Abstract
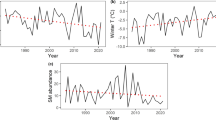
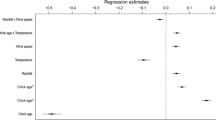
The ongoing climate change has improved our understanding of how climate affects the reproduction of animals. However, the interaction between food availability and climate on breeding has rarely been examined. While it has been shown that breeding of boreal birds of prey is first and foremost determined by prey abundance, little information exists on how climatic conditions influence this relationship. We studied the joint effects of main prey abundance and ambient weather on timing of breeding and reproductive success of two smaller (pygmy owl Glaucidium passerinum and Tengmalm’s owl Aegolius funereus) and two larger (tawny owl Strix aluco and Ural owl Strix uralensis) avian predator species using long-term nation-wide datasets during 1973–2004. We found no temporal trend either in vole abundance or in hatching date and brood size of any studied owl species. In the larger species, increasing late winter or early spring temperature advanced breeding at least as much as did high autumn abundance of prey (voles). Furthermore, increasing snow depth delayed breeding of the largest species (Ural owl), presumably by reducing the availability of voles. Brood size was strongly determined by spring vole abundance in all four owl species. These results show that climate directly affects the breeding performance of vole-eating boreal avian predators much more than previously thought. According to earlier studies, small-sized species should advance their breeding more than larger species in response to increasing temperature. However, we found an opposite pattern, with larger species being more sensitive to temperature. We argue that this pattern is caused by a difference in the breeding tactics of larger mostly capital breeding and smaller mostly income breeding owl species.


Similar content being viewed by others
References
Aars J, Ims, RA (2002) Intrinsic and climatic determinants of population demography: the winter dynamics of tundra voles. Ecology 83:3449–3456
Altwegg R, Roulin A, Kestenholz M, Jenni L (2006) Demographic effects of extreme winter weather in the barn owl. Oecologia 149:44–51. doi:10.1007/s00442-006-0430-3
Begon M, Townsend CR, Harper JL (2006) Ecology, individuals, populations and communities, 4th edn. Blackwell, Boston
Bierman SM, Fairbairn JP, Petty SJ, Elston DA, Tidhar D, Lambin X (2006) Changes over time in the spatiotemporal dynamics of cyclic population of field voles (Microtus agrestis L.). Am Nat 167:583–590. doi:10.1086/501076
Birkhead M, Bacon P, Walter P (1983) Factors affecting the breeding success of the mute swan Cygnus olor. J Anim Ecol 52:727–741. doi:10.2307/4450
Both C, Bouwhuis S, Lessels CM, Visser ME (2006) Climate change and population declines in a long-distance migratory bird. Nature 441:81–83. doi:10.1038/nature04539
Brinkhof MWG, Cavé AJ, Hage FJ, Verhulst S (1993) Timing of reproduction and fledging success in the coot Fulica atra: evidence for a causal relationship. J Anim Ecol 62:577–587
Brommer JE, Pietiäinen H, Kolunen H (2002) Reproduction and survival in a variable environment: Ural owls (Strix uralensis) and the three-year vole cycle. Auk 119:544–550. doi:10.1642/0004-8038(2002)119[0544:RASIAV]2.0.CO;2
Brommer JE, Karell P, Pietiainen H (2004) Supplementary fed Ural owls increase their reproductive output with a one year time lag. Oecologia 139:354–358. doi:10.1007/s00442-004-1528-0
Brommer JE, Pietiäinen H, Ahola K, Karell P, Karstinen T, Kolunen H (2009) The return of the vole cycle in southern Finland refutes the generality of the loss of cycles through ‘climatic forcing’. Glob Change Biol 15. doi:10.1111/j.1365-2486.2009.02012.x
Burnhamn K, Anderson D (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Charmantier A, McCleery RH, Cole LR, Perrins C, Kruuk LEB, Sheldon BC (2008) Adaptive phenotypic plasticity in response to climate change in a wild bird population. Science 320:800–803. doi:10:1126/science.1157174
Clobert J, Danchin E, Dhondt AA, Nichols JD (2001) Dispersal. Oxford University Press, Oxford
Cox CB, Moore PD (2005) Biogegraphy. An ecological and evolutionary approach, 7th edn. Blackwell, Boston
Cramp S (1985) The birds of the Western Palearctic, vol IV. Oxford University Press, Oxford
