Abstract
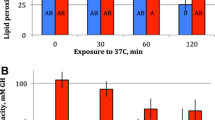
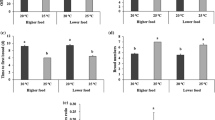
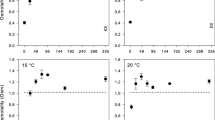
The dietary supply of polyunsaturated fatty acids (PUFA) crucially affects animals’ performance at different temperatures. However, the underlying physiological mechanisms are still insufficiently understood. Here, we analyzed lifespan and heat tolerance of four genotypes of Daphnia magna reared on either the green alga Scenedesmus obliquus that lacks long-chain (> C18) PUFA, or the heterokont alga Nannochloropsis limnetica that contains C20 PUFA, both either at saturating and near-starvation levels. A significant genotype-by-diet interaction in lifespan was observed at saturating diets. The C20 PUFA-rich diet eliminated differences in lifespan among genotypes on the PUFA-deficient diet. Corrected for body length, acute heat tolerance was higher at low than at high food concentration, at least in the older of the two age groups analyzed. Genotypes differed significantly in heat tolerance, but there were no genotype-by-diet interactions. As predicted, the C20 PUFA-rich diet resulted in higher lipid peroxidation (LPO) and a lower mitochondrial membrane potential (ΔΨm). LPO levels averaged across clones and rearing conditions were inversely related to acute heat tolerance. Yet, heat tolerance was higher on the PUFA-rich diet than on the PUFA-deficient diet, particularly in older Daphnia, indicating that the C20 PUFA-rich diet allowed Daphnia to compensate for higher LPO. In contrast, Daphnia with intermediate levels of ΔΨm showed the lowest heat tolerance. Neither LPO nor ΔΨm explained the diet effects on lifespan. We hypothesize that antioxidants present in the PUFA-rich diet may have enabled higher heat tolerance of Daphnia despite higher LPO, which may also explain the lifespan expansion of otherwise short-lived genotypes.






Similar content being viewed by others
Availability of data and materials (data transparency)
All data generated or analyzed during this study are available at faculty.etsu.edu/yampolsk/data/ScenNanno.
Code availability (software application or custom code)
N/A.
References
Anderson CE, Ekwudo MN, Jonas-Closs RA, Cho Y, Peshkin L, Kirschner MW, Yampolsky LY (2022) Lack of age-related respiratory changes in Daphnia. Biogerontology 23:85–97. https://doi.org/10.1007/s10522-021-09947-6
Angilletta MJ Jr (2011) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press
Ballweg S, Sezgin E, Doktorova M, Covino R, Reinhard J, Wunnicke D, Hänelt I, Levental I, Hummer G, Ernst R (2020) Regulation of lipid saturation without sensing membrane fluidity. Nat Commun 11:756. https://doi.org/10.1038/s41467-020-14528-1
Betancor MB, Oboh A, Ortega A, Mourente G, Navarro JC, de la Gándara F et al (2020) Molecular and functional characterisation of a putative elovl4 gene and its expression in response to dietary fatty acid profile in Atlantic bluefin tuna (Thunnus thynnus). Comp Biochem Physiol B Biochem Mol Biol 240:110372
Boyen J, Fink P, Mensens C, Hablützel PI, De Troch M (2020) Fatty acid bioconversion in harpacticoid copepods in a changing environment: a transcriptomic approach. Phil Trans R Soc B 375:20190645. https://doi.org/10.1098/rstb.2019.0645
Brett MT, Muller-Navarra DC, Ballantyne AP, Ravet JL, Goldman CR (2006) Daphnia fatty acid composition reflects that of their diet. Limnol Oceanogr 51:2428–2437. https://doi.org/10.4319/lo.2006.51.5.2428
Christensen JJ, Telle-Hansen VH, Ulven SM, Kovanen PT, Jauhiainen M, Öörni K, Holven KB (2021) The homeoviscous adaptation to dietary lipids (HADL) hypothesis is probably incorrect. Am J Clin Nutr 113:1711–1712. https://doi.org/10.1093/ajcn/nqab110
