Abstract
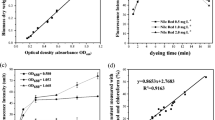
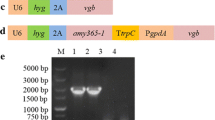
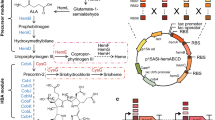
To block and weaken the bacterial branched VB12 synthetic metabolic pathway, homologous recombination technology was used to knock out the sirohaem synthase gene cysG located in the chromosome and the endogenous A plasmid of the Ensifer adhaerens Casida A strain, and the expression of the uroporphyrinogen III decarboxylase gene hemE was weakened by weak promoter substitution. The growth of the engineered strains and the production of VB12 and haem were analysed and measured in the engineered strains, aiming to provide a new strategy for enhancement of VB12 biosynthesis. The results showed that the chromosomal cysG gene knockout strain ΔcysG, endogenous A plasmid cysG gene knockout strain ΔpAcysG and cysG gene double knockout strain ΔcysGΔpAcysG grew normally, with VB12 yield increases of 19.9%, 11.2%, and 27.4% compared to the starting strain, respectively. In the background of the cysG gene knockout strain, the expression of the hemE gene was weakened, resulting in the generation of the strain ΔcysGΔpAcysG-E-pdnaD, and the VB12 yield of ΔcysGΔpA cysG-E-pdnaD reached 114.17 ± 5.77 mg L−1, an increase of 45.1% compared to the yield of the original strain. The above results indicate that the strategy of increasing VB12 production by knocking out the haem synthesis pathway and weakening the haem synthesis pathway is effective.




Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Lawrence AD, Evelyne D, Mclean KJ et al (2008) Identification, characterization, and structure/function analysis of a corrin reductase involved in adenosylcobalamin biosynthesis. J Biol Chem 283(16):10813–10821. https://doi.org/10.1074/jbc.M710431200
Ravnum S, Dan IA (2001) An adenosyl-cobalamin (coenzyme-B12)-repressed translational enhancer in the cob mRNA of Salmonella typhimurium. Mol Microbiol 39(6):1585–1594. https://doi.org/10.1046/j.1365-2958.2001.02346.x
Sally P, Stabler M (2014) Vitamin B12 deficiency. N Engl J Med 368:149–160. https://doi.org/10.1056/NEJMcp1113996
Andrès E (2014) Oral cobalamin (vitamin B12) therapy in pernicious anemia. Autoimmun Rev 13(7):778. https://doi.org/10.1016/j.autrev.2014.01.057
Urits I, Yilmaz M, Bahrun E et al (2020) Utilization of B12 for the treatment of chronic migraine. Baillière’s Best Pract Res Clin Anaesthesiol 34(3):479–491. https://doi.org/10.1016/j.bpa.2020.07.009
Julian T, Syeed R, Glascow N, Angelopoulou E, Zis P (2020) B12 as a treatment for peripheral neuropathic pain: a systematic review. Nutrients 12(8):2221. https://doi.org/10.3390/nu12082221
Rakuša ŽT, Roškar R, Hickey N, Geremia S (2022) Vitamin B12 in foods, food supplements, and medicines—a review of its role and properties with a focus on its stability. Molecules 28(1):240. https://doi.org/10.3390/molecules28010240
European Food Safety et al (2008) 5′-Deoxyadenosylcobalamin and methylcobalamin as sources for vitamin B12 added as a nutritional substance in food supplements—scientific opinion of the Scientific Panel on Food Additives and Nutrient Sources added to food. Efsa J 815:1–21. https://doi.org/10.2903/j.efsa.2008.815
Guido R, Gabriele A, Giovanna A et al (2018) Safety and efficacy of vitamin B12 (in the form of cyanocobalamin) produced by Ensifer spp. as a feed additive for all animal species based on a dossier submitted by VITAC EEIG. EFSA J 16(7):5336. https://doi.org/10.2903/j.efsa.2018.5336
Hodgkin DC, Kamper J, Mackay M et al (1956) Structure of vitamin B12. Nature 178(4524):64–66. https://doi.org/10.1038/178064a0
Woodward RB (1973) The total synthesis of vitamin B12. Pure Appl Chem 33:145–177. https://doi.org/10.1351/pac197333010145
Xia W, Chen W, Peng WF et al (2015) Industrial vitamin B-12 production by Pseudomonas denitrificans using maltose syrup and corn steep liquor as the cost-effective fermentation substrates. Bioprocess Biosyst Eng 38(6):1065–1073. https://doi.org/10.1007/s00449-014-1348-5
Wang P, Zhang ZW, Jiao YJ et al (2015) Improved propionic acid and 5,6-dimethylbenzimidazole control strategy for vitamin B12 fermentation by Propionibacterium freudenreichii. J Biotechnol 193(6):123–129. https://doi.org/10.1016/j.jbiotec.2014.11.019
Blanche F, Cameron B, Crouzet J, Debussche L, Levy-Schil S, Thibaut D (1998) Rhône-Poulenc Biochimie. Eur Patent B1:0516647
Hörig J, Renz P (1977) Biosynthesis of vitamin B12. formation of free 5,6-dimethylbenzimidazole and alpha-ribazole from riboflavin by Propionibacterium freudenreichii. FEBS Lett 80(2):337–339. https://doi.org/10.1016/0014-5793(77)80470-5
