Abstract
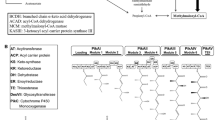
Bitespiramycin is composed of nine main acylated spiramycin components with isovaleryspiramycin as the major component. However, even with excellent therapeutic effects, its application and industrialization are restricted due to its low titer. In this study, the exogenous addition of A-Factor analogue 1,4-butyrolactone (1,4-BL) stimulated an improvement in bitespiramycin biological titer by 29% with a tiny influence on concentration of major component. Moreover, the mechanism of 1,4-BL stimulating effect was preliminarily explored by the analyses of three key enzyme activities, intracellular metabolite profiling and metabolic flux distribution. All results coordinately revealed that the extensive accumulation of methylmalonyl-CoA and acetyl-CoA was the direct reason for the enhanced bitespiramycin biosynthesis. This study would provide theoretical and technical basis for the application of 1,4-BL addition strategy to industrial bitespiramycin production.









Similar content being viewed by others
References
Biarnes-Carrera M, Breitling R, Takano E (2015) Butyrolactone signalling circuits for synthetic biology. Curr Opin Chem Biol 28:91–98
Niu G, Chater KF, Tian Y, Zhang J, Tan H (2016) Specialised metabolites regulating antibiotic biosynthesis in Streptomyces spp. FEMS Microbiol Rev 40(4):554–573
Mariam J, Anand R (2018) Fluorescence Quenching Studies of γ-Butyrolactone-Binding Protein (CprB) from Streptomyces coelicolor A3 (2) Quorum Sensing. Humana Press 2018:131–143
Takano E (2006) γ-Butyrolactones: Streptomyces signalling molecules regulating antibiotic production and differentiation. Curr Opin Microbiol 9(3):287–294
Yang YK, Shimizu H, Shioya S, Suga KI, Nihira T, Yamada Y (1995) Optimum autoregulator addition strategy for maximum virginiamycin production in batch culture of Streptomyces virginiae. Biotechnol Bioeng 46(5):437–442
Yang YK, Morikawa M, Shimizu H, Shioya S, Suga KI, Nihira T, Yamada Y (1996) Maximum virginiamycin production by optimization of cultivation conditions in batch culture with autoregulator addition. Biotechnol Bioeng 49(4):437–444
Yang YH, Kim TW, Park SH, Lee K, Park HY, Song E, Joo HS, Kim YG, Hahn JS, Kim BG (2009) Cell-free Escherichia coli-based system to screen for quorum-sensing molecules interacting with quorum receptor proteins of Streptomyces coelicolor. Appl Environ Microb 75(19):6367–6372
Miyake K, Horinouchi S, Yoshida M, Chiba N, Mori K, Nogawa N, Morikawa N, Beppu T (1989) Detection and properties of A-factor-binding protein from Streptomyces griseus. J Bacteriol 171(8):4298–4302
Liu, X, Sun, X, He, W, Tian, X, Zhuang, Y, Chu, J (2019) Dynamic changes of metabolomics and expression of candicidin biosynthesis gene cluster caused by the presence of a pleiotropic regulator AdpA in Streptomyces ZYJ-6. Bioprocess Biosyst Eng 1–13
Tan GY, Bai L, Zhong JJ (2013) Exogenous 1,4-butyrolactone stimulates A-factor-like cascade and validamycin biosynthesis in Streptomyces hygroscopicus 5008. Biotechnol Bioeng 110(11):2984–2993
Wang YH, Wu CH, Chu J (2010) Regulation of branched-chain amino acid catabolism: glucose limitation enhances the component of isovalerylspiramycin for the bitespiramycin production. Bioprocess Biosyst Eng 33(2):257–265
Hong M, Huang M, Chu J (2016) Impacts of proline on the central metabolism of an industrial erythromycin-producing strain Saccharopolyspora erythraea via 13C labeling experiments. J Biotechnol 231:1–8