Clayton PR, Szabolcs L (2015) From alga to omega; have we reached peak (fish) oil? J R Soc Med 108:351–357. https://doi.org/10.1177/0141076815599673
Coggins BL, Collins JW, Holbrook KJ, Yampolsky LY (2017) Antioxidant capacity, lipid peroxidation, and lipid composition changes during long-term and short-term thermal acclimation in Daphnia. J Comp Physiol B 187:1091–1106. https://doi.org/10.1007/s00360-017-1090-9
Coggins BL, Pearson AC, Yampolsky LY (2021) Does geographic variation in thermal tolerance in Daphnia represent trade-offs or conditional neutrality? J Therm Biol 98:102934. https://doi.org/10.1016/j.jtherbio.2021.102934
Conde TA, Neves BF, Couto D, Melo T, Neves B, Costa M, Silva J, Domingues P, Domingues MR (2021) Microalgae as sustainable bio-factories of healthy lipids: evaluating fatty acid content and antioxidant activity. Mar Drugs 19:357. https://doi.org/10.3390/md19070357
Cook HW, McMaster CR (2002) Fatty acid desaturation and chain elongation in eukaryotes. In: Vance DE, Vance JE (eds) New comprehensive biochemistry, vol 36. Elsevier, Paris, pp 181–204
Daenen K, Andries A, Mekahli D, Van Schepdael A, Jouret F, Bammens B (2019) Oxidative stress in chronic kidney disease. Pediatr Nephrol 34:975–991. https://doi.org/10.1007/s00467-018-4005-4
Devkota R, Kaper D, Bodhicharla R, Henricsson M, Borén J, Pilon M (2021) A genetic titration of membrane composition in Caenorhabditis elegans reveals its importance for multiple cellular and physiological traits. Genetics 219:iyab093. https://doi.org/10.1093/genetics/iyab093
Ebert D (2005) Ecology, epidemiology, and evolution of parasitism in Daphnia. National Center for Biotechnology Information (US), Bethesda
Ekwudo MN, Malek MC, Anderson CE, Yampolsky LY (2022) The interplay between prior selection mild intermittent exposure and acute severe exposure in phenotypic and transcriptional response to hypoxia. Ecol Evol. https://doi.org/10.1002/ece3.9319
Frankel EN, Satué-Gracia T, Meyer AS, German JB (2002) Oxidative stability of fish and algae oils containing long-chain polyunsaturated fatty acids in bulk and in oil-in-water emulsions. J Agric Food Chem 50:2094–2099. https://doi.org/10.1021/jf0111458
Ginjupalli GK, Gerard PD, Baldwin WS (2015) Arachidonic acid enhances reproduction in Daphnia magna and mitigates changes in sex ratios induced by pyriproxyfen. Environ Toxicol Chem 34:527–535. https://doi.org/10.1002/etc.2804
Gophen M, Geller W (1984) Filter mesh size and food particle uptake by Daphnia. Oecologia 64:408–412. https://doi.org/10.1007/BF00379140
Gower JD (1988) A role for dietary lipids and antioxidants in the activation of carcinogens. Free Radic Biol Med 5:95–111. https://doi.org/10.1016/0891-5849(88)90035-4
Hazel JR (1995) Thermal adaptation in biological membranes: is homeoviscous adaptation the explanation? Annu Rev Physiol 57:19–42
Hazel JR, Williams E (1990) The role of alterations in membrane lipid composition in enabling physiological adaptation of organisms to their physical environment. Prog Lipid Res 29:167–277
Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford University Press, New York
Isanta-Navarro J, Arnott SH, Klauschies T, Martin-Creuzburg D (2021) Dietary lipid quality mediates salt tolerance of a freshwater keystone herbivore. Sci Total Environ 769:144657. https://doi.org/10.1016/j.scitotenv.2020.144657
Ishikawa A, Stuart YE, Bolnick DI, Kitano J (2021) Copy number variation of a fatty acid desaturase gene Fads2 associated with ecological divergence in freshwater stickleback populations. Biol Lett 17:20210204. https://doi.org/10.1098/rsbl.2021.0204
