Abstract
An efficient strategy for laccase production in Trametes versicolor cultures was developed using vanillic acid as the inducer. The optimized vanillic acid treatment strategy consisted of exposing 2-day-old mycelia cultures to 80 mg/L vanillic acid. After 4 days, laccase activity of 588.84 U/L was achieved in flasks which represented a 1.79-fold increase compared to the control. In 200-L airlift bioreactor, the maximal laccase activity reached up to 785.12 U/L using the optimized vanillic acid treatment strategy. The zymograms of culture supernatants revealed three bands with laccase activity, among which Lac1 and Lac2 were abundant laccase isoforms constitutively expressed, and Lac3 was an inducible isozyme by vanillic acid. The results of real-time quantitative PCR showed that the transcription level of lcc in T. versicolor cultures grown with vanillic acid for 7 days was about 5.64-fold greater than that without vanillic acid in flasks. In 200-L airlift bioreactor cultures of T. versicolor with addition of vanillic acid, the transcript level of lcc at day 7 was 2.62-fold higher than that in flasks with vanillic acid due to the good mass transfer and oxygen supply in the bioreactor system. This study provides a basis for understanding the induction mechanism of vanillic acid for laccase production and has good potential for industrial applications.





Similar content being viewed by others
References
Murugesan K, Arulmani M, Nam IH, Kim YM, Chang YS, Kalaichelvan PT (2006) Purification and characterization of laccase produced by a white rot fungus Pleurotus sajor-caju under submerged culture condition and its potential in decolorization of azo dyes. Appl Microbiol Biot 72:939–946
Jin XC, Ning Y (2013) Laccase production optimization by response surface methodology with Aspergillus fumigatus AF1 in unique inexpensive medium and decolorization of different dyes with the crude enzyme or fungal pellets. J Hazard Mater 262:870–877
Saparrat M, Balatti PA, Martínez MJ, Jurado M (2010) Differential regulation of laccase gene expression in Coriolopsis rigida LPSC No. 232. Fungal Biol-UK 114:999–1006
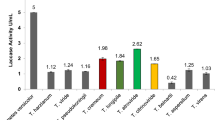
Terrón MC, González T, Carbajo JM, Yagüe S, Arana-Cuenca A, Téllez A, Dobson AD, González AE (2004) Structural close-related aromatic compounds have different effects on laccase activity and on lcc gene expression in the ligninolytic fungus Trametes sp. I-62. Fungal Genet Biol 41:954–962
Scheel T, Höfer M, Ludwig S, Hölker U (2000) Differential expression of manganese peroxidase and laccase in white-rot fungi in the presence of manganese or aromatic compounds. Appl Microbiol Biot 54:686–691
Munoz C, Guillen F, Martinez A, Martinez M (1997) Induction and characterization of laccase in the ligninolytic fungus Pleurotus eryngii. Curr Microbiol 34:1–5
Couto SR, Herrera JLT (2007) Laccase production at reactor scale by filamentous fungi. Biotechnol Adv 25:558–569
Couto SR, Herrera JLT (2006) Industrial and biotechnological applications of laccases: a review. Biotechnol Adv 24:500–513
Baldrian P (2006) Fungal laccases–occurrence and properties. FEMS Microbiol Rev 30:215–242
Hou H, Zhou J, Wang J, Du C, Yan B (2004) Enhancement of laccase production by Pleurotus ostreatus and its use for the decolorization of anthraquinone dye. Process Biochem 39:1415–1419
Dhillon GS, Kaur S, Brar SK (2012) In-vitro decolorization of recalcitrant dyes through an ecofriendly approach using laccase from Trametes versicolor grown on brewer’s spent grain. Int Biodeter Biodegr 72:67–75
Lorenzo M, Moldes D, Couto SR, Sanroman A (2002) Improving laccase production by employing different lignocellulosic wastes in submerged cultures of Trametes versicolor. Bioresource Technol 82:109–113
Wang F, Guo C, Wei T, Zhang T, Liu CZ (2012) Heat shock treatment improves Trametes versicolor laccase production. Appl Biochem Biotech 168:256–265
Wang F, Ma AZ, Guo C, Zhuang GQ, Liu CZ (2013) Ultrasound-intensified laccase production from Trametes versicolor. Ultrason Sonochem 20:118–124
Wang HL, Li P, Yang YH, Liu YF (2015) Overproduction of laccase from a newly isolated Ganoderma lucidum using the municipal food waste as main carbon and nitrogen supplement. Bioprocess Biosyst Eng 38:957–966
Collins PJ, Dobson A (1997) Regulation of laccase gene transcription in Trametes versicolor. Appl Environ Microb 63:3444–3450
Dong JL, Zhang YW, Zhang RH, Huang WZ, Zhang YZ (2005) Influence of culture conditions on laccase production and isozyme patterns in the white-rot fungus Trametes gallica. J Basic Microb 45:190–198
Singha S, Panda T (2014) Improved production of laccase by Daedalea flavida: consideration of evolutionary process optimization and batch-fed culture. Bioprocess Biosyst Eng 37:493–503
