, Abbott CR, et al. (2001) Ghrelin causes hyperphagia and obesity in rats. Diabetes 50(11):2540–7
Geloneze B, Tambascia MA, Pilla VF, et al. (2003) Ghrelin: a gut-brain hormone: effect of gastric bypass surgery. Obes Surg 13(1):17–22
Fruhbeck G, Diez-Caballero A, Gil MJ, et al. (2004) The decrease in plasma ghrelin concentrations following bariatric surgery depends on the functional integrity of the fundus. Obes Surg 14(5):606–12
Small CJ, Bloom SR (2004) Gut hormones and the control of appetite. Trends Endocrinol Metab 15(6):259–63
Dakin CL, Small CJ, Batterham RL, et al. (2004) Peripheral oxyntomodulin reduces food intake and body weight gain in rats. Endocrinology 145(6):2687–95
Verdich C, Flint A, Gutzwiller JP, et al. (2001) A meta-analysis of the effect of glucagon-like peptide-1 (7–36) amide on ad libitum energy intake in humans. J Clin Endocrinol Metab 86(9):4382–9
Cohen MA, Ellis SM, Le Roux CW, et al. (2003) Oxyntomodulin suppresses appetite and reduces food intake in humans. J Clin Endocrinol Metab 88(10):4696–701
Konturek SJ, Konturek JW, Pawlik T, Brzozowski T (2004) Brain-gut axis and its role in the control of food intake. J Physiol Pharmacol 55(1Pt2):137–54
Batterham RL, Cohen MA, Ellis SM, et al. (2003) Inhibition of food intake in obese subjects by peptide YY3-36. N Engl J Med 349(10):941–8
Korner J, Bessler M, Cirilo L, Conwell I (2004) Effects of Roux-en-Y gastric bypass on fasting and postprandial concentrations of plasma ghrelin, PYY and insulin. J Clin Endocrinol Metab Online
Rubino F, Gagner M, Gentileschi P, et al. (2004) The early effect of the Roux-en-Y gastric bypass on hormones involved in body weight regulation and glucose metabolism. Ann Surg 240(2):236–42
Stoeckli R, Chanda R, Langer I, Keller U (2004) Changes of body weight and plasma ghrelin levels after gastric banding and gastric bypass. Obes Res 12(2):346–50
Adami GF, Cordera R, Marinari G, et al. (2003) Plasma ghrelin concentratin in the short-term following biliopancreatic diversion. Obes Surg 13(6):889–92
Frezza EE (2004) Are we closer to finding the treatment for type 2 diabetes mellitus in morbid obesity? Are the incretins the key to success? Obes Surg 14(7):999–1005
Pories WJ (2004) Diabetes: the evolution of a new paradigm. Ann Surg 239(1):12–3
Mason EE (1999) Ileal [correction of ilial] transposition and enteroglucagon/GLP-1 in obesity (and diabetic?) surgery. Obes Surg 9(3):223–8
Patriti A, Facchiano E, Sanna A, et al. (2004) The enteroinsular axis and the recovery from type 2 diabetes after bariatric surgery. Obes Surg 14(6):840–8
Rubino F, Marescaux J (2004) Effect of duodenal-jejunal exclusion in a non-obese animal model of type 2 diabetes: a new perspective for an old disease. Ann Surg 239(1):1–11
Naslund E, Backman L, Holst JJ, et al. (1998) Importance of small bowel peptides for the improved glucose metabolism 20 years after jejunoileal bypass for obesity. Obes Surg 8(3):253–60
Regan JP, Inabnet WB, Gagner M, Pomp A (2003) Early experience with two-stage laparoscopic Roux-en-Y gastric bypass as an alternative in the super-super obese patient. Obes Surg 13(6):861–4
Koopmans HS, Sclafani A, Fichtner C, Aravich PF (1982) The effects of ileal transposition on food intake and body weight loss in VMH-obese rats. Am J Clin Nutr 35(2):284–93
Koopmans HS, Ferri GL, Sarson DL, et al. (1984) The effects of ileal transposition and jejunoileal bypass on food intake and GI hormone levels in rats. Physiol Behav 33(4):601–9
Strader AD, Vahl TP, Jandacek RJ, et al. (2005) Weight loss through ileal transposition is accompanied by increased ileal hormone secretion and synthesis in the rat. Am J Physiol Endocrinol Metab 288(2):E447–53
Ferri GL, Koopmans HS, Ghatei MA, et al. (1983) Ileal enteroglucagon cells after ileal-duodenal transposition in the rat. Digestion 26(1):10–6
De Paula AL, Macedo AL, Prudente AS, et al. (2006) Laparoscopic sleeve gastrectomy with ileal interposition (“neuroendocrine brake”)–pilot study of a new operation. Surg Obes Relat Dis 2(4):464–7
Langer FB, Reza Hoda MA, Bohdjalian A, et al. (2005) Sleeve gastrectomy and gastric banding: effects on plasma ghrelin levels. Obes Surg 15(7):1024–9
Melissas J, Koukouraki S, Askoxylakis J, et al. (2007) Sleeve gastrectomy: a restrictive procedure? Obes Surg 17(1):57–62
Gagner M (2005) La transposition iléale avec ou sans gastrectomie par laparoscopie chez l’homme (TIG). J Coeliochirurgie 54:4–9
Acknowledgments
The study was supported in part by funds from Ethicon Endosurgery, Cincinnati, OHIO. Dr Boza’s fellowship was funded by the Division of Surgery of Catholic University School of Medicine, Pontificia Universidad Católica de Chile, and the Scholarship Presidente de la República
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boza, C., Gagner, M., Devaud, N. et al. Laparoscopic sleeve gastrectomy with ileal transposition (SGIT): A new surgical procedure as effective as gastric bypass for weight control in a porcine model. Surg Endosc 22, 1029–1034 (2008). https://doi.org/10.1007/s00464-007-9685-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-007-9685-y