Daan S, Tinbergen JM (1997) Adaptation of life history. In: Krebs JR, Davies NB (eds) Behavioural ecology, 4th edn. Blackwell, Oxford, pp 311–333
Drent RH, Daan S (1980) The prudent parent: energetic adjustments in avian breeding. Ardea 68:225–252
Dunn P (2004) Breeding dates and reproductive performance. In: Møller AP et al (eds) Advances in ecological research: birds and climate change. Elsevier, London, pp 69–87
Francis CM, Saurola P (2004) Estimating components of variance in demographic parameters of tawny owls, Strix aluco. Anim Biodivers Conserv 27(1):489–502
Hakkarainen H, Korpimäki E, Koivunen V, Ydenberg R (2002) Survival of male Tengmalm’s owls under temporally varying food conditions. Oecologia 131:83–88. doi:10.1007/s00442-001-0865-5
Halonen M, Mappes T, Meri T, Suhonen J (2007) Influence of snow cover on food hoarding in pygmy owls Glaucidium passerinum. Ornis Fenn 84:105–111
Hanski I, Hansson L, Henttonen H (1991) Specialist predators, generalist predators and the microtine rodent cycle. J Anim Ecol 60:353–367
Hansson L, Henttonen H (1985) Gradients in density variations of small rodents: the importance of latitude and snow cover. Oecologia 67:394–402. doi:10.1007/BF00384946
Hansson L, Henttonen H (1988) Rodent dynamics as community processes. Trends Ecol Evol 3:195–200. doi:10.1016/0169-5347(88)90006-7
Heikkinen RK, Luoto M, Virkkala R, Pearson RG, Körber JH (2007) Biotic interactions improve predictions of boreal bird distributions at macro-scales. Glob Ecol Biogeogr 16:754–763. doi:10.1111/j.1466-8238.2007.00345.x
Hirons GJM, Hardy AR, Stanley PI (1984) Body weight, gonad development and moult in the tawny owl (Strix aluco). J Zool 202:145–164
Hörnfeldt B, Eklund U (1990) The effect of food on laying date and clutch-size in Tengmalm’s owl Aegolius funereus. Ibis 132:395–406
Hörnfeldt B, Carlsson BG, Löfgren O, Eklund U (1990) Effects of cyclic food-supply on breeding performance in Tengmalm’s owl (Aegolius funereus). Can J Zool 68:522–530
Huitu O, Laaksonen J, Klemola T, Korpimäki E (2008) Spatial dynamics of Microtus vole populations in continuous and fragmented agricultural landscapes. Oecologia 155:53–61. doi:10.1007/s00442-007-0885-x
Ims RA, Henden J-A, Killengreen ST (2008) Collapsing population cycles. Trends Ecol Evol 23:79–86. doi:10.1016/j.tree.2007.10.010
Jokinen M (1975) Lehtopöllöpoikasten kasvu ja energiankäyttö ja niiden vaikutus pesimätuloksen määrittämiseen. MSc thesis, University of Turku
Jönsson KI (1997) Capital and income breeding as alternative tactics of resource use in reproduction. Oikos 78:57–66. doi:10.2307/3545800
Karell P (2007) Short- and long-term consequences of food resources on Ural owl Strix uralensis reproduction. PhD thesis, University of Helsinki
Karell P, Kontiainen P, Pietiäinen H, Siitari H, Brommer JE (2008) Maternal effects on offspring Igs and eggsize in relation to natural and experimentally improved food supply. Funct Ecol 22:682–690. doi:10:1111/j.1365-2435.2008.01425.x
Korpimäki E (1981) On the ecology and biology of Tengmalm’s Owl (Aegolius funereus) in Southern Ostrabothnia and Suomenselkä, western Finland. Acta Universitatis Ouluensis A 118. Biol 13:1–84
Korpimäki E (1986) Seasonal changes in the food of Tengmalm’s owl Aegolius funereus in western Finland. Ann Zool Fenn 23:339–344
Korpimäki E (1987a) Clutch size, breeding success and brood size experiments in Tengmalm’s owl Aegolius funereus: a test of hypotheses. Ornis Scan 18:277–284
Korpimäki E (1987b) Timing of breeding of Tengmalm’s owl Aegolius funereus in relation to vole dynamics in western Finland. Ibis 129:58–68
Korpimäki E (1990) Body mass of breeding Tengmalm’s owls Aegolius funereus: seasonal, between year, site and age related variation. Ornis Scand 21:169–178
Korpimäki E, Hakkarainen H (1991) Fluctuating food supply affects the clutch size of Tengmalm’s owl independent of laying date. Oecologia 85:543–552. doi:10.1007/BF00323767
Korpimäki E, Lagerström M, Saurola P (1987) Field evidence for nomadism in Tengmalm owl Aegolius funereus. Ornis Scan 18:1–4
Korpimäki E, Norrdahl K, Huitu O, Klemola T (2005) Predator-induced synchrony in population oscillations of co-existing small mammal species. Proc R Soc Lond B 272:193–202. doi:10.1098/rspb.2004.2860
Krüger O (2004) The importance of competition, food, habitat, weather and phenotype for the reproduction of Buzzard Buteo buteo. Bird Study 51:125–132. doi:10.1080/00063650409461344