Chen Y, Huang M, Wang Z (2013) Controlling the feed rate of glucose and propanol for the enhancement of erythromycin production and exploration of propanol metabolism fate by quantitative metabolic flux analysis. Bioprocess Biosyst Eng 36:1445
Choi KR, Kim WJ, Lee SY (2018) Metabolomics for industrial fermentation. Bioprocess Biosyst Eng 41(7):1073–1077
Vu-Trong K, Bhuwapathanapun S, Grav PP (1980) Metabolic regulation in tylosin-producing Streptomyces fradiae: regulatory role of adenylate nucleotide pool and enzyme involved in biosynthesis of tylonolide precursors. Antimicrob Agents Chemother 17(4):519
Coze F, Gilard F, Tcherkez G (2013) Carbon-flux distribution within Streptomyces coelicolor metabsolism: a comparison between the actinorhodin-producing strain M145 and its non-producing derivative M1146. PLoS ONE 8(12):e84151
Borodina I, Siebring J, Zhang J (2008) Antibiotic overproduction in Streptomyces coelicolor A3 2 mediated by phosphofructokinase deletion. J Biol Chem 283(37):25186
Biarnes Carrera M, Lee CK, Nihira T (2018) Orthogonal regulatory circuits for Escherichia coli based on the γ-butyrolactone system of Streptomyces coelicolor. ACS Synthetic Biol 7(4):1043–1055
Acknowledgments
This work was financially supported by the National Basic Research Program of China (973 Program) (No. 2012CB721006), National Natural Science Foundation of China (No. 21276081), the National Scientific and Technological Major Special Project (Significant Creation of New drugs), No. 2011ZX09203-001-03, Research Fund for the Doctoral Program of Higher Education of China (No. 20110074110015).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A
-
1.
G6P + 2NADP → Ru5P + CO2 + 2NADPH
-
2.
Ru5P → 0.667F6P + 0.333G3P
-
3.
G6P → F6P
-
4.
F6P + ATP → F1,6P
-
5.
F1,6P → DHAP + G3AP
-
6.
DHAP → G3P
-
7.
G3P + NAD + ADP → 3PG + NADH + ATP
-
8.
3PG → PEP
-
9.
PEP → Pyr + ATP
-
10.
Pyr + NAD → AcCoA + CO2 + NADH
-
11.
AcCoA + OXA → Citrate
-
12.
Citrate + NAD → AKG + CO2 + NADH
-
13.
AKG + FAD + GDP + 2NAD → OXA + CO2 + GTP + FADH + 2NADH
-
14.
Pyr + CO2 → OXA
-
15.
FADH + ADP + 0.5O2 → FAD + 0.5H2O + ATP
-
16.
NADH + ADP + 0.5O2 → NAD + 0.5H2O + 2ATP
-
17.
SUCCoA → METCoA
-
18.
AcCoA + HCO3−1 + ATP → MalCoA
-
19.
4Malonyl-CoA + Methylmalonyl-CoA + Ethylmalonyl-CoA + Methoxymalonyl-ACP → Platenolide
-
20.
Glc + ATP → G6P + ADP
-
21.
0.067G6P + 0.064Ru5P + 0.009G3P + 0.065PG + 0.05PEP + 0.176Pyr + 0.095OXA + 0.102AKG + 0.249AcCoA + NADPH + NAD + ATP → C3.93H7.35O2.83N0.58 (Biomass) + NADP + NADH + CO2
Appendix B
1 | 3PG | 3-Phosphoglycerate |
2 | AcCoA | Acetyl coenzyme A |
3 | DHAP | Dihydroxyacetone phosphate |
4 | F6P | Fructose-6-phosphate |
5 | FADH | Flavin adenine dinucleotide, reduced |
6 | G6P | Glucose-6-phosphate |
7 | G3P | Glyceraldehyde-3-phosphate |
8 | Glc | Glucose |
9 | AKG | α-Ketoglutarate |
10 | NADH | Nicotinamide adenine dinucleotide,reduced |
11 | OAA | Oxaloacetate |
12 | PEP | Phosphoenolpyruvate |
13 | PYR | Pyruvate |
14 | Ru5P | Ribulose-5-phosphate |
Rights and permissions
About this article
Cite this article
Gao, X., Wang, Y. & Chu, J. A preliminary study on the impact of exogenous A-Factor analogue 1,4-butyrolactone on stimulating bitespiramycin biosynthesis. Bioprocess Biosyst Eng 42, 1903–1913 (2019). https://doi.org/10.1007/s00449-019-02184-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-019-02184-9