Abstract
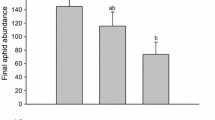
Predators and pathogens often regulate the population dynamics of their prey or hosts. When species interact with both their predators and their pathogens, understanding each interaction in isolation may not capture the system’s dynamics. For instance, predators can influence pathogen transmission via consumptive effects, such as feeding on infected prey, or non-consumptive effects, such as changing the prey’s susceptibility to infection. A prey species' infection status can, in turn, influence predator’s choice of prey and have negative fitness consequences for the predator. To test how intraguild predation (IGP), when predator and pathogen share the same prey/host, affects pathogen transmission, predator preference, and predator fitness, we conducted a series of experiments using a crop pest (Pseudoplusia includens), a generalist predator (Podisus maculiventris), and a generalist pathogen (Autographa californica multicapsid nuclear polyhedrovirus, AcMNPV). Using a field experiment, we quantified the effects of consumptive and non-consumptive predators on pathogen transmission. We found that a number of models provided similar fits to the data. These models included null models showing no effects of predation and models that included a predation effect. We also found that predators consumed infected prey more often when choosing between live infected or live healthy prey. Infected prey also reduced predator fitness. Developmental times of predators fed infected prey increased by 20% and longevity decreased by 45%, compared with those that consumed an equivalent number of non-infected prey. While this research shows an effect of the pathogen on intraguild predator fitness, we found no support that predators affected pathogen transmission.


Similar content being viewed by others
References
Abbas MST, Boucias DG (1984) Interaction between nuclear polhyedrosis virus-infected Anticarsia gematalis (Lepidoptera: Noctuidae) larvae and predators Podisus maculiventris (Say) (Hemiptera: Pentatomidae). Environ Entomol 13:599–602
Anderson RM, May RM (1980) Infectious diseases and population cycles of forest insects. Science 210:658–661
Arim M, Marquet PA (2004) Intraguild predation: a widespread interaction related to species biology. Ecol Lett 7:557–564
Bate AM, Hilker FM (2014) Disease in group-defending prey can benefit predators. Theor Ecol 7:87–100
Beach MR, Todd JW (1988) Foliage consumption and developmental parameters of the soybean looper and the velvetbean caterpillar (Lepidoptera: Noctuidae) reared on susceptible and resistant soybean genotypes. J Econ Entomol 81:310–316
Bell HA, Down RE, Kirkbride-Smith AE, Edwards JP (2004) Effect of microsporidian infection in Lacanobia oleracea (Lep., Noctuidae) on prey selection and consumption by the spined soldier bug Podisus maculiventris (Het., Pentatomidae). J Appl Entomol 128:548–553
Bernardi O, Malvestiti GS, Dourado PM, Oliveira WS, Martinelli S, Berger GU, Head GP, Omoto C (2012) Assessment of the high-dose concept and level of control provided by MON 87701 × MON 89788 soybean against Anticarsia gemmatalis and Pseudoplusia includens (Lepidoptera: Noctuidae) in Brazil. Pest Manag Sci 68:1083–1091
Bolker B, R Development Core Team (2017). bbmle: Tools for general maximum likelihood estimation. R package version 1.0.20. https://CRAN.R-project.org/package=bbmle. Accessed 15 Mar 2020
Bolker BM (2008) Ecological models and data in R. Princeton University Press, Princeton
Borer ET, Briggs CJ, Holt RD (2007) Predators, parasitoids, and pathogens: a cross-cutting examination of intraguild predation theory. Ecology 88:2681–2688
Burnham K, Anderson D (2003) Model selection and multi-model inference. Springer, New York
Cáceres CE, Knight CJ, Hall SR (2009) Predator-spreaders: predation can enhance parasite success in a planktonic host-parasite system. Ecology 90:2850–2858
