Abstract
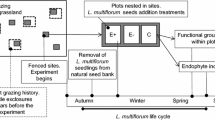
When symbionts are inherited by offspring, they can have substantial ecological and evolutionary consequences because they occur in all host life stages. Although natural frequencies of inherited symbionts are commonly <100 %, few studies investigate the ecological drivers of variation in symbiont prevalence. In plants, inherited fungal endophytes can improve resistance to herbivory, growth under drought, and competitive ability. We evaluated whether native ungulate herbivory increased the prevalence of a fungal endophyte in the common, native bunchgrass, Festuca campestris (rough fescue, Poaceae). We used large-scale (1 ha) and long-term (7–10 year) fencing treatments to exclude native ungulates and recorded shifts in endophyte prevalence at the scale of plant populations and for individual plants. We characterized the fungal endophyte in F. campestris, Epichloë species FcaTG-1 (F. campestris taxonomic group 1) for the first time. Under ungulate exclusion, endophyte prevalence was 19 % lower in plant populations, 25 % lower within plant individuals, and 39 % lower in offspring (seeds) than in ungulate-exposed controls. Population-level endophyte frequencies were also negatively correlated with soil moisture across geographic sites. Observations of high within-plant variability in symbiont prevalence are novel for the Epichloë species, and contribute to a small, but growing, literature that documents phenotypic plasticity in plant-endophyte symbiota. Altogether, we show that native ungulates can be an important driver of symbiont prevalence in native plant populations, even in the absence of evidence for direct mechanisms of mammal deterrence. Understanding the ecological controls on symbiont prevalence could help to predict future shifts in grasslands that are dominated by Epichloë host plants.




Similar content being viewed by others
References
Afkhami ME, Rudgers JA (2008) Symbiosis lost: imperfect vertical transmission of fungal endophytes in grasses. Am Nat 172:405–416. doi:10.1086/589893
Afkhami ME, McIntyre PJ, Strauss SY (2014) Mutualist-mediated effects on species’ range limits across large geographic scales. Ecol Lett 17:1265–1273. doi:10.1111/ele.12332
Anisimova M, Gascuel O (2006) Approximate likelihood ratio test for branches: a fast, accurate and powerful alternative. Syst Biol 55:539–552. doi:10.1080/10635150600755453
Bacon CW, White JF Jr (1994) Stains, media, and procedures for analyzing endophytes. In: Bacon CW, White JF Jr (eds) Biotechnology of endophytic fungi of grasses. CRC, Boca Raton, pp 47–56
Barkworth ME, Capels KM, Long S, Anderton LK, Piep MB (eds) (2007) Flora of north America volume 24: north of Mexico: Magnoliophyta: Commelinidae (in part): Poaceae, part 1. Oxford University Press, New York
Bazely DR, Vicari M, Emmerich S, Filip L, Lin D, Inman A (1997) Interactions between herbivores and endophyte-infected Festuca rubra from the Scottish islands of St. Kilda, Benbecula and Rum. J Appl Ecol 34:847–860
Belesky DP, Bacon CW (2009) Tall fescue and associated mutualistic toxic fungal endophytes in agroecosystems. Toxin Rev 28:102–117. doi:10.1080/15569540903082143
Bibian A, Rudgers JA, Miller TEX (accepted) The role of host demographic storage in the ecological dynamics of heritable symbionts. Am Nat
Charlton ND, Craven KD, Afkhami ME, Hall BA, Ghimire SR, Young CA (2014) Interspecific hybridization and bioactive alkaloid variation increases diversity in endophytic Epichloë species of Bromus laevipes. FEMS Microb Ecol 90:276–289. doi:10.1111/1574-6941.12393
