Abstract
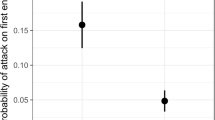
Acacia trees in Costa Rica have an obligate mutualism with three species of Pseudomyrmex ants, which vigorously defend their host tree from insect and mammalian herbivores. Depending on the size and species of ant colony, individual acacia trees may be differentially protected. For animals able to discern between weakly and highly aggressive ant colonies, costs of ant stings from less active colonies might be offset by nutritional value acquired from feeding on acacia fruit or ant larvae in swollen thorns. We examined foraging selectivity of capuchin monkeys on acacia trees in Santa Rosa National Park, Costa Rica. We measured four characteristics of the acacia trees from which capuchins fed and of acacias immediately adjacent to those in which the monkeys fed: diameter at breast height (DBH), accessibility, species of closest tree and ant species present. We found that capuchins prefer to forage in acacias that are large and accessible. We also made two measurements of ant colony activity on each tree, one before and one after disturbing the ant colony. We found that the three species of mutualistic ants differ in baseline activity levels and that mutualistic ants are more active than non-mutualistic ant species found in acacia trees. We also found that capuchins foraged more frequently in trees colonized by non-mutualistic ants, but the explanatory value (r 2) of this model was low. Furthermore, monkeys did not discriminate between acacias on the basis of baseline ant activity or the ant colony’s response to disturbance. We conclude that these monkeys select acacia trees in which to forage based on characteristics of the trees rather than the ants. In addition, our study suggests that white-faced capuchins act as predators on the acacia ants but they probably benefit the dispersal and reproductive success of acacia trees. Capuchins may in fact function as an additional mutualistic partner for acacia trees via seed dispersal, but they must overcome the ants’ defense of the trees to do so.



Similar content being viewed by others
References
Beattie AJ (1985) The evolutionary ecology of ant–plant mutualisms. Cambridge University Press, Cambridge
Bronstein JL (1991) The non-pollinating wasp fauna of Ficus pertusa: exploitation of a mutualism? Oikos 61:175–186
Cheney KL, Côté IM (2005) Frequency dependent success of aggressive mimics in a cleaning symbiosis. Proc R Soc Lond B 272:2635–2639
Connor RC (1995) The benefits of mutualism: a conceptual framework. Biol Rev Camb Philos Soc 70:427–457
Crawley MJ (2005) Statistics. An introduction using R. Wiley, Chichester
Cronin G (1998) Between-species and temporal variation in acacia-ant herbivore interactions. Biotropica 30:135–139
Ebinger JE, Seigler DS, Clarke HD (2000) Taxonomic revision of South American species of the genus Acacia subgenus Acacia (Fabaceae: Mimosoideae). Syst Bot 25:588–617
Fedigan LM, Jack K (2005) The demographic and reproductive context of male replacements in Cebus capucinus. Behaviour 141:755–775
Fragaszy D, Visalberghi E, Fedigan LM (2004) The complete capuchin. Cambridge University Press, Cambridge
Handel SN, Beattie AJ (1990) Seed dispersal by ants. Sci Am 263:76–83
Hibbett DS (2002) Plant–fungal interactions: when good relationships go bad. Nature 419:345–346
Janson CH (1985) Aggressive competition and individual food consumption in wild brown capuchin monkeys (Cebus apella). Behav Ecol Sociobiol 18:125–138
Janzen DH (1966) Co-evolution of mutualism between ants and acacias in Central America. Evolution 120:249–275
Janzen DH (1967) Fire, vegetation structure and the ant X acacia interaction in Central America. Ecology 48:26–35
Janzen DH (1969) Birds and the ant × acacia interaction in Central America, with notes on birds and other myrmecophytes. Condor 71:240–256
Janzen DH (1975) Pseudomyrmex nigropilosa: a parasite of a mutualism. Science 188:936–937
Janzen DH (1983) Costa Rican natural history. University of Chicago Press, Chicago, Ill.
