Abstract
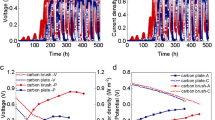
External resistance is important for the anode and cell performance. However, little attentions were paid on the effect of external resistance on the variation of biofilm structure. Here, we used external resistance ranged from 4000 to 500 Ω for anodic acclimation to investigate the correlation between anode performance and biofilm structure. With the reduce of external resistance, the maximum current density of anode increased from 1.0 to 3.4 A/m2, which was resulted from a comprehensive effect of reduced charge transfer resistance and increased diffusion resistance. Biological analysis showed that with the reduce of external resistance, biomass and extracellular polymeric substances content increased by 109 and 286%, cell viability increased by 22.7%, which contributed to the reduced charge transfer resistance. But the porosity of anodic biofilm decreased by 27.8%, which led to an increased diffusion resistance of H+. This work provided a clear correlation between the electrochemical performance and biofilm structure.






Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Topcu Ş, Taşkan E (2021) Effect of the tetracycline antibiotics on performance and microbial community of microbial fuel cell. Bioproc Biosyst Eng 44:595–605. https://doi.org/10.1007/s00449-020-02473-8
Mokwatlo SC, Brink HG, Nicol W (2020) Effect of shear on morphology, viability and metabolic activity of succinic acid-producing Actinobacillus succinogenes biofilms. Bioproc Biosyst Eng 43:1253–1263. https://doi.org/10.1007/s00449-020-02322-8
Armato C, Ahmed D, Agostino V, Traversi D, Degan R, Tommasi T, Margaria V, Sacco A, Gilli G, Quaglio M, Saracco G, Schilirò T (2019) Anodic microbial community analysis of microbial fuel cells based on enriched inoculum from freshwater sediment. Bioproc Biosyst Eng 42:697–709. https://doi.org/10.1007/s00449-019-02074-0
Sun D, Cheng S, Wang A, Li F, Logan BE, Cen K (2015) Temporal-Spatial changes in viabilities and electrochemical properties of anode biofilms. Environ Sci Technol 49:5227–5235. https://doi.org/10.1021/acs.est.5b00175
Sun D, Cheng S, Zhang F, Logan BE (2017) Current density reversibly alters metabolic spatial structure of exoelectrogenic anode biofilms. J Power Sources 356:566–571. https://doi.org/10.1016/j.jpowsour.2016.11.115
Yang C, Chen M, Qian Y, Zhang L, Lu M, Xie X, Huang L, Huang W (2019) Packed anode derived from cocklebur fruit for improving long-term performance of microbial fuel cells. Sci China Mater 62:645–652. https://doi.org/10.1007/s40843-018-9368-y
Du Q, Mu Q, Cheng T, Li N, Wang X (2018) Real-Time imaging revealed that exoelectrogens from wastewater are selected at the center of a gradient electric field. Environ Sci Technol 52:8939–8946. https://doi.org/10.1021/acs.est.8b01468
Yang J, Cheng S, Li C, Sun Y, Huang H (2019) Shear stress affects biofilm structure and consequently current generation of bioanode in microbial electrochemical systems (MESs). Front Microbiol. https://doi.org/10.3389/fmicb.2019.00398
Li T, Zhou L, Qian Y, Wan L, Du Q, Li N, Wang X (2017) Gravity settling of planktonic bacteria to anodes enhances current production of microbial fuel cells. Appl Energ 198:261–266. https://doi.org/10.1016/j.apenergy.2017.04.078
Yang G, Huang L, Yu Z, Liu X, Chen S, Zeng J, Zhou S, Zhuang L (2019) Anode potentials regulate Geobacter biofilms: new insights from the composition and spatial structure of extracellular polymeric substances. Water Res 159:294–301. https://doi.org/10.1016/j.watres.2019.05.027
Li T, Zhou Q, Zhou L, Yan Y, Liao C, Wan L, An J, Li N, Wang X (2020) Acetate limitation selects Geobacter from mixed inoculum and reduces polysaccharide in electroactive biofilm. Water Res 177:115776. https://doi.org/10.1016/j.watres.2020.115776
Yang J, Cheng S, Li P, Huang H, Cen K (2019) Sensitivity to oxygen in microbial electrochemical systems biofilms. iScience 13:163–172. https://doi.org/10.1016/j.isci.2019.01.022
Zhang L, Zhu X, Li J, Liao Q, Ye D (2011) Biofilm formation and electricity generation of a microbial fuel cell started up under different external resistances. J Power Sources 196:6029–6035. https://doi.org/10.1016/j.jpowsour.2011.04.013
Rossi R, Logan BE (2020) Impact of external resistance acclimation on charge transfer and diffusion resistance in bench-scale microbial fuel cells. Bioresource Technol 318:123921. https://doi.org/10.1016/j.biortech.2020.123921
Lyon DY, Buret F, Vogel TM, Monier J (2010) Is resistance futile? Changing external resistance does not improve microbial fuel cell performance. Bioelectrochemistry 78:2–7. https://doi.org/10.1016/j.bioelechem.2009.09.001