Cirtwill AR, Stouffer DB (2015) Concomitant predation on parasites is highly variable but constrains the ways in which parasites contribute to food web structure. J Anim Ecol 84:734–744
Coors A, De Meester L (2011) Fitness and virulence of a bacterial endoparasite in an environmentally stressed crustacean host. Parasitology 138:122–131
D’Amico V, Elkinton JS, Podgwaite JD, Buonaccorsi J, Dwyer G (2005) Pathogen clumping: an explanation for non-linear transmission of an insect virus. Ecol Entomol 30:383–390
de Nardo EAB, Maia AHN, Watanabe MA (2001) Effect of a formulation of Anticarsia gemmatalis (Lepidoptera: Noctuidae) nuclear polyhedrosis virus on the predator Podisus nigrispinus (Heteroptera: Pentatomidae: Asopinae), using the fertility life table parameters. Environ Entomol 30:1164–1173
Down RE, Bell HA, Matthews HJ, Kirkbride-Smith AE, Edwards JP (2004) Dissemination of the biocontrol agent Vairimorpha necatrix by the spined soldier bug, Podisus maculiventris. Entomol Exp Appl 110:103–114
Duffy MA, Housley JM, Penczykowski RM, Caceres CE, Hall SR (2011) Unhealthy herds: indirect effects of predators enhance two drivers of disease spread. Funct Ecol 25:945–953
Dwyer G, Elkinton JS, Buonaccorsi JP (1997) Host heterogeneity in susceptibility and disease dynamics: tests of a mathematical model. Am Nat 150:685–707
Elderd BD (2018) Modeling insect epizootics and their population-level consequences. In: Hajek A, Shapiro-Ilan D (eds) Ecology of invertebrate diseases. Wiley, Hoboken
Elderd BD (2013) Developing models of disease transmission: insights from ecological studies of insects and their baculoviruses. PLoS Pathog 9:e1003372
Elderd BD, Rehill BJ, Haynes KJ, Dwyer G (2013) Induced plant defenses, host–pathogen interactions, and forest insect outbreaks. Proc Natl Acad Sci USA 110:14978–14983
Elderd BD, Reilly JR (2014) Warmer temperatures increase disease transmission and outbreak intensity in a host–pathogen system. J Anim Ecol 83:838–849
Finke DL (2012) Contrasting the consumptive and non-consumptive cascading effects of natural enemies on vector-borne pathogens. Entomol Exp Appl 144:45–55
Flick AJ, Acevedo MA, Elderd BD (2016) The negative effects of pathogen-infected prey on predators: a meta-analysis. Oikos 125:1554–1560
Goodman CL, El Sayed GN, Mcintosh AH, Grasela JJ, Stiles B (2001) Establishment and characterization of insect cell lines from 10 lepidopteran species. Vitro Cell Dev Biol Anim 37:367–373
Gottelli NJ, Ellison AM (2004) A primer of ecological statistics. Sinauer Associates, Inc., Sunderland
Granados RR, Lawler KA (1981) In vivo pathway of Autographica californica baculovirus invasion and infection. Virology 176:297–308
Hermann SL, Thaler JS (2014) Prey perception of predation risk: volatile chemical cues mediate non-consumptive effects of a predator on a herbivorous insect. Oecologia 176:669–676
Herzog DC (1980) Sampling soybean looper on soybean. Sampling methods in soybean entomology. Springer, New York, pp 141–168
Hilker FM, Schmitz K (2008) Disease-induced stabilization of predator–prey oscillations. J Theor Biol 255:299–306
Hochberg ME (1991) Non-linear transmission rates and the dynamics of infectious disease. J Theor Biol 153:301–321
Holt RD, Polis GA (1997) A theoretical framework for intraguild predation. Am Nat 145:745–764
Inouye BD (2001) Surface experimental designs for investigating interspecific competition. Ecology 82:2696–2706
Johnson PT, Dobson A, Lafferty KD, Marcogliese DJ, Memmott J, Orlofske SA, Poulin R, Thieltges DW (2010) When parasites become prey: ecological and epidemiological significance of eating parasites. Trends Ecol Evol 25:362–371
Katsuma S, Koyano Y, Kang W, Kokusho R, Kamita SG, Shimada T (2012) The baculovirus uses a captured host phosphatase to induce enhanced locomotory activity in host caterpillars. PLoS Pathog 8:e1002644
