Abstract
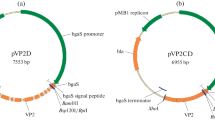
Complementary DNA (cDNA) encoding the new versatile peroxidase from the ligninolytic basidiomycete Pleurotus eryngii has been expressed in the ascomycete Emericella nidulans. In recombinant E. nidulans cultures, the pH reached values as high as 8.3, correlating with a sharp decrease in peroxidase activity. Peroxidase was rapidly inactivated at alkaline pH, but was comparatively stable at acidic pH. The peroxidase inactivation in alkaline buffer could be reversed by adding Ca2+ and lowering the pH. However, reactivation did not result after incubating the enzyme in non-buffered E. nidulans cultures that reached pH 7.5. To optimize recombinant peroxidase production, the effect of controlling the pH in E. nidulans bioreactor cultures was studied. An extended growth period, and a significant increase in the recombinant peroxidase level (5.3-fold higher activity than in the bioreactor without pH control) was obtained when the pH was maintained at 6.8, showing that culture pH is an important parameter for recombinant peroxidase production.




Similar content being viewed by others
References
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Annu Rev Microbiol 41:465–505
Cai DY, Tien M (1993) Lignin-degrading peroxidases of Phanerochaete chrysosporium. J Biotechnol 30:79–90
Hatakka A (1994) Lignin-modifying enzymes from selected white-rot fungi—production and role in lignin degradation. FEMS Microbiol Rev 13:125–135
Gold MH, Youngs HL, Gelpke MD (2000) Manganese peroxidase. Met Ions Biol Syst 37:559–586
Mester T, Field JA (1998) Characterization of a novel manganese peroxidase-lignin peroxidase hybrid isozyme produced by Bjerkandera species strain BOS55 in the absence of manganese. J Biol Chem 273:15412–15417
Heinfling A, Ruiz-Dueñas FJ, Martínez MJ, Bergbauer M, Szewzyk U, Martínez AT (1998) A study on reducing substrates of manganese-oxidizing peroxidases from Pleurotus eryngii and Bjerkandera adusta. FEBS Lett 428:141–146
Martínez AT (2002) Molecular biology and structure-function of lignin-degrading peroxidases. Enzyme Microb Technol 30:425–444
Ruiz-Dueñas FJ, Martínez MJ, Martínez AT (1999) Molecular characterization of a novel peroxidase isolated from the ligninolytic fungus Pleurotus eryngii. Mol Microbiol 31:223–236
Camarero S, Ruiz-Dueñas FJ, Sarkar S, Martínez MJ, Martínez AT (2000) The cloning of a new peroxidase found in lignocellulose cultures of Pleurotus eryngii and sequence comparison with other fungal peroxidases. FEMS Microbiol Lett 191:37–43
Ruiz-Dueñas FJ, Martínez MJ, Martínez AT (1999) Heterologous expression of Pleurotus eryngii peroxidase confirms its ability to oxidize Mn2+ and different aromatic substrates. Appl Environ Microbiol 65:4705–4707
Devchand M, Gwynne DI (1991) Expression of heterologous proteins in Aspergillus. J Biotechnol 17:3–9
Cove DJ (1966) The induction and repression of nitrate reductase in the fungus Aspergillus nidulans. Biochim Biophys Acta 113:51–56
Andersen HD, Jensen EB, Welinder KG (1992) A process for producing heme proteins. Patent (International) No. WO9216634
Stewart P, Whitwam RE, Kersten PJ, Cullen D, Tien M (1996) Efficient expression of a Phanerochaete chrysosporium manganese peroxidase gene in Aspergillus oryzae. Appl Environ Microbiol 62:860–864
Martínez MJ, Ruiz-Dueñas FJ, Guillén F, Martínez AT (1996) Purification and catalytic properties of two manganese-peroxidase isoenzymes from Pleurotus eryngii. Eur J Biochem 237:424–432
Somogyi M (1945) A new reagent for the determination of sugars. J Biol Chem 160:61–73
Ruiz-Dueñas FJ (1998) Caracterización molecular de un nuevo tipo de peroxidasa ligninolítica. PhD Thesis, Universidad Complutense de Madrid, Spain
Larrondo LF, Lobos S, Stewart P, Cullen D, Vicuña R (2001) Isoenzyme multiplicity and characterization of recombinant manganese peroxidases from Ceriporiopsis subvermispora and Phanerochaete chrysosporium. Appl Environ Microbiol 67:2070–2075