Salminen S et al (1998) Demonstration of safety of probiotics—a review. Int J Food Microbiol 44(1):93–106. https://doi.org/10.1016/s0168-1605(98)00128-7
Fang H, Kang J, Zhang D (2017) Microbial production of vitamin B12: a review and future perspectives. Microb Cell Fact 16(1):1–15. https://doi.org/10.1186/s12934-017-0631-y
EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP) (2015) Scientific Opinion on safety and efficacy of vitamin B12 (cyanocobalamin) produced by Ensifer adhaerens when used as a feed additive for all animal species based on a dossier submitted by Lohmann Animal Health. EFSA J 13(5):4112–4130. https://doi.org/10.2903/j.efsa.2015.4112
Choby JE, Skaar EP (2016) Heme synthesis and acquisition in bacterial pathogens. J Mol Biol 428(17):3408–3428. https://doi.org/10.1016/j.jmb.2016.03.018
Anzaldi LL, Skaar EP (2010) Overcoming the heme paradox: heme toxicity and tolerance in bacterial pathogens. Infect Immun 78(12):4977–4989. https://doi.org/10.1128/IAI.00613-10
Kakhki MP, Heidary M (2014) Trizol-based RNA extraction: a reliable method for gene expression studies. Univ Tehran 25(1):13–17
Fenghuan W (2012) Effects of budC gene knockout and ldhA overexpression on D-lactic acid production by Klebsiella pneumoniae. J Beijing Univ Chem Technol Nat Sci Ed 39(4):84–89. https://doi.org/10.1007/s11783-011-0280-z
Liu T, Liu B, Zhou H, Zhang J (2021) Knockout of the das gene increases S-adenosylmethionine production in Komagataella phaffii. Biotechnol Biotechnol Equip 35(1):29–36. https://doi.org/10.1080/13102818.2020.1837012
Crook N, Sun J, Morse N, Schmitz A, Alper HS (2016) Identification of gene knockdown targets conferring enhanced isobutanol and 1-butanol tolerance to Saccharomyces cerevisiae using a tunable RNAi screening approach. Appl Microbiol Biotechnol 100(23):1–14
Yawei C, Shuangyan L, Lihai F, Xu Z, Tianwei T (2015) Control of ATP concentration in Escherichia coli using synthetic small regulatory RNAs for enhanced S-adenosylmethionine production. FEMS Microbiol Lett 362(15):1–7. https://doi.org/10.1093/femsle/fnv115
Dailey H, Dailey T, Gerdes S et al (2017) Prokaryotic heme biosynthesis: multiple pathways to a common essential product. Microbiol Mol Biol Rev 81(1):48–56. https://doi.org/10.1128/mmbr.00048-16
Evelyne R, Helen KL, Richard B et al (2003) Identification and functional analysis of enzymes required for precorrin-2 dehydrogenation and metal ion insertion in the biosynthesis of sirohaem and cobalamin in Bacillus megaterium. Biochemistry 370(2):505–516. https://doi.org/10.1042/BJ20021443
Cui Y, Dong H, Tong B et al (2022) A versatile Cas12k-based genetic engineering toolkit (C12KGET) for metabolic engineering in genetic manipulation-deprived strains. Nucleic Acids Res 50(15):8961–8973. https://doi.org/10.1093/NAR/GKAC655
Huang Y, Xu X, Song Y et al (2019) Structure and product relationship analysis of acyl homoserine lactone synthases among Ensifer adhaerens reveals distinct chromosome and plasmid origins. Biochem Biophys Res Commun 516(3):907–913. https://doi.org/10.1016/j.bbrc.2019.06.131
Lee MJ, Kim HJ, Lee JY et al (2013) Effect of gene amplifications in porphyrin pathway on heme biosynthesis in a recombinant Escherichia coli. J Microbiol Biotechnol 23(5):668–673. https://doi.org/10.4014/jmb.1302.02022
Kang Z, Zhang J, Zhou J et al (2012) Recent advances in microbial production of delta-aminolevulinic acid and vitamin B12. Biotechnol Adv 30(6):1533–1542. https://doi.org/10.1016/j.biotechadv.2012.04.003
Acknowledgements
The authors thank AJE for providing polishing services for this article.
Funding
This work was supported by the Key Research and Development Program of Ningxia 2021BEG02003.
Author information
Authors and Affiliations
Contributions
YHL, YYC and JYS contributed to the conception and design of the researches and the interpretation of the data. YHL and YYC performed the experiments. YHL, YYC and QW performed some strains construction and VB12 detection. CLM and WH performed strains cultivation. YHL, QW and WH contributed to manuscript writing and figures making. All authors critically viewed, edited and approved the manuscript. All authors read and approved the final manuscript. YHL and YYC contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Chang, Y., Wang, Q. et al. Effect of blocking the haem synthesis pathway and weakening the haem synthesis pathway for sirohaem on the growth of and vitamin B12 synthesis in Ensifer adhaerens Casida A. Bioprocess Biosyst Eng 46, 1825–1835 (2023). https://doi.org/10.1007/s00449-023-02939-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-023-02939-5