Koelle K, Pascual M (2004) Disentangling extrinsic from intrinsic factors in disease dynamics: a nonlinear time series approach with an application to cholera. Am Nat 163:901–913
Krebs CJ, Boonstra R, Boutin S, Sinclair AR (2001) What Drives the 10-year Cycle of Snowshoe Hares? The ten-year cycle of snowshoe hares—one of the most striking features of the boreal forest—is a product of the interaction between predation and food supplies, as large-scale experiments in the Yukon have demonstrated. Bioscience 51:25–35
Kunimi Y, Fuxa J, Richter A (1997) Survival times and lethal doses for wild and recombinant Autographa californica Nuclear Polyhedrosis Viruses in different instars of Pseudoplusia includens. Biol Control 9:129–135
Lafferty KD (1992) Foraging on prey that are modified by parasites. Am Nat 140:854–867
Lee Y, Fuxa JR (2000) Ingestion and defecation of recombinant and wild-type nucleopolyhedroviruses by scavenging and predatory arthropods. Environ Entomol 29:950–957
Marston NL, Schmidt GT, Biever KD, Dickerson WA (1978) Reaction of five species of soybean caterpillars to attack, by the predator, Podisus maculiventris. Environ Entomol 7:53–56
McCauley SJ, Rowe L, Fortin MJ (2011) The deadly effects of “nonlethal” predators. Ecology 92:2043–2048
McCullagh P, Nelder JA (1989) Generalized linear models. Chapman & Hall, Boca Raton
Orrock JL, Grabowski JH, Pantel JH, Peacor SD, Peckarsky BL, Sih A, Werner EE (2008) Consumptive and nonconsumptive effects of predators on metacommunities of competing prey. Ecology 89:2426–2435
O’Neil RJ (1995) Know your friends: spined soldier bug. Midwest Biol Control News 2(8):3
Packer C, Holt RD, Hudson PJ, Lafferty KD, Dobson AP (2003) Keeping the herds healthy and alert: implications of predator control for infectious disease. Ecol Lett 6:797–802
Polis GA, Myers CA, Holt RD (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Annu Rev Ecol Syst 20:297–330
Preisser EL, Bolnick DI, Benard MF (2005) Scared to death? The effects of intimidation and consumption in predator–prey interactions. Ecology 86:501–509
R Core Team (2013) R: a language and environment for statistical computing. Version 3.1. 2. R Foundation for Statistical Computing, Vienna
Raffel TR, Hoverman JT, Halstead NT, Michel PJ, Rohr JR (2010) Parasitism in a community context: trait-mediated interactions with competition and predation. Ecology 91:1900–1907
Ramirez RA, Snyder WE (2009) Scared sick? Predator–pathogen facilitation enhances exploitation of a shared resource. Ecology 90:2832–2839
Reed JM, Levine SH (2005) A model for behavioral regulation of meta population dynamics. Ecol Model 183:411–423
Richards SA (2008) Dealing with overdispersed count data in applied ecology. J Appl Ecol 45:218–227
Rohr JR, Civitello DJ, Crumrine PW, Halstead NT, Miller AD, Schotthoefer AM, Stenoien C, Johnson LB, Beasley VR (2015) Predator diversity, intraguild predation, and indirect effects drive parasite transmission. Proc Natl Acad Sci USA 112:3008–3013
Rosenheim JA et al (1995) Intraguild predation among biological-control agents: theory and evidence. Biol Control 5:303–335
Roy M, Holt RD (2008) Effects of predation on host–pathogen dynamics in SIR models. Theor Popul Biol 73:319–331
Silva RBD, Corrêa AS, Della Lucia TMC, Pereira AIA, Cruz I, Zanuncio JC (2012) Does the aggressiveness of the prey modify the attack behavior of the predator Supputius cincticeps (Stål)(Hemiptera, Pentatomidae)? Revista Brasileira de Entomologia 56:244–248
Smith K, Acevedo-Whitehouse K, Pedersen A (2009) The role of infectious diseases in biological conservation. Anim Conserv 12:1–12
Smith RH, Freeman B, Foshee W (1994) Soybean loopers: late season foliage feeders on cotton. Alabama Cooperative Extension System, Alabama A&M University and Auburn University.
