Abstract
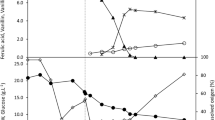
Biovanillin production by a wild strain of Bacillus cereus NCIM-5727 is studied using eugenol as the precursor aiming to achieve maximum vanillin productivity. Based on shake flask optimization, molar yield and global volumetric productivity of vanillin reached up to 71.2% (6.6 gL−1) and 0.18 g(Lh)−1, respectively, at 36 h by resting cells of B. cereus NCIM-5727 at the optimum cell concentration of 3 gL−1 using eugenol concentration of 10 gL−1 at 37 ºC, buffer pH 7.0, buffer volume 10%, and shaking speed 180 rpm. Furthermore, small-scale optimization in a bioreactor at the controlled aeration rate of 0.5 Lmin−1, agitation rate of 210 rpm, and pH 7.0 enhanced the global volumetric productivity of vanillin up to 0.28 g(Lh)−1 at 25 h of bioconversion. The highest vanillin molar yield (75.2%) is reported using resting cells of B. cereus NCIM-5727 upon eugenol biotransformation and found stable for 10 h.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Code availability
Not applicable.
References
Chen P, Yan L, Wu Z, Li S, Bai Z, Yan X, Wang N, Liang N, Li H (2016) A microbial transformation using Bacillus subtilis B7-S to produce natural vanillin from ferulic acid. Sci Rep 6:20400. https://doi.org/10.1038/srep20400
Rao RS, Ravishankar GA (2000) Review: Vanilla flavour: production by conventional and biotechnological routes. J Sci Food Agric 80:289–304. https://doi.org/10.1002/1097-0010(200002)
Ding P, Garrett M, Oystein L, Nienow AW, Pacek AW (2012) Generation of Hydrogen Gas during the Catalytic Oxidation of Sodium Lignosulfonate to Vanillin: initial results. Ind Eng Chem Res 51:184–188. https://doi.org/10.1021/ie201607t
Xu P, Hua D, Ma C (2007) Microbial transformation of propenylbenzenes for natural flavour production. Trends Biotechnol 25:571–576. https://doi.org/10.1016/j.tibtech.2007.08.011
Krings U, Berger RG (1998) Biotechnological production of flavours and fragrances. Appl Microbiol Biotechnol 49:1–8. https://doi.org/10.1007/s002530051129
Shimoni E, Ravid U, Shoham Y (2000) Isolation of a Bacillus sp. capable of transforming isoeugenol to vanillin. J Biotechnol 78:1–9. https://doi.org/10.1016/s0168-1656(99)00199-6
Hua D, Ma C, Lin S, Song L, Deng Z, Maomy Z, Zhang Z, Yu B, Xu P (2007) Biotransformation of isoeugenol to vanillin by a newly isolated Bacillus pumilus strain: identification of major metabolites. J Biotechnol 130:463–470. https://doi.org/10.1016/j.jbiotec.2007.05.003
Yamada M, Okada Y, Yoshida T, Nagasawa T (2007) Biotransformation of isoeugenol to vanillin by Pseudomonas putida IE27 cells. Appl Microbiol Biotechnol 73:1025–1030. https://doi.org/10.1007/s00253-006-0569-1
Zamzuri NA, Abd-Aziz S (2013) Biovanillin from agro wastes as an alternative food flavour. J Sci Food Agric 93:429–438. https://doi.org/10.1002/jsfa.5962
Gallage NJ, Moller BL (2015) Vanillin-bioconversion and bioengineering of the most popular plant flavor and its de novo biosynthesis in the vanilla orchid. Mol Plant 8:40–57. https://doi.org/10.1016/j.molp.2014.11.008
Paz A, Outeirino D, de Souza P, Oliveira R, Dominguez JM (2018) Fed-batch production of vanillin by Bacillus aryabhattai BA03. New Biotechnol 40:186–191. https://doi.org/10.1016/j.nbt.2017.07.012
Tadasa K (1977) Degradation of eugenol by a microorganism. Argic Biol Chem 41:925–929. https://doi.org/10.1080/00021369.1977.10862621
Rabenhorst J (1996) Production of methoxyphenol type natural aroma chemicals by biotransformation of eugenol with a new Pseudomonas sp. Appl Microbiol Biotechnol 46:470–474. https://doi.org/10.1007/s002530050846
Overhage J, Steinbuchel A, Priefert H (2006) Harnessing eugenol as a substrate for production of aromatic compounds with recombinant strains of Amycolatopsissp HR167. J Biotechnol 125:369–376. https://doi.org/10.1016/j.jbiotec.2006.03.024
Unno T, Kim SJ, Kanaly RA, Ahn JH, Kang SI, Hur HG (2007) Metabolic characterization of newly isolated Pseudomonas nitroreducensJin1 growing on eugenol and isoeugenol. J Agric Food Chem 55:8556–8561. https://doi.org/10.1021/jf0715929
Kadakol JC, Kamanavalli CM (2010) Biodegradation of eugenol by Bacillus cereus strain PN24. E-J Chem 7:74–80. https://doi.org/10.1155/2010/364637
Ashengroph M, Nahvi I, Zarkesh-Esfahani H, Momenbeik F (2011) Pseudomonas resinovorans SPR1, a newly isolated strain with potential of transforming eugenol to vanillin and vanillic acid. New Biotechnol 28:656–664. https://doi.org/10.1016/j.nbt.2011.06.009
