Abstract
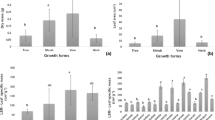
Functional responses to environmental variation do not only depend on the genetic potential of a species to express different trait values, but can also be limited by characteristics, such as the timing of organ (pre-) formation, aboveground longevity or the presence of a storage organ. In this experiment we tested to what degree variation in tuber size and organ preformation constrain the responsiveness to environmental quality and whether responsiveness is modified by the availability of stored resources by exposing the spring geophyte Bunium bulbocastanum to different light and nutrient regimes. Growth and biomass partitioning were affected by initial tuber size and resource availability. On average, tuber weight amounted to 60%, but never less than 30% of the total plant biomass. Initial tuber size, considered an estimate of the total carbon pool available at the onset of treatments, affected plant growth and reproduction throughout the experiment but had little effect on the responsiveness of plants to the treatments. The responsiveness was partly constrained by organ preformation: in the second year variation of leaf number was considerably larger than in the first year of the treatments. The results indicate that a spring geophyte with organ preformation has only limited possibilities to respond to short-term fluctuations of the environment, as all leaves and the inflorescence are preformed in the previous growing season and resources stored in tubers are predominantly used for survival during dormancy and are not invested into plastic adjustments to environmental quality. Such spring geophytes have only limited possibilities to buffer environmental variation. This explains their restriction to habitats characterized by predictable changes of the environmental conditions.




Similar content being viewed by others
References
Abrahamson WG (1979) Patterns of resource allocation in wildflower populations of fields and woods. Am J Bot 66:71–79
Aydelotte AR, Diggle PK (1997) Analysis of developmental preformation in the alpine herb Caltha leptosepala. Am J Bot 84:1646–1657
Beard JS (1983) Ecological control of the vegetation of southwestern Australia: moisture versus nutrients. In: Kruger FJ, et al (eds) Mediterranean-type ecosystems. Springer, Berlin Heidelberg New York, pp 66–73
Billings WD, Mooney HA (1968) The ecology of arctic and alpine plants. Biol Rev 43:481–529
Bobbink R, Willems JH (1987) Increasing dominance of Brachypodium pinnatum (L.) Beauv. in chalk grasslands: a threat to a species-rich ecosystem. Biol Cons 40:301–314
Chapin FS, Schulze ED, Mooney HA (1990) The ecology and economics of storage in plants. Annu Rev Ecol Syst 21:423–477
Diggle PK (1997) Extreme preformation in alpine Polygnonum viviparum: An architectural and developmental analysis. Am J Bot 84:154–169
Ehrlen J, van Groenendael J (2001) Storage and the delayed costs of reproduction in the understorey perennial Lathyrus vernus. J Ecol 89:237–246
Elemans M (2005) Plant traits in forest understory herbs—a modeling study. PhD thesis, Utrecht University
Geber MA, de Kroon H, Watson MA (1997a) Organ preformation in mayapple as a mechanism for historical effects on demography. J Ecol 85:211–223
Geber MA, Watson MA, de Kroon H (1997b) Organ preformation, development and resource allocation in perennials. In: Bazzaz FA, Grace J (eds) Plant resource allocation. Academic, New York, pp 113–141
Harper JL (1977) Population biology of plants. Academic, London
Hegi G (1975) Illustrierte Flora von Mitteleuropa. Band V/2. P.Parey, Berlin
Huber H, Stuefer JF, Willems JH (1996) Environmentally induced carry-over effects on seed production, germination and seedling performance in Bunium bulbocastanum (Apiaceae). Flora 191:353–361
Huber H, Fijan A, During HJ (1998) A comparative study of spacer plasticity in erect and stoloniferous herbs. Oikos 81:576–586
Huber H, Whigham DF, O’Neill J (2004) Time of disturbance can change life history decisions in the clonal forest understory herb Uvularia perfoliata. Evol Ecol 18:521–539
Inouye DW (1986) Long-term preformation of leaves and inflorescences by a long-lived perennial monocarp, Frasera speciosa (Gentianaceae). Am J Bot 73:1535–1540
Irmisch T (1854) Beiträge zur vergleichenden Morphologie der Pflanzen I. Abhandlungen der naturforschenden Gesellschaft zu Halle 2:31–80
Iwasa Y, Cohen D (1989) Optimal growth schedule of a perennial plant. Am Nat 133:480–505
Iwasa Y, Kubo T (1997) Optimal storage for recovery after unpredictable disturbances. Evol Ecol 11:41–65