Aifa MS, Sayadi S, Gargouri A (1999) Heterologous expression of lignin peroxidase of Phanerochaete chrysosporium in Aspergillus niger. Biotechnol Lett 21:849–853
Conesa A, van den Hondel CAMJJ, Punt PJ (2000) Studies on the production of fungal peroxidases in Aspergillus niger. Appl Environ Microbiol 66:3016–3023
Conesa A, Punt PJ, van den Hondel CAMJJ (2002) Fungal peroxidases: molecular aspects and applications. J Biotechnol 93:143–158
Johnson TM, Li JKK (1991) Heterologous expression and characterization of an active lignin peroxidase from Phanerochaete chrysosporium using recombinant baculovirus. Arch Biochem Biophys 291:371–378
Johnson TM, Pease EA, Li JKK, Tien M (1992) Production and characterization of recombinant lignin peroxidase isozyme H2 from Phanerochaete chrysosporium using recombinant baculovirus. Arch Biochem Biophys 296:660–666
Whitwam RE, Gazarian IG, Tien M (1995) Expression of fungal Mn peroxidase in E. coli and refolding yield active enzyme. Biochem Biophys Res Commun 216:1013–1017
Doyle WA, Smith AT (1996) Expression of lignin peroxidase H8 in Escherichia coli: folding and activation of the recombinant enzyme with Ca2+ and haem. Biochem J 315:15–19
Nie G, Reading NS, Aust SD (1998) Expression of the lignin peroxidase H2 gene from Phanerochaete chrysosporium in Escherichia coli. Biochem Biophys Res Commun 249:146–150
Pérez-Boada M, Doyle WA, Ruiz-Dueñas FJ, Martínez MJ, Martínez AT, Smith AT (2002) Expression of Pleurotus eryngii versatile peroxidase in Escherichia coli and optimisation of in vitro folding. Enzyme Microb Tech 30:518–524
Tuisel H, Sinclair R, Bumpus JA, Ashbaugh W, Brock BJ, Aust SD (1990) Lignin peroxidase H2 from Phanerochaete chrysosporium: purification, characterization and stability to temperature and pH. Arch Biochem Biophys 279:158–166
Youngs HL, Moënne-Loccoz P, Loehr TM, Gold MH (2000) Formation of a bis(histidyl) heme iron complex in manganese peroxidase at high pH and restoration of the native enzyme structure by calcium. Biochemistry 39:9994–10000
George SJ, Kvaratskhelia M, Dilworth MJ, Thorneley RNF (1999) Reversible alkaline inactivation of lignin peroxidase involves the release of both the distal and proximal site calcium ions and bishistidine co-ordination of the haem. Biochem J 344:237–244
Felenbok B (1991) The ethanol utilization regulon of Aspergillus nidulans: the alcA-alcR system as a tool for the expression of recombinant proteins. J Biotechnol 17:11–18
Panozzo C, Cornillot E, Felenbok B (1998) The CreA represor is the sole DNA-binding protein responsible for carbon catabolite repression of the alcA gene in Aspergillus nidulans via its binding to a couple of specific sites. J Biol Chem 273:6367–6372
Flipphi M, Mathieu M, Cirpus I, Panozzo C, Felenbok B (2001) Regulation of the aldehyde dehydrogenase gene (aldA) and its role in the control of the co-inducer level necessary for induction of the ethanol utilization pathway in Aspergillus nidulans. J Biol Chem 276:6950–6958
Lin W, Feldberg RS, De Bernardez-Clark E (1993) Kinetics of cell growth and heterologous glucoamylase production in recombinant Aspergillus nidulans. Biotechnol Bioeng 41:273–279
Acknowledgements
The authors thank M.A. Peñalva (CIB, CSIC, Madrid) for providing the E. nidulans strain and the plasmid palcA. This work was partially supported by the EU contract “Fungal metalloenzymes oxidizing aromatic compounds of industrial interest” (QLK3-99-590), the Comunidad de Madrid, and the Spanish Commission of Science and Technology (BIO98-610 and BIO99-908).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lú-Chau, T.A., Ruiz-Dueñas, F.J., Camarero, S. et al. Effect of pH on the stability of Pleurotus eryngii versatile peroxidase during heterologous production in Emericella nidulans. Bioprocess Biosyst Eng 26, 287–293 (2004). https://doi.org/10.1007/s00449-004-0365-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-004-0365-1