Jaganjac M, Cindrić M, Jakovčević A, Žarković K, Žarković N (2021) Lipid peroxidation in brain tumors. Neurochem Int 149:105118. https://doi.org/10.1016/j.neuint.2021.105118
Kabeya N, Ogino M, Ushio H, Haga Y, Satoh S, Navarro JC, Monroig Ó (2021) A complete enzymatic capacity for biosynthesis of docosahexaenoic acid (DHA, 22: 6n–3) exists in the marine Harpacticoida copepod Tigriopus californicus. Open Biol 11:200402. https://doi.org/10.1098/rsob.200402
Kelley NS, Yoshida Y, Erickson KL (2014) Do n-3 polyunsaturated fatty acids increase or decrease lipid peroxidation in humans? Metab Syndr Relat Disord 12:403–415. https://doi.org/10.1089/met.2014.0045
Kerfoot WC, Kirk KL (1991) Degree of taste discrimination among suspension-feeding cladocerans and copepods: Implications for detritivory and herbivory. Limnol Oceanogr 36:1107-l123
Kilham SS, Kreeger DA, Lynn SG, Goulden CE, Herrera L (1998) COMBO: a defined freshwater culture medium for algae and zooplankton. Hydrobiologia 377:147–159
Koch U, von Elert E, Straile D (2009) Food quality triggers the reproductive mode in the cyclical parthenogen Daphnia (Cladocera). Oecologia 159:317–324. https://doi.org/10.1007/s00442-008-1216-6
Krienitz L, Wirth M (2006) The high content of polyunsaturated fatty acids in Nannochloropsis limnetica (Eustigmatophyceae) and its implication for food web interactions, freshwater aquaculture and biotechnology. Limnol Ecol Manag Inland Waters 36:204–210. https://doi.org/10.1016/j.limno.2006.05.002
Lands WE (1992) Biochemistry and physiology of n-3 fatty acids. FASEB J 6:2530–2536. https://doi.org/10.1096/fasebj.6.8.1592205. (PMID: 1592205)
Martin-Creuzburg D, Merkel P (2016) Sterols of freshwater microalgae: potential implications for zooplankton nutrition. J Plankt Res 38:865–877. https://doi.org/10.1093/plankt/fbw034
Martin-Creuzburg D, von Elert E (2009) Good food against bad food: the role of sterols and polyunsaturated fatty acids in determining growth and reproduction in Daphnia magna. Aquat Ecol 43:843950
Martin-Creuzburg D, Sperfeld E, Wacker A (2009) Colimitation of a freshwater herbivore by sterols and polyunsaturated fatty acids. Proc Biol Sci 276:805–1814. https://doi.org/10.1098/rspb.2008.1540
Martin-Creuzburg D, Wacker A, Ziese C, Kainz MJ (2012) Dietary lipid quality affects temperature-mediated reaction norms of a freshwater key herbivore. Oecologia 168:901–912. https://doi.org/10.1007/s00442-011-2155-1
Martin-Creuzburg D, Oexle S, Wacker A (2014) Thresholds for sterol-limited growth of Daphnia magna: a comparative approach using 10 different sterols. J Chem Ecol 40:1039–1050. https://doi.org/10.1007/s10886-014-0486-1
Martin-Creuzburg D, Coggins BL, Ebert D, Yampolsky LY (2019) Rearing temperature and fatty acid supplementation jointly affect lipid fluorescence polarization and heat tolerance in Daphnia. Physiol Biochem Zool 92:408–418. https://doi.org/10.1086/704365
Matsushita Y, Miyoshi K, Kabeya N, Sanada S, Yazawa R, Haga Y et al (2020) Flatfishes colonised freshwater environments by acquisition of various DHA biosynthetic pathways. Communications Biology 3:516
Meydani SN (1996) Effect of (n-3) polyunsaturated fatty acids on cytokine production and their biologic function. Nutrition 12:S8-14
Montoya-Arroyo A, Lehnert K, Muñoz-González A, Schmid-Staiger U, Vetter W, Frank J (2022) Tocochromanol profiles in Chlorella sorokiniana, Nannochloropsis limnetica and Tetraselmis suecica confirm the presence of 11′-α-tocomonoenol in cultured microalgae independently of species and origin. Foods 11:396. https://doi.org/10.3390/foods11030396
