Abstract
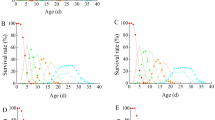
To what extent the combined effect of several parasite species co-infecting the same host (i.e. polyparasitism) affects the host’s fitness is a crucial question of ecological parasitology. We investigated whether the ecological setting can influence the co-infection’s outcome with the mosquito Aedes aegypti and two parasites: the microsporidium Vavraia culicis and the gregarine Ascogregarina culicis. The cost of being infected by the two parasites depended on the interaction between the two infectious doses and host food availability. The age at pupation of the mosquito was delayed most when the doses of the two parasites were highest and little food was available. As infectious dose increases with the parasites’ prevalence and intensity of transmission, the cost of being co-infected depends on the epidemiological status of the two parasite species.

Similar content being viewed by others
References
Alizon S (2008) Decreased overall virulence in coinfected hosts leads to the persistence of virulent parasites. Am Nat 172:E67–E79
Andreadis TG (2007) Microsporidian parasites of mosquitoes. J Am Mosq Control Assoc 23:3–29
Bedhomme S, Agnew P, Sidobre C, Michalakis Y (2004) Virulence reaction norms across a food gradient. Proc R Soc Lond B 271:739–744
Bonsall MB, Benmayor R (2005) Multiple infections alter density dependence in host–pathogen interactions. J Anim Ecol 74:937–945
Brown SP, Hochberg ME, Grenfell BT (2002) Does multiple infection select for raised virulence? Trends Microbiol 10:401–405
Brunner JL, Richards K, Collins JP (2005) Dose and host characteristics influence virulence of ranavirus infections. Oecologia 144:399–406
Chen WJ (1999) The life cycle of Ascogregarina taiwanensis (Apicomplexa:Lecudinidae). Parasitol Today 15:153–156
Christophers SR (1960) Aëdes aegypti (L.). The yellow fever mosquito. Its life history, bionomics and structure. Cambrige University Press, Cambridge
Cox FEG (2001) Concomitant infections, parasites and immune responses. Parasitology 122:S23–S38
de Roode JC, Helinski MEH, Anwar MA, Read AF (2005) Dynamics of multiple infection and within-host competition in genetically diverse malaria infections. Am Nat 166:531–542
de Roode JC, Gold LR, Altizer S (2007) Virulence determinants in a natural butterfly-parasite system. Parasitology 134:657–668
Ebert D, Zschokke-Rohringer CD, Carius HJ (2000) Dose effects and density-dependent regulation of two microparasites of Daphnia magna. Oecologia 122:200–209
Fellous S, Koella JC (2009) Infectious dose affects the outcome of the within-host competition between parasites. Am Nat 173:E177–E184
Fellous S, Salvaudon L (2009) How can your parasites become your allies? Trends Parasitol 25:62–66
Frank SA (1992) A kin selection model for the evolution of virulence. Proc R Soc Lond B 250:195–197
Gower CM, Webster JP (2005) Intraspecific competition and the evolution of virulence in a parasitic trematode. Evolution 59:544–553
Hochberg ME (1991) Intra-host interactions between a braconid endoparasitoid, Apanteles glomeratus, and a baculovirus for larvae of Pieris brassicae. J Anim Ecol 60:51–63
Jokela J, Lively CM, Taskinen J, Peters AD (1999) Effect of starvation on parasite-induced mortality in a freshwater snail (Potamopyrgus antipodarum). Oecologia 119:320–325
Jokela J, Taskinen J, Mutikainen P, Kopp K (2005) Virulence of parasites in hosts under environmental stress: experiments with anoxia and starvation. Oikos 108:156–164
Lambrechts L, Chavatte JM, Snounou G, Koella JC (2006) Environmental influence on the genetic basis of mosquito resistance to malaria parasites. Proc R Soc Lond B 273:1501–1506
Marzal A, Bensch S, Reviriego M, Balbontin J, De Lope F (2008) Effects of malaria double infection in birds: one plus one is not two. J Evol Biol 21:979–987
Michalakis Y, Bedhomme S, Biron DG, Rivero A, Sidobre C, Agnew P (2008) Virulence and resistance in a mosquito–microsporidium interaction. Evol Appl 1:49–56
Pullan R, Brooker S (2008) The health impact of polyparasitism in humans: are we under-estimating the burden of parasitic diseases? Parasitology 135:783–794
Regoes RR, Ebert D, Bonhoeffer S (2002) Dose-dependent infection rates of parasites produce the Allee effect in epidemiology. Proc R Soc Lond B 269:271–279
Reyes-Villanueva F, Becnel JJ, Butler JF (2003) Susceptibility of Aedes aegypti and Aedes albopictus larvae to Ascogregarina culicis and Ascogregarina taiwanensis (Apicomplexa: Lecudinidae) from Florida. J Invertebr Pathol 84:47–53
Roychoudhury S, Kobayashi M (2006) New findings on the developmental process of Ascogregarina taiwanensis and Ascogregarina culicis in Aedes albopictus and Aedes aegypti. J Am Mosq Control Assoc 22:29–36
Schmid-Hempel P, Frank SA (2007) Pathogenesis, virulence, and infective dose. PLoS Pathogens 3:e147
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Sulaiman I (1992) Infectivity and pathogenicity of Ascogregarina culicis (Eugregarinida: Lecudinidae) to Aedes aegypti (Diptera: Culicidae). J Med Entomol 29:1–4
Thomas MB, Watson EL, Valverde-Garcia P (2003) Mixed infections and insect–pathogen interactions. Ecol Lett 6:183–188
Vale PF, Salvaudon L, Kaltz O, Fellous S (2008) The role of the environment in the evolutionary ecology of host parasite interactions. Infect Genet Evol 8:302–305
Van Baalen M, Sabelis MW (1995) The dynamics of multiple infection and the evolution of virulence. Am Nat 146:881–910
Voordouw MJ, Anholt BR, Taylor PJ, Hurd H (2009) Rodent malaria-resistant strains of the mosquito, Anopheles gambiae, have slower population growth than susceptible strains. BMC Evol Biol 9:76
Acknowledgments
Thanks to Claudy Haussy for help in the lab and Jacqui Shykoff, Christophe Boëte, Alfonso Marzal and an anonymous reviewers for comments on earlier versions of this manuscript. S. F. was funded by an Allocation de Recherche du Ministère délégué à la Recherche et à l’Education Supérieure.
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jay Rosenheim.
Rights and permissions
About this article
Cite this article
Fellous, S., Koella, J.C. Cost of co-infection controlled by infectious dose combinations and food availability. Oecologia 162, 935–940 (2010). https://doi.org/10.1007/s00442-009-1535-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1535-2