Giedraityte G, Kalediene L (2014) Biotransformation of eugenol via protocatechuic acid by thermophilic Geobacillus sp. AY 946034 strain. J Microbiol Biotechnol 24:475–482. https://doi.org/10.4014/jmb.1309.09069
Singh A, Mukhopadhyay K, Ghosh Sachan S (2019) Biotransformation of eugenol to vanillin by a novel strain Bacillus safensis SMS1003. Biocatal Biotransfor 37:291–303. https://doi.org/10.1080/10242422.2018.1544245
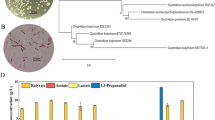
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Kamble AL, Meena VS, Banerjee UC (2010) Effect of agitation and aeration on the production of nitrile hydratase by Rhodococcuserythropolis MTCC 1526 in a stirred tank reactor. Lett Appl Microbiol 51:413–420. https://doi.org/10.1111/j.1472-765X.2010.02909.x
Barghini P, Di Gioia D, Fava F, Ruzzi M (2007) Vanillin production using metabolically engineered Escherichia coli under non-growing conditions. Microb Cell Fact 6:13. https://doi.org/10.1186/1475-2859-6-13
Di Gioia D, Luziatelli F, Negroni A, Ficca AG, Fava F, Ruzzi M (2011) Metabolic engineering of Pseudomonas fluorescens for the production of vanillin from ferulic acid. J Biotechnol 156:309–316. https://doi.org/10.1016/j.jbiotec.2011.08.014
Narbad A, Gasson MJ (1998) Metabolism of ferulic acid via vanillin using a novel CoA-dependent pathway in a newly-isolated strain of Pseudomonas fluorescens. Microbiol 144:1397–1405. https://doi.org/10.1099/00221287-144-5-1397
Ghosh S, Sachan A, Sen SK, Mitra A (2007) Microbial transformation of ferulic acid to vanillic acid by Streptomyces sannanensis MTCC 6637. J Ind Microbiol Biotechnol 34:131–138. https://doi.org/10.1007/s10295-006-0177-1
Song SH, Yeom SH, Choi SS, Yoo YJ (2002) Effect of aeration on denitrification by Ochrobactrum anthropic SY509. Biotechnol Bioprocess Eng 7:352–356. https://doi.org/10.1007/BF02933520
Tilay A, Bule M, Annapure U (2010) Production of biovanillin by one-step biotransformation using fungus Pycnoporous cinnabarinus. J Agric Food Chem 58:4401–4405. https://doi.org/10.1021/jf904141u
Ashengroph M, Nahvi I, Zarkesh-Esfahani H, Momenbeik F (2010) Optimization of media composition for improving conversion of isoeugenol into vanillin with Pseudomonas sp. strain KOB10 using the Taguchi method. Biocatal Biotransfor 28:339–347. https://doi.org/10.3109/10242422.2010.530660
Rivas Torres B, Aliakbarian N, Torre P, Perego P, Domínguez JM, Zilli M, Converti A (2009) Vanillin bioproduction from alkaline hydrolyzate of corn cob by Escherichia coli JM109/pBB1. Enzyme Microb Tech 44:154–158. https://doi.org/10.1016/j.enzmictec.2008.10.003
Sindhwani G, Uk I, Aeri V (2012) Microbial transformation of eugenol to vanillin. J Microbiol Biotechnol Res 2:313–318
Acknowledgements
This work was supported by the Ministry of Science and Technology of India through its funding agency DST SERB (Department of Science and Technology-Science and Engineering Research Board) under Grant No. SB/YS/LS-308/2013 in the form of research projects. The authors highly acknowledge Birla Institute of Technology for the infrastructure facilities. The authors express their gratitude to Dr. VK Nigam for assistance in operating the CSTR.
Funding
This work was supported by the Ministry of Science and Technology of India through its funding agency DST SERB (Department of Science and Technology-Science and Engineering Research Board) under Grant No. SB/YS/LS-308/2013 in the form of research projects.
Author information
Authors and Affiliations
Contributions
Each author has contributed significantly to this work. AS conducted experiments, literature search, prepared the tables or figures, and drafted the manuscript. SGS and KM contributed to the conception and design of the manuscript, and reviewed and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
Not applicable.
Consent to participate
The authors declare that they consent to participate.
Consent to publish
The authors declare that they consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, A., Mukhopadhyay, K. & Ghosh Sachan, S. Enhanced vanillin production from eugenol by Bacillus cereus NCIM-5727. Bioprocess Biosyst Eng 45, 1811–1824 (2022). https://doi.org/10.1007/s00449-022-02787-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-022-02787-9