Cheplick GP (2007) Costs of fungal endophyte infection in Lolium perenne genotypes from Eurasia and North Africa under extreme resource limitation. Environ Exp Bot 60:202–210
Cheplick GP, Faeth SH (2009) Ecology and evolution of grass-endophyte symbiosis. Oxford University Press, Oxford
Chevenet F, Brun C, Baenuls AL, Jacq B, Christen R (2006) TREEDYN: towards dynamic graphics and annotations for analyses of trees. BMC Bioinform 7:439
Clay K, Holah J, Rudgers JA (2005) Herbivores cause a rapid increase in hereditary symbiosis and alter plant community composition. Proc Natl Acad Sci USA 102:12465–12470
Dereeper A et al (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:W465–W469. doi:10.1093/nar/gkn180
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Elmi AA, West CP (1995) Endophyte infection effects on stomatal conductance, osmotic adjustment and drought recovery of tall fescue. New Phytol 131:61–67
Emery SM, Thompson D, Rudgers JA (2010) Variation in endophyte symbiosis, herbivory and drought tolerance of Ammophila breviligulata populations in the great lakes region. Am Midl Nat 163:186–196
Garcia Parisi PA, Casas C, Gundel PE, Omacini M (2012) Consequences of grazing on the vertical transmission of a fungal Neotyphodium symbiont in an annual grass population. Austral Ecol 37:620–628. doi:10.1111/j.1442-9993.2011.02325.x
Gass TM, Binkley D (2011) Soil nutrient losses in an altered ecosystem are associated with native ungulate grazing. J Appl Ecol 48:952–960. doi:10.1111/j.1365-2664.2011.01996.x
Goff BM, Aiken GE, Witt WW, Sleugh BB, Burch PL (2012) Steer consumption and ergovaline recovery from in vitro digested residues of tall fescue seedheads. Crop Sci 52:1437–1440. doi:10.2135/cropsci2011.07.0378
Guindon S, Gascuel O (2003) A simple, fast and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704. doi:10.1080/10635150390235520
Hart SC, Stark JM, Davidson EA, Firestone MK (1994) Nitrogen mineralization, immobilization, and nitrification. In: Weaver RW, Angle S, Bottomley P, Bezdicek D, Smith S, Tabatabai A, Wollum A (eds), Methods of Soil Analysis. Part 2. Microbiological and Biochemical Properties, Soil Science Society of America, Madison, WI, pp 985–1018
Hobbs NT (1996) Modification of ecosystems by ungulates. J Wildl Manage 60:695–713. doi:10.2307/3802368
Iannone LJ, Cabral D, Schardl CL, Rossi MS (2009) Phylogenetic divergence, morphological and physiological differences distinguish a new Neotyphodium endophyte species in the grass Bromus auleticus from South America. Mycologia 101:340–350. doi:10.3852/08-156
Iannone LJ, Irisarri JGN, Mc Cargo PD, Pérez LI, Gundel PE (2015) Occurrence of Epichloë fungal endophytes in the sheep-preferred grass Hordeum comosum from Patagonia. J Arid Environ 115:19–26
Jaenike J, Unckless R, Cockburn SN, Boelio LM, Perlman SJ (2010) Adaptation via symbiosis: recent spread of a Drosophila defensive symbiont. Science 329:212–215. doi:10.1126/science.1188235
Karban R (2011) The ecology and evolution of induced resistance against herbivores. Funct Ecol 25:339–347. doi:10.1111/j.1365-2435.2010.01789.x
Koh S, Hik DS (2007) Herbivory mediates grass–endophyte relationships. Ecology 88:2752–2757
Kucht S, Grob J, Hussein Y, Grothe T, Keller U, Basar S, Konig WA, Steiner U, Leister E (2004) Elimination of ergoline alkaloids following treatment of Ipomoea asarifolia (Convolvulaceae) with fungicides. Planta 219:619–625
Leuchtmann A, Bacon CW, Schardl CL, White JF Jr, Tadych M (2014) Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 106:202–215. doi:10.3852/106.2.202
