Abstract
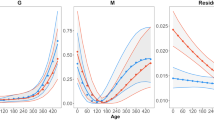
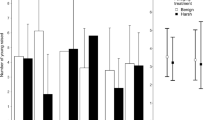
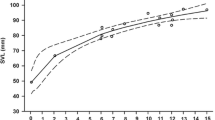
Food availability is a major environmental factor that can influence life history within and across generations through direct effects on individual quality and indirect effects on the intensity of intra- and intercohort competition. Here, we investigated in yearling and adult common lizards (Zootoca vivipara) the immediate and delayed life-history effects of a prolonged food deprivation in the laboratory. We generated groups of fully fed or food-deprived yearlings and adults at the end of one breeding season. These lizards were released in 16 outdoor enclosures together with yearlings and adults from the same food treatment and with food-deprived or fully fed juveniles, creating four types of experimental populations. Experimental populations were then monitored during 2 years, which revealed complex effects of food on life-history trajectories. Food availability had immediate direct effects on morphology and delayed direct effects on immunocompetence and female body condition at winter emergence. Also, male annual survival rate and female growth rate and body size were affected by an interaction between direct effects of food availability and indirect effects on asymmetric competition with juveniles. Reproductive outputs were insensitive to past food availability, suggesting that female common lizards do not solely rely on stored energy to fuel reproduction. Finally, food conditions had socially-mediated intergenerational effects on early growth and survival of offspring through their effects on the intensity of competition. This study highlights the importance of social interactions among cohorts for life-history trajectories and population dynamics in stage-structured populations.





Similar content being viewed by others
References
Andrews RM (1982) Patterns of growth in reptiles. In: Gans G, Pough FH (eds) Biology of the reptilia, vol 13. Academic, New York, pp 273–320
Aubret F, Bonnet X, Shine R, Lourdais O (2002) Fat is sexy for females but not males: the influence of body reserves on reproduction in snakes (Vipera aspis). Horm Behav 42:135–147
Avery RA (1971) Estimates of food consumption by the lizard Lacerta vivipara Jacquin. J Anim Ecol 40:351–365
Avery RA (1975) Clutch size and reproductive effort in the lizard Lacerta vivipara Jacquin. Oecologia 19:165–170
Bateson P et al (2004) Developmental plasticity and human health. Nature 430:419–421
Bauwens D, Verheyen RF (1985) The timing of reproduction in the lizards Lacerta vivipara: differences between individual females. J Herpetol 19:353–364
Bauwens D, van Damme R, Verheyen RF (1989) Synchronization of spring molting behaviour with the onset of mating behavior in male lizards, Lacerta vivipara. J Herpetol 23:89–91
Beckerman A, Benton TG, Lapsley CT, Koesters N (2003) Talkin’ ‘bout my generation: environmental variability and cohort effects. Am Nat 162:754–767
Birkhead TR, Fletcher F, Pellatt EJ (1999) Nestling diet, secondary sexual traits and fitness in the zebra finch. Proc R Soc Lond B 266:385–390
Bolker BM et al (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135
Brommer JE, Karell P, Pietiainen H (2004) Supplementary fed Ural owls increase their reproductive output with a one year time lag. Oecologia 139:354–358
Bystrom P, Andersson J (2005) Size-dependent foraging capacities and intercohort competition in an ontogenetic omnivore (Arctic char). Oikos 110:523–536
Claessen D, de Roos AM, Persson L (2000) Dwarfs and giants: Cannibalism and competition in size-structured populations. Am Nat 155:219–237
de Roos AM, Persson L, McCauley E (2003) The influence of size-dependent life-history traits on the structure and dynamics of populations and communities. Ecol Lett 6:473–487
Drent RH, Daan S (1980) The prudent parent–energetic adjustments in avian breeding. Ardea 68:225–252
Fitze PS, Le Galliard JF (2008) Operational sex ratio, sexual conflict and the intensity of sexual selection. Ecol Lett 11:432–439
Fitze PS, Cote J, Martinez-Rica JP, Clobert J (2008) Determinants of male fitness: disentangling intra- and inter-sexual selection. J Evol Biol 21:246–255
González-Suárez M, Mugabo M, Decencière B, Perret S, Claessen D, Le Galliard JF (2011) Disentangling the effects of predator body size and prey density on prey consumption in a lizard. Funct Ecol 25:158–165
Grafen A (1988) On the uses of data on lifetime reproductive success. In: Clutton-Brock TH (ed) Reproductive success. University of Chicago Press, Chicago, pp 454–471
