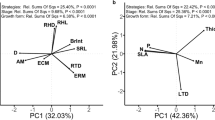
Abstract
The atmospheric CO2 concentration [CO2] has been increasing markedly since the industrial revolution and is predicted to reach 500–1,000 μmol mol−1 by the end of this century. Although the short-term and acclimatory responses to elevated [CO2] have been well studied, much less is understood about evolutionary responses to high [CO2]. We studied phenotypic and genetic differences in Plantago asiatica populations around a natural CO2 spring, where [CO2] has been consistently high over an evolutionary time scale. Our common-garden experiment revealed that plants transferred from habitats with higher [CO2] had higher relative growth rates, greater leaf to root ratios, lower photosynthetic rates, and lower stomatal conductance. The habitat-dependent differences were partly heritable because a similar trend of leaf to root ratio was found among their offsprings. Genetic analyses indicated that selfing or biparental inbreeding might promote local adaptation in areas with high [CO2] despite substantial gene flow across the [CO2] gradient. These results indicate that phenotypic and genetic differences have occurred between high and normal [CO2] populations.




Similar content being viewed by others
References
Andalo C, Goldringer I, Godelle B (2001) Inter- and intergenotypic competition under elevated carbon dioxide in Arabidopsis thaliana. Ecology 82:157–164
Antonovics J (1968) Evolution in closely adjacent plant populations. V. Evolution of self-fertility. Heredity 23:219–238
Bernacchi CJ, Coleman JS, Bazzaz FA, McConnaughay KDM (2000) Biomass allocation in old-field annual species grown in elevated CO2 environments: no evidence for optimal partitioning. Glob Change Biol 6:855–863
Bezemer TM, Thompson LJ, Jones TH (1998) Poa annua shows inter-generational differences in response to elevated CO2. Glob Change Biol 4:687–691
Bossdorf O, Richards CL, Pigliucci M (2008) Epigenetics for ecologists. Ecol Lett 11:106–115
Chinnusamy V, Zhu J-K (2009) Epigenetic regulation of stress responses in plants. Curr Opin Plant Biol 12:133–139
Edwards K, Johnstone C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA fro PCR analysis. Nucleic Acid Res 19:1349
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Excoffier L, Laval G, Schneider S (2005) Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evol Bioinfo Online 1:47–50
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 164:1567–1587
Fordham M, Barnes JD, Bettarini I, Polle A, Slee N, Raines C, Miglietta F, Raschi A (1997) The impact of elevated CO2 on growth and photosynthesis in Agrostis canina L. ssp. Monteluccii adapted to contrasting atmospheric CO2 concentrations. Oecologia 110:169–178
Grant-Downton RT, Dickinson HG (2005) Epigenetics and its implications for plant biology 1. The epigenetic network in plants. Ann Bot 96:1143–1164
Grant-Downton RT, Dickinson HG (2006) Epigenetics and its implications for plant biology 2. The ‘epigenetic epiphany’: epigenetics evolution and beyond. Ann Bot 97:11–27
Hewitt EJ, Smith TA (1975) Plant mineral nutrition. English University Press, London
Hikosaka K, Yamano T, Nagashima H, Hirose T (2003) Light-acquisition and use of individuals as influenced by elevated CO2 in even-aged monospecific stands of Chenopodium album. Funct Ecol 17:786–795
Hu XS, Li B (2003) On migration load of seeds and pollen grains in a local population. Heredity 90:162–168
Ishikawa N, Yokoyama J, Ikeda H, Takabe E, Tsukaya H (2006) Evolution of morphological variation in Plantago asiatica var. densiuscula, with special reference to the systematic treatment of Plantago asiatica var. yakusimensis. J Plant Res 119:385–395
Ishikawa K, Onoda Y, Hikosaka K (2007) Intraspecific variation in temperature dependence of gas exchange characteristics of Plantago asiatica ecotypes from different temperature regimes. New Phytol 176:356–364
Ishizaki S, Hikosaka K, Hirose T (2003) Increase in leaf mass per area benefits plant growth at elevated CO2 concentration. Ann Bot 91:905–914
Kawecki TJ, Ebert D (2004) Conceptual issues in local adaptation. Ecol Lett 7:1225–1241
Körner C, Bazzaz FA (1996) Carbon dioxide, populations, and communities. Academic, San Diego
Lau JA, Peiffer J, Reich PB, Tiffin P (2008) Transgenerational effects of global environmental change: long-term CO2 and nitrogen treatments influence offspring growth response to elevated CO2. Oecologia 158:141–150