Lewis GC, Ravel C, Naffaa W, Astier C, Charmet G (1997) Occurrence of Acremonium endophytes in wild populations of Lolium spp. in European countries and a relationship between level of infection and climate in France. Ann Appl Biol 130:227–238
Mack KML, Rudgers JA (2008) Balancing multiple mutualists: asymmetric interactions among plants, arbuscular mycorrhizal fungi, and fungal endophytes. Oikos 117:310–320. doi:10.1111/j.2007.0030-1299.15973.x
Malinowski DP, Belesky DP (2000) Adaptations of endophyte-infected cool-season grasses to environmental stresses: mechanisms of drought and mineral stress tolerance. Crop Sci 40:923–940
Margulis L (1996) Archaeal–eubacterial mergers in the origin of Eukarya: phylogenetic classification of life. Proc Natl Acad Sci USA 93:1071–1076
Maron JL, Pearson DE, Potter T, Ortega YK (2012) Seed size and provenance mediate the joint effects of disturbance and seed predation on community assembly. J Ecol 100:1492–1500. doi:10.1111/j.1365-2745.2012.02027.x
McInenly LE, Merrill EH, Cahill JF, Juma NG (2010) Festuca campestris alters root morphology and growth in response to simulated grazing and nitrogen form. Funct Ecol 24:283–292. doi:10.1111/j.1365-2435.2009.01642.x
McNaughton SJ, Banyikwa FF, McNaughton MM (1997) Promotion of the cycling of diet-enhancing nutrients by African grazers. Science 278:1798–1800. doi:10.1126/science.278.5344.1798
Moran NA, McCutcheon JP, Nakabachi A (2008) Genomics and evolution of heritable bacterial symbionts. Annu Rev Genet 42:165–190. doi:10.1146/annurev.genet.41.110306.130119
Nagabhyru P, Dinkins RD, Wood CL, Bacon CW, Schardl CL (2013) Tall fescue endophyte effects on tolerance to water-deficit stress. BMC Plant Biol 13:127. doi:10.1186/1471-2229-13-127
Oliver KM, Campos J, Moran NA, Hunter MS (2008) Population dynamics of defensive symbionts in aphids. Proc R Soc B Biol Sci 275:293–299
Omacini M, Semmartin M, Perez LI, Gundel PE (2012) Grass–endophyte symbiosis: a neglected aboveground interaction with multiple belowground consequences. Appl Soil Ecol 61:273–279. doi:10.1016/j.apsoil.2011.10.012
Orians CM, Jones CG (2001) Plants as resource mosaics: a functional model for predicting patterns of within-plant resource heterogeneity to consumers based on vascular architecture and local environmental variability. J Chem Ecol 94:493–504. doi:10.1034/j.1600-0706.2001.940311.x
Rasmussen S, Parsons AJ, Bassett S, Christensen MJ, Hume DE, Johnson LJ, Johnson RD, Simpson WR, Stacke C, Voisey CR, Xue H, Newman JA (2007) High nitrogen supply and carbohydrate content reduce fungal endophyte and alkaloid concentration in Lolium perenne. New Phytol 173:787–797
Ren A, Wei M, Yin L, Wu L, Zhou Y, Li X, Gao Y (2014) Benefits of a fungal endophyte in Leymus chinensis depend more on water than on nutrient availability. Environ Exp Bot 108:71–78. doi:10.1016/j.envexpbot.2013.11.019
Rodriguez RJ, White JF, Arnold AE, Redman RS (2009) Fungal endophytes: diversity and functional roles. New Phytol 182:314–330. doi:10.1111/j.1469-8137.2009.02773.x
Rodriguez-Saona C, Thaler JS (2005) Herbivore-induced responses and patch heterogeneity affect abundance of arthropods on plants. Ecol Entomol 30:156–163. doi:10.1111/j.0307-6946.2005.00682.x
Rudgers JA, Holah J, Orr SP, Clay K (2007) Forest succession suppressed by an introduced plant-fungal symbiosis. Ecology 88:18–25
Rudgers JA, Miller TEX, Ziegler SM, Craven KD (2012) There are many ways to be a mutualist: endophytic fungus reduces plant survival but increases population growth. Ecology 93:565–574
Sachs JL, Simms EL (2006) Pathways to mutualism breakdown. Trends Ecol Evol 21:585–592. doi:10.1016/j.tree.2006.06.018