Heino M, Kaitala V (1999) Evolution of resource allocation between growth and reproduction in animals with indeterminate growth. J Evol Biol 12:423–429
Kooijman SALM (2000) Dynamic energy and mass budgets in biological systems. Cambridge University Press, Cambridge
Koskela E, Jonsson P, Hartikainen T, Mappes T (1998) Limitation of reproductive success by food availability and litter size in the bank vole, Clethrionomys glareolus. Proc R Soc Lond B 265:1129–1134
Kratochvil L, Fokt M, Rehak I, Frynta D (2003) Misinterpretation of character scaling: a tale of sexual dimorphism in body shape of common lizards. Can J Zool Rev Can Zool 81:1112–1117
Kubicka L, Kratochvil L (2009) First grow, then breed and finally get fat: hierarchical allocation to life-history traits in a lizard with invariant clutch size. Funct Ecol 23:595–601
Le Galliard J-F, Ferrière R, Clobert J (2005) Juvenile growth and survival under dietary restriction: are males and females equal? Oikos 111:368–376
Lecomte J, Clobert J, Massot M, Barbault R (1994) Spatial and behavioural consequences of a density manipulation in the common lizard. Ecoscience 1:300–310
Lummaa V, Clutton-Brock T (2002) Early development, survival and reproduction in humans. Trends Ecol Evol 17:141–147
Marquis O, Massot M, Le Galliard J-F (2008) Intergenerational effects of climate generate cohort variation in lizard reproductive performance. Ecology 89:2575–2583
Martin LB, Navara KJ, Weil ZM, Nelson RJ (2007) Immunological memory is compromised by food restriction in deer mice Peromyscus maniculatus. Am J Physiol Regul Integr Comp Physiol 292:R316–R320
Massot M, Clobert J (1995) Influence of maternal food availability on offspring dispersal. Behav Ecol Sociobiol 37:413–418
Massot M, Clobert J, Pilorge T, Lecomte J, Barbault R (1992) Density dependence in the common lizard: demographic consequences of a density manipulation. Ecology 73:1742–1756
Metcalfe NB, Monaghan P (2001) Compensation for a bad start: grow now, pay later? Trends Ecol Evol 16:254–260
Mugabo M, Marquis O, Perret S, Le Galliard JF (2010) Immediate and delayed life history effects caused by food deprivation early in life in a short-lived lizard. J Evol Biol 23:1886–1898
Nagy KA (1983) Ecological energetics. In: Huey RB, Pianka ER, Schoener TW (eds) Lizard ecology: studies of a model organism. Harvard University Press, Cambridge, pp 24–54
Olsson M, Shine R (1997) The limits to reproductive output: offspring size versus number in the sand lizard (Lacerta agilis). Am Nat 149:179–188
Pinheiro JC, Bates DM (2000) Mixed-Effects Models in S and S-PLUS. Springer, New York
Plaistow SJ, Lapsley CT, Benton TG (2006) Context-dependent intergenerational effects: the interaction between past and present environments and its effect on population dynamics. Am Nat 167:206–215
Saino N, Calza S, Ninni P, Moller AP (1999) Barn swallows trade survival against offspring condition and immunocompetence. J Anim Ecol 68:999–1009
Schwarzkopf L (1994) Measuring trade-offs: a review of studies of cost of reproduction in lizards. In: Vitt LJ, Pianka ER (eds) Lizard ecology: historical and experimental perspectives. Princeton University Press, Princeton, pp 7–29
Tschirren B, Rutstein AN, Postma E, Mariette M, Griffith SC (2009) Short- and long-term consequences of early developmental conditions: a case study on wild and domesticated zebra finches. J Evol Biol 22:387–395
van de Wolfshaar KE, de Roos AM, Persson L (2008) Population feedback after successful invasion leads to ecological suicide in seasonal environments. Ecology 89:259–268
Vinkler M, Bainova H, Albrecht T (2010) Functional analysis of the skin-swelling response to phytohaemagglutinin. Funct Ecol 24:1081–1086
Acknowledgments
We thank B. Decencière, M. Paquet and all students for assistance in the field. Raoul Van Damme and two anonymous reviewers made useful suggestions to increase the quality of a previous version of this article. This research was supported by the Centre National de la Recherche Scientifique (CNRS), an Agence Nationale de la Recherche (ANR) grant (07-JCJC-0120) to J.F.L.G., grants from the Institut Federatif Ecologie-Biodiversité-Evolution-Environnement (IFR 101) and a grant from the Région Île-de-France (Réseau de Recherche sur le Développement Soutenable R2DS 2007-006) to J.-F. L.G. and M.M.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Raoul Van Damme.
Rights and permissions
About this article
Cite this article
Mugabo, M., Marquis, O., Perret, S. et al. Direct and socially-mediated effects of food availability late in life on life-history variation in a short-lived lizard. Oecologia 166, 949–960 (2011). https://doi.org/10.1007/s00442-011-1933-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-011-1933-0