Pap EH, Drummen GP, Winter VJ, Kooij TW, Rijken P, Wirtz KW, Op den Kamp JA, Hage WJ, Post JA (1999) Ratio-fluorescence microscopy of lipid oxidation in living cells using C11- BODIPY(581/591). Febs Lett 453:278–282
Pilecky M, Kämmer SK, Mathieu-Resuge M, Wassenaar LI, Taipale SJ, Martin-Creuzburg D, Kainz MJ (2022) Hydrogen isotopes (δ2H) of polyunsaturated fatty acids track bioconversion by zooplankton. Funct Ecol 36:538–549. https://doi.org/10.1111/1365-2435.13981
Rahman NA, Katayama T, Wahid MEA, Kasan NA, Khatoon H, Yamada Y, Takahashi K (2020) Taxon- and growth phase-specific antioxidant production by chlorophyte, bacillariophyte, and haptophyte strains isolated from tropical waters. Front Bioeng Biotechnol 8:581628. https://doi.org/10.3389/fbioe.2020.581628
Rasband WS (2018) ImageJ. U. S. National Institutes of Health, Bethesda, Maryland, USA, 1997–2018. https://imagej.nih.gov/ij/. Accessed 13 May 2023
Rivers JP, Sinclair AJ, Craqford MA (1975) Inability of the cat to desaturate essential fatty acids. Nature 258:171–173
SAS Institute (2021) JMP® Statistical software. Cary, NC, USA. www.jmp.com. Accessed 13 May 2023
Schlotz N, Ebert D, Martin-Creuzburg D (2013) Dietary supply with polyunsaturated fatty acids and resulting maternal effects influence host–parasite interactions. BMC Ecol 13:41. https://doi.org/10.1186/1472-6785-13-41
Sinensky M (1974) Homeoviscous adaptation—a homeostatic process that regulates viscosity of membrane lipids in Escherichia coli. Proc Nat Acad Sci USA 71:522–525
Sperfeld E, Wacker A (2011) Temperature- and cholesterol-induced changes in eicosapentaenoic acid limitation of Daphnia magna determined by a promising method to estimate growth saturation thresholds. Limnol Oceanogr 56:1273–1284. https://doi.org/10.4319/lo.2011.56.4.1273
Spiteller G (2001) Lipid peroxidation in aging and age-dependent diseases. Exp Gerontol 36:1425–1457. https://doi.org/10.1016/s0531-5565(01)00131-0
Stillwell W, Wassall SR (2003) Docosahexaenoic acid: membrane properties of a unique fatty acid. Chem Phys Lipids 126:1–27. https://doi.org/10.1016/s0009-3084(03)00101-4
Stillwell W, Ehringer W, Jenski LJ (1993) Docosahexaenoic acid increases permeability of lipid bilayers and tumor cells. Lipids 28:103–108
Stillwell W, Jenski LJ, Crump FT, Ehringer W (1997) Effect of docosahexaenoic acid on mouse mitochondrial membrane properties. Lipids 32:497–506. https://doi.org/10.1007/s11745-997-0064-6
Stockwell BR, Friedmann Angeli JP, Bayir H et al (2017) Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171:273–285. https://doi.org/10.1016/j.cell.2017.09.021
Tao L, Sun T, Magnuson AD, Qamar TR, Lei XG (2018) Defatted microalgae-mediated enrichment of n–3 polyunsaturated fatty acids in chicken muscle is not affected by dietary selenium vitamin E or corn oil. J Nutr 148(10):1547–1555. https://doi.org/10.1093/jn/nxy164
Twining CW, Bernhardt JR, Derry AM, Hudson CM, Ishikawa A, Kabeya N, Kainz MJ, Kitano J, Kowarik C, Ladd SN, Leal MC, Scharnweber K, Shipley JR, Matthews B (2021) The evolutionary ecology of fatty-acid variation: implications for consumer adaptation and diversification. Ecol Lett 24:1709–1731. https://doi.org/10.1111/ele.13771
Villalba JM, López-Domínguez JA, Chen Y, Khraiwesh H, González-Reyes JA, Del Río LF, Gutiérrez-Casado E, Del Río M, Calvo-Rubio M, Ariza J, de Cabo R, López-Lluch G, Navas P, Hagopian K, Burón MI, Ramsey JJ (2015) The influence of dietary fat source on liver and skeletal muscle mitochondrial modifications and lifespan changes in calorie-restricted mice. Biogerontology 16:655–670. https://doi.org/10.1007/s10522-015-9572-1