Kullberg G (1995) Strategy of the pygmy owl while hunting avian and mammalian prey. Ornis Fenn 72:72–78
Lehikoinen A, Öst M, Kilpi M (2006) Winter climate affects subsequent breeding success of common eiders. Glob Change Biol 12:1355–1365. doi:10.1111/j.1365-2486.2006.01162.x
Lehikoinen A, Byholm P, Ranta E, Saurola P, Valkama J, Korpimäki E, Pietiäinen H, Henttonen H (2009) Reproduction of common buzzard at its northern range margin under climate change. Oikos 118:829–836. doi:10.1111/j.1600-0706.2008.17440.x
Ludwig GX, Alatalo RV, Helle P, Lindén H, Lindström J, Siitari H (2006) Short- and long-term population dynamical consequences of asymmetric climate change in black grouse. Proc R Soc Lond B 273:2009–2016. doi:10.1098/rspb.2006.3538
Martin TE (2007) Climate correlates of 20 years of trophic changes in high-elevation riparian system. Ecology 88:367–380. doi:10.1890/0012-9658(2007)88[367:CCOYOT]2.0.CO;2
Meijer T, Drent R (1999) Re-examination of the capital and income dichotomy in breeding birds. Ibis 141:399–414. doi:10.1111/j.1474-919X.1999.tb04409.x
Mikkola H (1983) Owls of Europe. Poyser, Calton
Newton I (1998) Population limitation in birds. Academic, London
Nooker JK, Dunn PO, Whittingham LA (2005) Effects of food abundance, weather, and female condition on reproduction in tree swallows (Tachycineta bicolor). Auk 122:1225–1238. doi:10.1642/0004-8038(2005)122[1225:EOFAWA]2.0.CO;2
Pietiäinen H (1989) Seasonal and individual variation in the production of offspring in the Ural owl Strix uralensis. J Anim Ecol 58:905–920. doi:10.2307/5132
Pietiäinen H, Kolunen H (1993) Female body condition and breeding of the Ural owl Strix uralensis. Funct Ecol 7:726–735. doi:10.2307/2390195
Pihlaja T (1999) Growth of nestling Ural owls. MSc thesis, University of Helsinki
Schönn S (1980) Der Sperlingkauz. Die Neue Brehm-Bücherei, Wittenberg-Lutherstadt
Solheim R (1984) Caching behaviour, prey choice and surplus killing by pygmy owls Glaucidium passerinum during winter, a functional response of a generalist predator. Ann Zool Fenn 21:301–308
Solonen T (2006) Overwinter population change of small mammals in Southern Finland. Ann Zool Fenn 43:295–302
Stevenson IR, Bryant DM (2000) Climate change and constraints on breeding. Nature 406:366–367. doi:10.1038/35019151
Sundell J, Huitu O, Henttonen H, Kaikusalo A, Korpimäki E, Pietiäinen H, Saurola P, Hanski I (2004) Large-scale spatial dynamics of vole populations in Finland revealed by the breeding success of vole eating avian predators. J Anim Ecol 73:167–178. doi:10.1111/j.1365-2656.2004.00795.x
Thomas DW, Blondel J, Perret P, Lambrechts MM, Speakman JR (2001) Energetic and fitness costs of mismatching resource supply and demand in seasonally breeding birds. Science 291:2598–2600. doi:10.1126/science.1057487
Visser ME, Both C, Lambrechts MM (2004) Global climate change leads to mistimed avian reproduction. In: Møller AP et al (eds) Advances in ecological research: birds and climate change. Elsevier, London, pp 89–110
Web-page of ageing of owls. http://www.nic.fi/~mattisj
Acknowledgments
Many voluntary bird ringers and observers have taken part in collecting the breeding and vole trapping data. The Ringing Centre of Finnish Museum of Natural History has maintained the electronic ringing databases since the 1970s and kindly helped with the data delivery. A large bulk of the long-term vole data have been collected in the national vole monitoring program by the Finnish Forest Research Institute; we thank Asko Kaikusalo for his help. We thank Nigel G. Yoccoz and an anonymous referee for their valuable comments. This work was supported by grants from Jenny and Antti Wihuri Foundation (to A.L.) and field work in Päijät-Häme was financed by the Academy of Finland (1985–1988 and 2000–2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Herwig Leirs.
Esa Ranta deceased during the preparation of the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lehikoinen, A., Ranta, E., Pietiäinen, H. et al. The impact of climate and cyclic food abundance on the timing of breeding and brood size in four boreal owl species. Oecologia 165, 349–355 (2011). https://doi.org/10.1007/s00442-010-1730-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-010-1730-1