Linhart YB, Grant MC (1996) Evolutionary significance of local genetic differentiation in plants. Annu Rev Ecol Syst 27:237–277
Marshall TC, Slate J, Kruuk LE, Pemberton JM (1998) Statistical confidence for likelihood-based paternity inference in natural populations. Mol Ecol 7:639–655
Miglietta F, Raschi A, Bettarini I, Resti R, Selvi F (1993) Natural CO2 springs in Italy: a resource for examining long-term response of vegetation to rising atmospheric CO2 concentrations. Plant Cell Environ 16:873–878
Nagashima H, Yamano T, Hikosaka K, Hirose T (2003) Effects of elevated CO2 on the size structure in even-aged monospecific stands of Chenopodium album. Glob Change Biol 9:619–629
Obeso JR (2002) The costs of reproduction in plants. New Phytol 155:321–348
Onoda Y, Hirose T, Hikosaka K (2007) Effect of elevated CO2 on leaf starch, nitrogen and photosynthesis of plants growing at three natural CO2 springs in Japan. Ecol Res 22:475–484
Onoda Y, Hirose T, Hikosaka K (2009) Does leaf photosynthesis adapt to CO2-enriched environments? An experiment on plants originating from three natural CO2 springs. New Phytol 182:698–702
Osada N, Onoda Y, Hikosaka K (2010) Effects of atmospheric CO2 concentration, irradiance and soil nitrogen availability on leaf photosynthetic traits on Polygonum sachalinense around the natural CO2 springs in northern Japan. Oecologia 164:41–52
Pfanz H, Vodnik D, Wittman C, Aschan G, Raschi A (2004) Plants and geothermal CO2 exhalations. Survival in and adaptation to a high CO2 environment. Prog Bot 65:499–537
Polle A, McKee I, Blaschke L (2001) Altered physiological and growth responses to elevated [CO2] in offspring from holm oak (Quercus ilex L.) mother trees with lifetime exposure to naturally elevated [CO2]. Plant Cell Environ 24:1075–1083
Porra RJ, Thompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure from multilocus genotype data. Genetics 155:945–959
Raschi A, Miglietta F, Tognetti R, van Gardingen PR (1999) Plant responses to elevated CO2. Evidence from natural springs. Cambridge University Press, Cambridge
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Reekie EG, Bazzaz FA (1992) Cost of reproduction as reduced growth in genotypes of two congeneric species with contrasting life histories. Oecologia 90:21–26
Saxe H, Ellsworth DS, Heath J (1998) Trees and forest functioning in an enriched CO2 atmosphere. New Phytol 139:395–436
Shitaka Y, Hirose T (1998) Effects of shift in flowering time on the reproductive output of Xanthium canadense in a seasonal environment. Oecologia 114:361–367
Squirrel J, Wolff K (2001) Isolation of polymorphic microsatellite loci in Plantago major and P. intermedia. Mol Ecol Note 1:179–181
Steinger T, Stephan A, Schmid B (2007) Predicting adaptive evolution under elevated atmospheric CO2 in the perennial grass Bromus erectus. Glob Change Biol 13:1028–1039
Ward JK, Kelly JK (2004) Scaling up evolutionary responses to elevated CO2: lessons from Arabidopsis. Ecol Lett 7:427–440
Ward JK, Antonovics J, Thomas RB, Strain BR (2000) Is atmospheric CO2 a selective agent on model C3 annuals? Oecologia 123:330–341
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:358–1370
Wieneke S, Prati D, Brandl R, Stöcklin J, Auge H (2004) Genetic variation in Sanguisorba minor after 6 years in situ selection under elevated CO2. Glob Change Biol 10:1389–1401
Wilson JB (1988) A review of evidence on the control of shoot: root ratio, in relation to models. Ann Bot 61:433–449
Acknowledgments
We thank Yudonosan-shrine, the landowner of the CO2 spring, for permission for the study. We thank Yayoi Koizumi, Risako Akita, Tomomi Kojima and Soichiro Nagano for technical support and Roger Butlin for comments on an early draft. The study was supported in part by KAKENHI (16687001, 19370008, 20677001, 21114009), Global Environment Research Fund (D-0904) and Global COE program (J03).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Evan DeLucia.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nakamura, I., Onoda, Y., Matsushima, N. et al. Phenotypic and genetic differences in a perennial herb across a natural gradient of CO2 concentration. Oecologia 165, 809–818 (2011). https://doi.org/10.1007/s00442-010-1900-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-010-1900-1