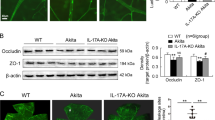
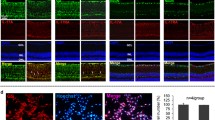
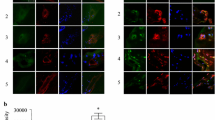
Abstract
T helper 22 (Th22) cells have been implicated in diabetic retinopathy (DR), but it remains unclear whether Th22 cells involve in the pathogenesis of DR. To investigate the role of Th22 cells in DR mice, the animal models were established by intraperitoneal injection of STZ and confirmed by fundus fluorescein angiography and retinal haematoxylin–eosin staining. IL-22BP was administered by intravitreal injection. IL-22 level was measured by ELISA in vivo and in vitro. The expression of IL-22Rα1 in the retina was assessed by immunofluorescence. We assessed GFAP, VEGF, ICAM-1, inflammatory-associated factors and the integrity of blood–retinal barrier in control, DR, IL-22BP, and sham group. Müller cells were co-cultured with Th22 cells, and the expression of the above proteins was measured by immunoblotting. Plasmid transfection technique was used to silence Act1 gene in Müller cells. Results in vivo and in vitro indicated that Th22 cells infiltrated into the DR retinal and IL-22Rα1 expressed in Müller cells. Th22 cells promoted Müller cells activation and inflammatory factor secretion by secreting IL-22 compared with high-glucose stimulation alone. In addition, IL-22BP ameliorated the pathological alterations of the retina in DR. Inhibition of the inflammatory signalling cascade through Act1 knockdown alleviated DR-like pathology. All in all, the results suggested that Th22 cells infiltrated into the retina and secreted IL-22 in DR, and then IL-22 binding with IL-22Rα1 activated the Act1/TRAF6 signal pathway, and promoted the inflammatory of Müller cells and involved the pathogenesis of DR.








Similar content being viewed by others
Availability of data and materials
All relevant data generated or analyzed during this study are included in this published article.
Abbreviations
- DR:
-
Diabetic retinopathy
- BRB:
-
Blood–retinal barrier
- iBRB:
-
Inner blood–retinal barrier
- oBRB:
-
Outer blood–retinal barrier
- STZ:
-
Streptozotocin
- FFA:
-
Fundus fluorescein angiography;
- HE:
-
Haematoxylin-eosin staining;
- GFAP:
-
Glial fibrillary acidic protein
- VEGF:
-
Vascular endothelial growth factor;
- ICAM-1:
-
Intercellular adhesion molecule-1
- GS:
-
Glutamate synthase
- IL-22BP:
-
IL-22 binding protein
References
Altmann C, Schmidt MHH (2018) The role of microglia in diabetic retinopathy: inflammation, microvasculature defects and neurodegeneration. Int J Mol Sci 19
Arif S, Pujol-Autonell I, Eichmann M (2020) Assessing effector T cells in type 1 diabetes. Curr Opin Endocrinol Diabetes Obes 27:240–247
Azizi G, Simhag A, El Rouby NM, Mirshafiey A (2015) Th22 cells contribution in immunopathogenesis of rheumatic diseases. Iran J Allergy Asthma Immunol 14:246–254
Bell K, Holz A, Ludwig K, Pfeiffer N, Grus FH (2017) Elevated regulatory T cell levels in glaucoma patients in comparison to healthy controls. Curr Eye Res 42:562–567
Bhuyan ZA, Asanoma M, Iwata A, Ishifune C, Maekawa Y, Shimada M, Yasutomo K (2014) Abrogation of Rbpj attenuates experimental autoimmune uveoretinitis by inhibiting IL-22-producing CD4+ T cells. PLoS ONE 9:e89266
Che Y, Su Z, Xia L (2020) Effects of IL-22 on cardiovascular diseases. Int Immunopharmacol 81:106277
Chen WY, Qian Y, Chen TY, Gu XP (2021) Research progress of CD4(+)T cells-mediated regulation of neuroinflammation involved in neurodegenerative diseases. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 43:628–633
Coughlin BA, Feenstra DJ, Mohr S (2017) Müller cells and diabetic retinopathy. Vision Res 139:93–100
Couturier A, Blot G, Vignaud L, Nanteau C, Slembrouck-Brec A, Fradot V, Acar N, Sahel JA, Tadayoni R, Thuret G, Sennlaub F, Roger JE, Goureau O, Guillonneau X, Reichman S (2021) Reproducing diabetic retinopathy features using newly developed human induced-pluripotent stem cell-derived retinal Müller glial cells. Glia 69:1679–1693
Cunha-Vaz J, Bernardes R, Lobo C (2011) Blood-retinal barrier. Eur J Ophthalmol 21(Suppl 6):S3-9
Das A, Stroud S, Mehta A, Rangasamy S (2015) New treatments for diabetic retinopathy. Diabetes Obes Metab 17:219–230
Dudakov JA, Hanash AM, van den Brink MR (2015) Interleukin-22: immunobiology and pathology. Annu Rev Immunol 33:747–785
Eyerich K, Dimartino V, Cavani A (2017) IL-17 and IL-22 in immunity: driving protection and pathology. Eur J Immunol 47:607–614
Eynard AR, Repossi G (2019) Role of ω3 polyunsaturated fatty acids in diabetic retinopathy: a morphological and metabolically cross talk among blood retina barriers damage, autoimmunity and chronic inflammation. Lipids Health Dis 18:114
