Abstract
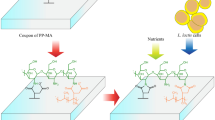
Polypropylene was modified to contain chitosan and evaluate its ability to generate Lactobacillus casei biofilms and their lactic acid production. Biofilm formation was carried out in either rich or minimal media. The chitosan-modified polypropylene harbored ~ 37% more cells than the control polypropylene. The biofilms from the chitosan-modified polypropylene grown in rich medium produced ~ 2 times more lactic acid after 72 h of incubation than the control suspended cells. There was no significant difference in the production of lactic acid after 72 h by L. casei biofilms on the chitosan-modified polypropylene grown in minimal media as compared with cells in suspension after 48 h and 72 h of incubation. Infrared spectroscopy confirmed higher deposition of nutrients and biomass on the chitosan-modified polypropylene as compared to the chitosan-free polypropylene. Electron and atomic force microscopy confirmed thicker biofilms when rich media were used to grow them as compared to minimal medium.








Similar content being viewed by others
References
Ercan D, Pongtharangkul T, Demirci A, Pometto AL III (2015) Applications of biofilm reactors for production of value-added products by microbial fermentation. In: Anthony LP, Ali D (eds) Biofilms in the food environment. Wiley, pp 255–283
Verbelen PJ, De Schutter DP, Delvaux F et al (2006) Immobilized yeast cell systems for continuous fermentation applications. Biotechnol Lett 28:1515–1525. https://doi.org/10.1007/s10529-006-9132-5
Klein J, Ziehr H (1990) Immobilization of microbial cells by adsorption. J Biotechnol 16:1–15. https://doi.org/10.1016/0168-1656(90)90061-f
Oliveira R (1997) Understanding adhesion: a means for preventing fouling. Exp Therm Fluid Sci 14:316–322. https://doi.org/10.1016/S0894-1777(96)00134-3
Hermanson GT (2008) Bioconjugate techniques, 2nd edn. Academic Press, Boston
Genari AN, Passos FV, Passos FML (2003) Configuration of a bioreactor for milk lactose hydrolysis. J Dairy Sci 86:2783–2789. https://doi.org/10.3168/jds.S0022-0302(03)73875-2
Krasaekoopt W, Bhandari B, Deeth H (2003) Evaluation of encapsulation techniques of probiotics for yoghurt. Int Dairy J 13:3–13. https://doi.org/10.1016/S0958-6946(02)00155-3
Lamboley L, St-Gelais D, Champagne CP, Lamoureux M (2003) Growth and morphology of thermophilic dairy starters in alginate beads. J Gen Appl Microbiol 49:205–214
Lemay MJ, Champagne CP, Gariépy C, Saucier L (2002) A comparison of the effect of meat formulation on the heat resistance of free or encapsulated cultures of Lactobacillus sakei. J Food Sci 67:3428–3434. https://doi.org/10.1111/j.1365-2621.2002.tb09601.x
Champagne CP, Lee BH, Saucier L (2010) Immobilization of cells and enzymes for fermented dairy or meat products. In: Zuidam NJ, Nedović V (eds) Encapsulation technologies for active food ingredients and food processing. Springer, New York, pp 345–365
Champagne CP, Gardner NJ, Lacroix C (2007) Fermentation technologies for the production of exopolysaccharide-synthesizing Lactobacillus rhamnosus concentrated cultures. Electron J Biotechnol 10:211–220
Morin N, Bernier-Cardou M, Champagne CP (1992) Production of Lactococcus lactis biomass by immobilized cell technology. J Ind Microbiol 9:131–135. https://doi.org/10.1007/BF01569745
Goddard JM, Hotchkiss JH (2007) Polymer surface modification for the attachment of bioactive compounds. Prog Polym Sci 32:698–725
Bastarrachea LJ, Denis-Rohr A, Goddard JM (2015) Antimicrobial food equipment coatings: applications and challenges. Annu Rev Food Sci Technol 6:97–118. https://doi.org/10.1146/annurev-food-022814-015453
Backer WE, Hu G (2001) Introduction. In: Baker WE, Scott CE, Hu G (eds) Reactive polymer blending. Cincinnati, OH, pp 2–12
Gagon AT, Britt DW, Bastarrachea LJ (2020) Antimicrobial light-activated polypropylene modified with chitosan: characterization and reusability. J Agric Food Chem 68:13076–13082. https://doi.org/10.1021/acs.jafc.9b06009
Gagon AT, Britt DW, Bastarrachea LJ (2020) Zein-modified antimicrobial polypropylene: characterization and reusability upon UV-A light exposure. LWT 121:108983. https://doi.org/10.1016/j.lwt.2019.108983