Strauss AT, Shocket MS, Civitello DJ, Hite JL, Penczykowski RM, Duffy MA, Cáceres CE, Hall SR (2016) Habitat, predators, and hosts regulate disease in Daphnia through direct and indirect pathways. Ecol Monogr 86:393–411
Thaler JS, McArt SH, Kaplan I (2012) Compensatory mechanisms for ameliorating the fundamental trade-off between predator avoidance and foraging. Proc Natl Acad Sci USA 109:12075–12080
Thieltges DW, Amundsen PA, Hechinger RF, Johnson PT, Lafferty KD, Mouritsen KN, Preston DL, Reise K, Zander CD, Poulin R (2013) Parasites as prey in aquatic food webs: implications for predator infection and parasite transmission. Oikos 122:1473–1482
Thiemann GW, Wassersug RJ (2000) Patterns and consequences of behavioural responses to predators and parasites in Rana tadpoles. Biol J Lin Soc 71:513–528
Thomas F, Adamo S, Moore J (2005) Parasitic manipulation: where are we and where should we go? Behav Proc 68:185–199
Thomas MB, Arthurs SP, Watson EL (2006) Trophic and guild interactions and the influence of multiple species on disease. Trophic and guild in biological interactions control. Springer, New York, pp 101–122
van Houte S, Ros VI, Mastenbroek TG, Vendrig NJ, Hoover K, Spitzen J, van Oers MM (2012) Protein tyrosine phosphatase-induced hyperactivity is a conserved strategy of a subset of baculoviruses to manipulate lepidopteran host behavior. PLoS ONE 7:e46933
Vance-Chalcraft HD et al (2007) The influence of intraguild predation on prey suppression and prey release: a meta-analysis. Ecology 88:2689–2696
Wittmeyer J, Coudron T, Adams T (2001) Ovarian development, fertility and fecundity in Podisus maculiventris Say (Heteroptera: Pentatomidae): an analysis of the impact of nymphal, adult, male and female nutritional source on reproduction. Invertebr Reprod Dev 39:9–20
Yang L (2000) Effects of body size and plant structure on the movement ability of a predaceous stink bug, Podisus maculiventris (Heteroptera: Pentatomidae). Oecologia 125:85–90
Young S, Yearian W (1986) Movement of a nuclear polyhedrosis virus from soil to soybean and transmission in Anticarsia gemmatalis (Hübner) (Lepidoptera: Noctuidae) populations on soybean. Environ Entomol 15:573–580
Acknowledgements
The extensiveness of this study would not have been possible without field assistance, insect rearing assistance, and lab assistance from Warwick Allen, Matthew Faldyn, Tatum Flick, Miriam Sadiq, James Smith, Chad Finkenbinder, Forrest Dillemuth, Ben Van Allen, McCayn McDaniels, David Clark, Scott Kosiba, and Olivia Barry. The authors also thank Roland Brandl, Sven Bacher, Alex Strauss, Jim Cronin, Kyle Harms, and two anonymous reviewers for valuable insights. This work was supported by the Louisiana Environmental Education Commission (Grant #43010), a Microryza (now Experiment) Grant (https://doi.org/10.18258/0812), and a Louisiana State University BioGrads research award. BDE was supported by NSF-DEB-1316334.
Author information
Authors and Affiliations
Contributions
AJF designed the experiments, conducted field and lab studies, and wrote the manuscript. TAC provided insects, advised in insect rearing and experimental use, and made substantial edits to the manuscript. BDE advised in designing the experiment, assisted with field work, and advised writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sven Bacher.
In support of being a highlighted student project, we provide a unique look at intraguild predation focusing on predators, prey, and prey pathogens. Predators preferred infected prey, infected prey reduced predator fitness. However, we found no support that predators changed pathogen transmission dynamics. Our combination of field and lab experiments to examine both sides of these interactions is unique in these communities and our results point to interesting scenarios playing out in natural and agricultural systems. AJF designed the experiments, conducted field and lab studies, and wrote the manuscript. TAC provided insects, advised in insect rearing and experimental use, and made substantial edits to the manuscript. BDE advised in designing the experiment, assisted with field work, and advised writing of the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Flick, A.J., Coudron, T.A. & Elderd, B.D. Intraguild predation decreases predator fitness with potentially varying effects on pathogen transmission in a herbivore host. Oecologia 193, 789–799 (2020). https://doi.org/10.1007/s00442-020-04665-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04665-1