Letourneau DK (1990) Code of ant-plant mutualism broken by parasite. Science 248:215–217
Maddala GS (1983) Limited-dependent and qualitative variables in econometrics. Cambridge University Press, Cambridge
McCabe G, Fedigan LM (2007) Effects of reproductive status on energy intake, ingestion rates and dietary composition of female white-faced capuchins (Cebus capucinus) at Santa Rosa, Costa Rica. Int J Primatol 28:837–851
Melin A, Fedigan LM, Hiramatsu C, Sendall C, Kawamura S (2007) Effect of colour vision phenotype on insect capture by a free-ranging population of white-faced capuchins (Cebus capucinus). Anim Behav 73:205–214
Miller MF (1996) Dispersal of Acacia seeds by ungulates and ostriches in an African savanna. J Trop Ecol 12:345–356
O’Malley R, Fedigan LM (2005) Variability in food-processing behavior among white-faced capuchins (Cebus capucinus) in Santa Rosa National Park, Costa Rica. Am J Phys Anthropol 128:63–73
O’Malley R, Fedigan LM (2006) Age/sex class differences in foraging on Acacia-dwelling ants by white-faced capuchins (Cebus capucinus). Am J Phys Anthropol [Suppl] 42:141
Or K, Ward D (2003) Three-way interactions between Acacia, large mammalian herbivores and bruchid beetles—a review. Afr J Ecol 41:257–265
Panger M, Perry S, Rose L, Gros-Louis J, Vogel E, MacKinnon K, Baker M (2002) Food processing technique differences across three capuchin (Cebus capucinus) populations in Costa Rica. Am J Phys Anthropol 119:52–66
R Development Core Team (2006) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org. Cited 6 Mar 2003
Seigler DS, Ebinger JE (1995) Taxonomic revision of the ant-acacias (Fabaceae, Mimosoideae, Acacia, series Gummiferae) of the new world. Ann Mo Bot Gard 82:117–138
Stanton ML, Palmer TM, Young TP, Evans A, Turner ML (1999) Sterilization and canopy modification of a swollen thorn acacia tree by a plant-ant. Nature 401:578–581
Valenta K (2007) Quantity, quality and spatial patterns of seed dispersal in Cebus capucinus. MA thesis, University of Calgary
Veall MR, Zimmermann KF (1996) Pseudo-R2 measures for some common limited dependent variable models. J Econ Surv 10(3):241–259
Ward PS (1993) Systematic studies on Pseudomyrmex acacia-ants (Hymenoptera: Formicidae: Pseudomyrmecinae). J Hymenopt Res 2:117–168
Wehnke EV, Hubbell SP, Foster RB, Dalling JW (2003) Seed dispersal patterns produced by white-faced monkeys: implications for the dispersal limitation of neotropical tree species. J Ecol 91:677–685
Wehnke EV, Valdez CN, Dominguez CA (2004) Seed dispersal and defecation patterns of Cebus capucinus and Alouatta palliata: consequences for seed dispersal effectiveness. J Trop Ecol 20:535–543
Willmer PG, Stone GN (1997) How aggressive ant-guards assist seed-set in Acacia flowers. Nature 388:165–167
Young BE, Kaspari M, Martin TE (1990) Species-specific nest site selection by birds in ant-acacia trees. Biotropica 22:310–315
Acknowledgements
We thank the staff of ACG, especially R. Blanco Segura for help with this study and permission to do research in the park. We are grateful to Dr J. Longino for help with ant identification and to Dr R. Longair for advice on methodology. This project was funded by a Sigma XI Grant (H. Y.), by the Canada Research Chairs Program (L. M. F.) and by NSERC Grants (L. M. F. and J. F. A.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Craig Osenberg.
Rights and permissions
About this article
Cite this article
Young, H., Fedigan, L.M. & Addicott, J.F. Look before leaping: foraging selectivity of capuchin monkeys on acacia trees in Costa Rica. Oecologia 155, 85–92 (2008). https://doi.org/10.1007/s00442-007-0883-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0883-z