Saikkonen K, Helander M, Faeth SH, Schulthess F, Wilson D (1999) Endophyte–grass–herbivore interactions: the case of Neotyphodium endophytes in Arizona fescue populations. Oecologia 121:411–420
SAS Institute, Inc. (2012) SAS version 9.3. SAS Institute, Cary, NC
Schardl CL, Craven KD, Speakman S, Stromberg A, Lindstrom A, Yoshida R (2008) A novel test for host-symbiont codivergence indicates ancient origin of fungal endophytes in grasses. Syst Biol 57:483–498. doi:10.1080/10635150802172184
Schardl CL, Young CA, Hesse U, Amyotte SG, Andreeva K, Calie PJ, Fleetwood DJ, Haws DC, Moore N, Oeser B, Panaccione DG, Schweri KK, Voisey CR, Farman ML, Jaromczyk JW, Roe BA, O’Sullivan DM, Scott B, Tudzynski P, An Z, Arnaoudova EG, Bullock CT, Charlton ND, Chen L, Cox M, Dinkins RD, Florea S, Glenn AE, Gordon A, Gueldener U, Harris DR, Hollin W, Jaromczyk J, Johnson RD, Khan AK, Leistner E, Leuchtmann A, Li C, Liu J, Liu J, Liu M, Mace W, Machado C, Nagabhyru P, Pan J, Schmid J, Sugawara K, Steiner U, Takach JE, Tanaka E, Webb JS, Wilson EV, Wiseman JL, Yoshida R, Zeng Z (2013) Plant-symbiotic fungi as chemical engineers: multi-genome analysis of the Clavicipitaceae reveals dynamics of alkaloid loci. PLoS Genet 9:e1003323. doi:10.1371/journal.pgen.1003323
Schardl CL, Young CA, Moore N, Krom N, Dupont P-Y, Pan J, Florea S, Webb JS, Jaromczyk J, Jaromczyk JW, Cox MP, Farman ML (2014) Genomes of plant-associated Clavicipitaceae. Adv Bot Res 70:291–327
Semmartin M, Omacini M, Gundel PE, Hernández-Agramonte IM (2015) Broad-scale variation of fungal-endophyte incidence in temperate grasses. J Ecol 103:184–190
Strauss SY, Rudgers JA, Lau JA, Irwin RE (2002) Direct and ecological costs of resistance to herbivory. Trends Ecol Evol 17:278–285
Takach JE, Young CA (2014) Alkaloid genotype diversity of tall fescue endophytes. Crop Sci 54:667–678. doi:10.2135/cropsci2013.06.0423
Tanentzap AJ, Vicari M, Bazely DR (2014) Ungulate saliva inhibits a grass–endophyte mutualism. Biol Lett 10:20140460. doi:10.1098/rsbl.2014.0460
Thrower LB, Lewis DH (1973) Uptake of sugars by Epichloë typhina (Pers. Ex Fr.) Tul. in culture and from its host, Agrostis stolonifera L. New Phytol 72:501–508
Valdez Barillas JR, Paschke MW, Ralphs MH, Child RD (2007) White locoweed toxicity is facilitated by a fungal endophyte and nitrogen-fixing bacteria. Ecology 88:1850–1856
Werren JH, Baldo L, Clark ME (2008) Wolbachia: master manipulators of invertebrate biology. Nat Rev Microbiol 6:741–751. doi:10.1038/nrmicro1969
Acknowledgments
J. M. and D. E. P. were supported by the US National Science Foundation DEB-0614406. J. A. R. was supported by the US National Science Foundation DEB-1354972 and 1145588. The authors have no other conflicts of interest to declare. Thanks to Beth Haley and Ben Goodheart for field and laboratory assistance. This article does not contain any studies with human participants or animals performed by any of the authors.
Author contribution statement
J. L. M. and D. E. P. conceived, designed, and performed the experimental manipulations. R. A. F., E. O., and J. A. R. collected the endophyte data. J. A. R. analyzed the data. C. A. Y. and N. D. C. characterized the endophyte identity. J. A. R. led the writing, with substantial contributions from co-authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Maria J. Pozo.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rudgers, J.A., Fletcher, R.A., Olivas, E. et al. Long-term ungulate exclusion reduces fungal symbiont prevalence in native grasslands. Oecologia 181, 1151–1161 (2016). https://doi.org/10.1007/s00442-016-3620-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3620-7