Soden DM, Dobson AD (2001) Differential regulation of laccase gene expression in Pleurotus sajor-caju. Microbiology 147:1755–1763
Wang F, Hu JH, Guo C, Liu CZ (2014) Enhanced laccase production by Trametes versicolor using corn steep liquor as both nitrogen source and inducer. Bioresource technol 166:602–605
Piscitelli A, Giardina P, Lettera V, Pezzella C, Sannia G, Faraco V (2011) Induction and transcriptional regulation of laccases in fungi. Curr Genomics 12:104
Jung H, Xu F, Li K (2002) Purification and characterization of laccase from wood-degrading fungus Trichophyton rubrum LKY-7. Enzyme Microb Tech 30:161–168
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Fonseca MI, Ramos-Hryb AB, Farina JI, Afanasiuk SSS, Villalba LL, Zapata PD (2014) Effect of chemical and metallic compounds on biomass, mRNA levels and laccase activity of Phlebia brevispora BAFC 633. World J Mcrobiol Biotechnol 30:2251–2262
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Cambria MT, Ragusa S, Calabrese V, Cambria A (2011) Enhanced laccase production in white-rot fungus Rigidoporus lignosus by the addition of selected phenolic and aromatic compounds. Appl Biochem Biotech 163:415–422
Galhaup C, Wagner H, Hinterstoisser B, Haltrich D (2002) Increased production of laccase by the wood-degrading basidiomycete Trametes pubescens. Enzyme Microb Tech 30:529–536
Shakhova N, Golenkina S, Stepanova E, Loginov D, Psurtseva N, Fedorova T, Koroleva O (2011) Effect of submerged cultivation conditions and inducers on biosynthesis of extracellular laccase by a Trametes versicolor 1666 strain. Appl Biochem Microbiol 47:808–816
Cui YQ, van der Lans RGJM, Luyben K (1998) Effects of dissolved oxygen tension and mechanical forces on fungal morphology in submerged fermentation. Biotechnol Bioeng 57:409–419
Calvo AM, Wilson RA, Bok JW, Keller NP (2002) Relationship between secondary metabolism and fungal development. Microbiol Mol Biol R 66:447–459
Rancano G, Lorenzo M, Molares N, Couto SR, Sanromán MÁ (2003) Production of laccase by Trametes versicolor in an airlift fermentor. Process Biochem 39:467–473
Fenice M, Sermanni GG, Federici F, D’Annibale A (2003) Submerged and solid-state production of laccase and Mn-peroxidase by Panus tigrinus on olive mill wastewater-based media. J Biotechnol 100:77–85
Tavares A, Coelho M, Agapito M, Coutinho J, Xavier A (2006) Optimization and modeling of laccase production by Trametes versicolor in a bioreactor using statistical experimental design. Appl Biochem Biotech 134:233–248
Thiruchelvam A, Ramsay JA (2007) Growth and laccase production kinetics of Trametes versicolor in a stirred tank reactor. Appl Microbiol Biot 74:547–554
Giatti Marques De Souza C et al (2004) Production of laccase isoforms by Pleurotus pulmonarius in response to presence of phenolic and aromatic compounds. J Basic Microb 44:129–136
Chen S, Ma D, Ge W, Buswell JA (2003) Induction of laccase activity in the edible straw mushroom, Volvariella volvacea. FEMS Microbiol Lett 218:143–148
Bai Z, Harvey LM, McNeil B (2003) Oxidative stress in submerged cultures of fungi. Crit Rev Biotechnol 23:267–302
Coman C, Moţ AC, Gal E, Pârvu M, Dumitrescu RS (2013) Laccase is upregulated via stress pathways in the phytopathogenic fungus Sclerotinia sclerotiorum. Fungal Biol-UK 117:528–539
Eyre C, Muftah W, Hiscox J, Hunt J, Kille P, Boddy L, Rogers HJ (2010) Microarray analysis of differential gene expression elicited in Trametes versicolor during interspecific mycelial interactions. Fungal Biol-UK 114:646–660
Kronstad J, De Maria A, Funnell D, Laidlaw RD, Lee N, de Sá MM, Ramesh M (1998) Signaling via cAMP in fungi: interconnections with mitogen-activated protein kinase pathways. Arch Microbiol 170:395–404
Acknowledgments
This work was financially supported by the National Basic Research Program (973 Program) of China (No. 2013CB733600), the National Natural Science Foundation of China (No. 21476242), the National Key Technology Research and Development Program of China (No. 2015BAK45B01), the National High Technology Research and Development Program (863 Program) of China (No. 2012AA101803), the Knowledge Innovation Program of the Chinese Academy of Sciences (No. KSZD -EW-Z-015). We acknowledge Dr. Lim Jit Kang (the Chinese Academy of Sciences Fellowships for Young International Scientists, No. 2015VMC060) for help in editing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, KF., Hu, JH., Guo, C. et al. Scale-up laccase production from Trametes versicolor stimulated by vanillic acid. Bioprocess Biosyst Eng 39, 1041–1049 (2016). https://doi.org/10.1007/s00449-016-1582-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-016-1582-0