Rismani-Yazdi H, Christy AD, Carver SM, Yu Z, Dehority BA, Tuovinen OH (2011) Effect of external resistance on bacterial diversity and metabolism in cellulose-fed microbial fuel cells. Bioresource Technol 102:278–283. https://doi.org/10.1016/j.biortech.2010.05.012
Franks AE, Nevin KP, Jia H, Izallalen M, Woodard TL, Lovley DR (2009) Novel strategy for three-dimensional real-time imaging of microbial fuel cell communities: monitoring the inhibitory effects of proton accumulation within the anode biofilm. Energ Environ Sci 2:113–119. https://doi.org/10.1039/b816445b
Renslow RS, Babauta JT, Majors PD, Beyenal H (2013) Diffusion in biofilms respiring on electrodes. Energy Environ Sci 6:595–607. https://doi.org/10.1039/c2ee23394k
Xiao Y, Zhang E, Zhang J, Dai Y, Yang Z, Christensen H, Ulstrup J, Zhao F (2017) Extracellular polymeric substances are transient media for microbial extracellular electron transfer. Sci Adv 3:e1700623. https://doi.org/10.1126/sciadv.1700623
Gurumurthy DM, Bharagava RN, Kumar A, Singh B, Ashfaq M, Saratale GD, Mulla SI (2019) EPS bound flavins driven mediated electron transfer in thermophilic Geobacillus sp. Microbiol Res 229:126324. https://doi.org/10.1016/j.micres.2019.126324
Cheng S, Wu J (2013) Air-cathode preparation with activated carbon as catalyst, PTFE as binder and nickel foam as current collector for microbial fuel cells. Bioelectrochemistry 92:22–26. https://doi.org/10.1016/j.bioelechem.2013.03.001
Yang J, Cheng S (2019) Effects of using anode biofilm and cathode biofilm bacteria as inoculum on the start-up, electricity generation, and microbial community of air-cathode single-chamber microbial fuel cells. Pol J Environ Stud 28:693–700
Yang X, Beyenal H, Harkin G, Lewandowski Z (2000) Quantifying biofilm structure using image analysis. J Microbiol Meth 39:109–119. https://doi.org/10.1016/S0167-7012(99)00097-4
Beyenal H, Lewandowski Z, Harkin G (2004) Quantifying biofilm structure: facts and fiction. Biofouling 20:1–23. https://doi.org/10.1080/0892701042000191628
Yang J, Cheng S, Sun Y, Li C (2017) Improving the power generation of microbial fuel cells by modifying the anode with single-wall carbon nanohorns. Biotechnol Lett 39:1515–1520. https://doi.org/10.1007/s10529-017-2384-4
Ni B, Zeng RJ, Fang F, Xu J, Sheng G, Yu H (2009) A novel approach to evaluate the production kinetics of extracellular polymeric substances (EPS) by activated sludge using weighted nonlinear least-squares analysis. Environ Sci Technol 43:3743–3750. https://doi.org/10.1021/es9001289
Dhar BR, Sim J, Ryu H, Ren H, Santo Domingo JW, Chae J, Lee HS (2017) Microbial activity influences electrical conductivity of biofilm anode. Water Res 127:230–238. https://doi.org/10.1016/j.watres.2017.10.028
Rossi R, Logan BE (2020) Unraveling the contributions of internal resistance components in two-chamber microbial fuel cells using the electrode potential slope analysis. Electrochim Acta 348:136291. https://doi.org/10.1016/j.electacta.2020.136291
Sharma Y, Li B (2010) The variation of power generation with organic substrates in single-chamber microbial fuel cells (SCMFCs). Bioresource Technol 101:1844–1850. https://doi.org/10.1016/j.biortech.2009.10.040
Bond DR, Lovley DR (2002) Electricity production by Geobacter sulfurreducens attached to electrodes. Appl Environ Microb 69:1548–1555. https://doi.org/10.1128/AEM.69.3.1548
Yates MD, Kiely PD, Call DF, Rismani-Yazdi H, Bibby K, Peccia J, Regan JM, Logan BE (2012) Convergent development of anodic bacterial communities in microbial fuel cells. ISME J 6:2002–2013. https://doi.org/10.1038/ismej.2012.42
Strycharz-Glaven SM, Snider RM, Guiseppi-Elie A, Tender LM (2011) On the electrical conductivity of microbial nanowires and biofilms. Energ Environ Sci 4:4366–4379. https://doi.org/10.1039/c1ee01753e
Fan Y, Hu H, Liu H (2007) Sustainable power generation in microbial fuel cells using bicarbonate buffer and proton transfer mechanisms. Environ Sci Technol 41:8154–8158. https://doi.org/10.1021/es071739c
Stewart PS (2012) Mini-review: convection around biofilms. Biofouling (Chur, Switzerland) 28:187–198. https://doi.org/10.1080/08927014.2012.662641
Shen Y, Wang M, Chang IS, Ng HY (2013) Effect of shear rate on the response of microbial fuel cell toxicity sensor to Cu(II). Bioresource Technol 136:707–710. https://doi.org/10.1016/j.biortech.2013.02.069
Funding
This work was supported by the National Natural Science Foundation of China (no. 52070162, 51778562) and the National Key Research and Development Program of China (2018YFA0901300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, J., Cheng, S. External resistance acclimation regulates bio-anode: new perspective from biofilm structure and its correlation with anode performance. Bioprocess Biosyst Eng 45, 269–277 (2022). https://doi.org/10.1007/s00449-021-02658-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-021-02658-9