Bastarrachea LJ (2019) Antimicrobial polypropylene with ε-poly(lysine): effectiveness under UV-A light and food storage applications. LWT 102:276–283. https://doi.org/10.1016/j.lwt.2018.12.047
Bastarrachea LJ, Goddard JM (2016) Self-healing antimicrobial polymer coating with efficacy in the presence of organic matter. Appl Surf Sci 378:479–488. https://doi.org/10.1016/j.apsusc.2016.03.198
Machluf M (2005) Protein therapeutic delivery using encapsulated cell platform. In: Nedović V, Willaert R (eds) Applications of cell immobilisation biotechnology. Springer, Netherlands, pp 197–210
Nedović V, Willaert R, Leskošek-Čukalović I et al (2005) Beer production using immobilized cells. In: Nedović V, Willaert R (eds) Applications of cell immobilization biotechnology. Springer, Netherlands, pp 259–273
Willaert Ronnie G, Baron Gino V (1996) Gel eentrapment and micro-encapsulation: methods, applications and engineering principles. Rev Chem Eng 12:1–205. https://doi.org/10.1515/REVCE.1996.12.1-2.1
Ho KL, Pometto AL 3rd, Hinz PN et al (1997) Ingredient selection for plastic composite supports for L-(+)-lactic acid biofilm fermentation by Lactobacillus casei subsp. rhamnosus. Appl Environ Microbiol 63:2516–2523. https://doi.org/10.1128/AEM.63.7.2516-2523.1997
Malinconico M, Vink ETH, Cain A (2018) Applications of poly(lactic acid) in commodities and specialties. Adv Polym Sci 282:35–50
Vinay-Lara E, Wang S, Bai L et al (2016) Lactobacillus casei as a biocatalyst for biofuel production. J Ind Microbiol Biotechnol 43:1205–1213. https://doi.org/10.1007/s10295-016-1797-8
Kandler O, Weiss N (1986) Genus Lactobacillus. In: Sneath H, Mair N, Sharpe M, Holt J (eds) Bergey’s manual of systematic bacteriology. Williams and Wilkins, Baltimore, pp 1063–1065
Demirci A, Pometto AL, Johnson KE (1993) Evaluation of biofilm reactor solid support for mixed-culture lactic acid production. Appl Microbiol Biotechnol 38:728–733. https://doi.org/10.1007/BF00167135
Demirci A, Pometto AL (1995) Repeated-batch fermentation in biofilm reactors with plastic-composite supports for lactic acid production. Appl Microbiol Biotechnol 43:585–589. https://doi.org/10.1007/BF00164758
Fiehn O (2016) Metabolomics by gas chromatography-mass spectrometry: combined targeted and untargeted profiling. Curr Protoc Mol Biol 114:30.4.1-30.4.32. https://doi.org/10.1002/0471142727.mb3004s114
Smith BC (1998) Infrared spectral interpretation: a systematic approach. Taylor & Francis
Barish JA, Goddard JM (2013) Anti-fouling surface modified stainless steel for food processing. Food Bioprod Process 91:352–361. https://doi.org/10.1016/j.fbp.2013.01.003
Santos MI, Gerbino E, Tymczyszyn E, Gomez-Zavaglia A (2015) Applications of infrared and raman spectroscopies to probiotic investigation. Foods (Basel, Switzerland) 4:283–305. https://doi.org/10.3390/foods4030283
Liu J, Deng Y, Li L et al (2018) Discovery and control of culturable and viable but non-culturable cells of a distinctive Lactobacillus harbinensis strain from spoiled beer. Sci Rep 8:11446. https://doi.org/10.1038/s41598-018-28949-y
Acknowledgements
Authors would like to thank Dr. John Yun from SI group, Inc. for providing Polybond 7200 and Dr. FenAnn Shen from the Microscopy Core Facility at Utah State University (Logan, UT) for assistance in the Scanning Electron Microscopy analyses. This work was supported by the Utah Agricultural Experiment Station UTA01377 and approved as Journal Paper 9500.
Funding
Utah Agricultural Experiment Station project UTA01377.
Author information
Authors and Affiliations
Contributions
LJB designed and executed experiments, authors DWB and REW aided in surface and metabolite analytical techniques, author AD provided additional assistance in experimental design and results interpretation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bastarrachea, L.J., Britt, D.W., Ward, R.E. et al. Development of bioactive solid support for immobilized Lactobacillus casei biofilms and the production of lactic acid. Bioprocess Biosyst Eng 45, 217–226 (2022). https://doi.org/10.1007/s00449-021-02654-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-021-02654-z