Kawano S (1985) Life history characteristics of temperate woodland plants in Japan. In: White J (ed) The population structure of vegetation. (W.) Junk, The Hague, pp 515–549
Kleijn D, Treier UA, Müller-Schärer H (2005) The importance of nitrogen and carbohdrate storage for plant growth of the alpine herb Veratrum album. New Phytol 166:565–575
Körner C (2003) Alpine plant life, 2nd edn. Springer, Berlin Heidelberg New York
de Kroon H, Huber H, Stuefer JF, van Groenendael JM (2005) A modular concept of phenotypic plasticity in plants. New Phytol 166:73–82
Lovett Doust J (1980) Experimental manipulation of patterns of resource allocation in the growth cycle and reproduction of Smyrnium olustratum L. Biol J Linn Soc 13:155–166
Meloche CG, Diggle PK (2003) The pattern of carbon allocation supporting growth of preformed shoot primordia in Acomastylis rossii (Rosaceae). Am J Bot 90:1313–1320
Meusel H, Jaeger E, Rauschert S, Weinert E (1978) Vergleichende Chorologie der zentraleuropaeischen Flora. Band II. Fischer, Jena
Mössler JG (1833) Mösslers Handbuch der Gewächskunde. 1. Band 2. Abteilung 3. Auflage, JF Hammerich, Altona
Onipchenko V (ed) (2004) Alpine vegetation ecology of the western Caucasus. Kluwer, Dordrecht
Orshan G (1989) Plant pheno-morphological studies in Mediterranean type ecosystems. Kluwer, Dordrecht
Othen B, Wehrmeyer A (2004) Seasonal dynamics of non-structural carbohydrates in bulbs & shoots of the geophyte Galanthus nivalis. Physiol Plant 120:529–536
Packham JR, Harding DJL (1982) Ecology of woodland processes. Edward Arnold, London
Pantis JD (1993) Biomass and nutrient allocation patterns in the Mediterranean geophyte Asphodelus aestivus Brot (Thessaly, Greece). Acta Oecol 14:489–500
Pigliucci M (2001) Phenotypic plasticity: beyond nature and nurture. The Johns Hopkins University Press, Baltimore
Rauh M (1950) Morphologie der Nutzpflanzen. Quelle & Meyer, Heidelberg
Rockwood LL, Lobstein MB (1994) The effects of experimental defoliation on reproduction in four species of herbaceous perennials from northern Virginia. Castanea 59:41–50
Salisbury EJ (1925) The structure of woodlands. Veroff Geobot Inst Rübel Zürich 3:334–354
Schlichting CD, Pigliucci M (1998) Phenotypic evolution. A reaction norm perspective. Sinauer Associates Inc., Sunderland
Sohn JJ, Policansky D (1977) The costs of reproduction in the mayapple Podophyllum peltatum (Berberidaceae). Ecology 58:1366–1374
Suzuki JI, Stuefer JF (1999) On the ecological and evolutionary significance of storage in clonal plants. Plant Species Biol 14:11–17
von Willert DJ, Eller BM, Werger MJA, Brinckmann E, Ihlenfeldt HD (1992) Life strategies of succulents in deserts. Cambridge University Press, Cambridge
Werger MJA, van Laar EMJM (1985) Seasonal changes in the structure of the herb layer of a deciduous woodland. Flora 176:351–364
Whigham DF (1974) An ecological life-history study of Uvularia perfoliata. Am Midl Nat 91:343–359
Whigham DF (1990) The effects of experimental defoliation on the growth and reproduction of a woodland orchid, Tipularia discolor. Can J Bot 68:1812–1816
Wijensinghe DK, Whigham DF (1997) Cost of producing clonal offspring and the effects of plant size on population dynamics of the woodland herb Uvularia perfoliata (Liliaceae). J Ecol 85:907–919
Willems JH, Bobbink R (1990) Spatial processes in the succession of chalk grassland on old fields in the Netherlands. In: Krahulec F, Agnew AD, Willems JH (eds) Spatial processes in plant communities. SPB Academic Publishing, The Hague, pp 237–249
Worley AC, Harder LD (1999) Consequences of preformation for dynamic resource allocation by a carnivorous herb, Pinguicula vulgaris (Lentibulariaceae). Am J Bot 86:1136–1145
Wyka T (1999) Carbohydrate storage and use in an alpine population of the perennial herb, Oxytropis sericea. Oecologia 120:198–208
Wyka T (2000) Effects of nutrients on growth rate and carbohydrate storage in Oxytropis sericea: A test of the carbon accumulation hypothesis. Int J Plant Sci 161:381–386
Zimmerman JK, Whigham DF (1992) Ecological functions of carbohydrates stored in corms of Tipularia discolor (Orchidaceae). Funct Ecol 6:575–561
Acknowledgements
We thank Fan Zhang for invaluable help during the experiment, and Heinjo During and Josef Stuefer and two anonymous referees for insightful comments on an earlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Christian Koerner
Rights and permissions
About this article
Cite this article
Werger, M.J.A., Huber, H. Tuber size variation and organ preformation constrain growth responses of a spring geophyte. Oecologia 147, 396–405 (2006). https://doi.org/10.1007/s00442-005-0280-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-005-0280-4