von Elert E (2002) Determination of limiting polyunsaturated fatty acids in Daphnia galeata using a new method to enrich food algae with single fatty acids. Limnol Oceanogr 47:1764–1773. https://doi.org/10.4319/lo.2002.47.6.1764
Wacker A, Martin-Creuzburg D (2007) Allocation of essential lipids in Daphnia magna during exposure to poor food quality. Funct Ecol 21:738–747. https://doi.org/10.1111/j.1365-2435.2007.01274.x
Werner C, Ilic M, von Elert E (2019) Differences in heat tolerance within a Daphnia magna population: the significance of body PUFA content. Hydrobiologia 846:17–26. https://doi.org/10.1007/s10750-018-3769-7
Windisch HS, Fink P (2019) Transcriptome sequencing of a keystone aquatic herbivore yields insights on the temperature-dependent metabolism of essential lipids. BMC Genomics 20:894. https://doi.org/10.1186/s12864-019-6268-y
Yampolsky LY, Zeng E, Lopez J, Williams PJ, Dick KB, Colbourne JK, Pfrender ME (2014) Functional genomics of acclimation and adaptation in response to thermal stress in Daphnia. BMC Genomics 15:859
Zaloga GP (2021) Narrative Review of n-3 Polyunsaturated Fatty Acid Supplementation upon Immune Functions. Resolution Molecules and Lipid Peroxidation Nutrients 13:662. https://doi.org/10.3390/nu13020662
Zamaria N (2004) Alteration of polyunsaturated fatty acid status and metabolism in health and disease. Reprod Nutr Dev 44:273–282. https://doi.org/10.1051/rnd:2004034
Zeis B, Buchen I, Wacker A, Martin-Creuzburg D (2019) Temperature-induced changes in body lipid composition affect vulnerability to oxidative stress in Daphnia magna. Comp Biochem Physiol B Biochem Mol Biol 232:101–107. https://doi.org/10.1016/j.cbpb.2019.03.008
Zinöcker MK, Svendsen K, Dankel SN (2021) The homeoviscous adaptation to dietary lipids (HADL) model explains controversies over saturated fat, cholesterol, and cardiovascular disease risk. Am J Clin Nutr 113:277–289. https://doi.org/10.1093/ajcn/nqaa322
Zou Y, Schreiber SL (2020) Progress in understanding ferroptosis and challenges in its targeting for therapeutic benefit. Cell Chem Biol 27:463–471. https://doi.org/10.1016/j.chembiol.2020.03.015
Acknowledgements
We are grateful to Cora Anderson, Millicent Ekwudo, Morad Malek, and Rachael Lowman for help in the lab, to Gerardo Arceo-Gomez for the access to a fluorescent microscope and to Bret Coggins, Aruna Kilaru, and two anonymous reviewers for helpful suggestions on the manuscript. TM has been supported by ETSU McNair Program and LY by the Impetus Foundation.
Funding
LYY and TDM were funded by ETSU McNair program and ETSU Barclay-Moore award.
Author information
Authors and Affiliations
Contributions
LYY conceived and designed the experiments. TDM and LYY performed the experiments and analyzed the data. LYY wrote the manuscript. DMC conceptualized and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no conflict of interest.
Ethical approval (include appropriate approvals or waivers)
N/A.
Consent to participate (include appropriate statements)
N/A.
Consent for publication
N/A.
Additional information
Communicated by Ulrich Sommer.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moore, T.D., Martin-Creuzburg, D. & Yampolsky, L.Y. Diet effects on longevity, heat tolerance, lipid peroxidation and mitochondrial membrane potential in Daphnia. Oecologia 202, 151–163 (2023). https://doi.org/10.1007/s00442-023-05382-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-023-05382-1