Feng S, Yu H, Yu Y, Geng Y, Li D, Yang C, Lv Q, Lu L, Liu T, Li G, Yuan L (2018) Levels of inflammatory cytokines IL-1β, IL-6, IL-8, IL-17A, and TNF-α in aqueous humour of patients with diabetic retinopathy. J Diabetes Res 2018:8546423
Forrester JV, Kuffova L, Delibegovic M (2020) The role of inflammation in diabetic retinopathy. Front Immunol 11:583687
Fresta CG, Fidilio A, Caruso G, Caraci F, Giblin FJ, Leggio GM, Salomone S, Drago F, Bucolo C (2020) A new human blood-retinal barrier model based on endothelial cells, pericytes, and astrocytes. Int J Mol Sci 21
Fukaya T, Fukui T, Uto T, Takagi H, Nasu J, Miyanaga N, Arimura K, Nakamura T, Koseki H, Choijookhuu N, Hishikawa Y, Sato K (2018) Pivotal role of IL-22 binding protein in the epithelial autoregulation of interleukin-22 signaling in the control of skin inflammation. Front Immunol 9:1418
Guidry C, King JL, Mason JO 3rd (2009) Fibrocontractive Müller cell phenotypes in proliferative diabetic retinopathy. Invest Ophthalmol vis Sci 50:1929–1939
Hossein-Khannazer N, Zian Z, Bakkach J, Kamali AN, Hosseinzadeh R, Anka AU, Yazdani R, Azizi G (2021) Features and roles of T helper 22 cells in immunological diseases and malignancies. Scand J Immunol 93:e13030
Huang JC, Schleisman M, Choi D, Mitchell C, Watson L, Asquith M, Rosenbaum JT (2021) Preliminary report on interleukin-22, GM-CSF, and IL-17F in the pathogenesis of acute anterior uveitis. Ocul Immunol Inflamm 29:558–565
Ito T, Hirose K, Nakajima H (2019) Bidirectional roles of IL-22 in the pathogenesis of allergic airway inflammation. Allergol Int 68:4–8
Jadhav AS, Patil PB, Biradar S (2020) Analysis on diagnosing diabetic retinopathy by segmenting blood vessels, optic disc and retinal abnormalities. J Med Eng Technol 44:299–316
Jensen IJ, Jensen SN, Sjaastad FV, Gibson-Corley KN, Dileepan T, Griffith TS, Mangalam AK, Badovinac VP (2020) Sepsis impedes EAE disease development and diminishes autoantigen-specific naive CD4 T cells. Elife 9
Jia L, Wu C (2014) The biology and functions of Th22 cells. Adv Exp Med Biol 841:209–230
Jiang Q, Yang G, Xiao F, Xie J, Wang S, Lu L, Cui D (2021) Role of Th22 cells in the pathogenesis of autoimmune diseases. Front Immunol 12:688066
Kebir H, Kreymborg K, Ifergan I, Dodelet-Devillers A, Cayrol R, Bernard M, Giuliani F, Arbour N, Becher B, Prat A (2007) Human TH17 lymphocytes promote blood-brain barrier disruption and central nervous system inflammation. Nat Med 13:1173–1175
Keir M, Yi Y, Lu T, Ghilardi N (2020) The role of IL-22 in intestinal health and disease. J Exp Med 217:e20192195
Khalfaoui T, Lizard G, Ouertani-Meddeb A (2008) Adhesion molecules (ICAM-1 and VCAM-1) and diabetic retinopathy in type 2 diabetes. J Mol Histol 39:243–249
Khaloo P, Qahremani R, Rabizadeh S, Omidi M, Rajab A, Heidari F, Farahmand G, Bitaraf M, Mirmiranpour H, Esteghamati A, Nakhjavani M (2020) Nitric oxide and TNF-α are correlates of diabetic retinopathy independent of hs-CRP and HbA1c. Endocrine 69:536–541
Khanh Vu TH, Chen H, Pan L, Cho KS, Doesburg D, Thee EF, Wu N, Arlotti E, Jager MJ, Chen DF (2020) CD4(+) T-cell responses mediate progressive neurodegeneration in experimental ischemic retinopathy. Am J Pathol 190:1723–1734
Kim CJ, Nazli A, Rojas OL, Chege D, Alidina Z, Huibner S, Mujib S, Benko E, Kovacs C, Shin LY, Grin A, Kandel G, Loutfy M, Ostrowski M, Gommerman JL, Kaushic C, Kaul R (2012) A role for mucosal IL-22 production and Th22 cells in HIV-associated mucosal immunopathogenesis. Mucosal Immunol 5:670–680
Kinuthia UM, Wolf A, Langmann T (2020) Microglia and inflammatory responses in diabetic retinopathy. Front Immunol 11:564077
Kusuhara S, Fukushima Y, Ogura S, Inoue N, Uemura A (2018) Pathophysiology of diabetic retinopathy: the old and the new. Diabetes Metab J 42:364–376
Lee D, Jo H, Go C, Jang Y, Chu N, Bae S, Kang D, Kim Y, Kang JS (2022) The roles of IL-22 and its receptor in the regulation of inflammatory responses in the brain. Int J Mol Sci 23
Li D, Zhou J, Yang B, Yu Y (2019) microRNA-340-5p inhibits hypoxia/reoxygenation-induced apoptosis and oxidative stress in cardiomyocytes by regulating the Act1/NF-κB pathway. J Cell Biochem 120:14618–14627
Li J, Tomkinson KN, Tan XY, Wu P, Yan G, Spaulding V, Deng B, Annis-Freeman B, Heveron K, Zollner R, De Zutter G, Wright JF, Crawford TK, Liu W, Jacobs KA, Wolfman NM, Ling V, Pittman DD, Veldman GM, Fouser LA (2004) Temporal associations between interleukin 22 and the extracellular domains of IL-22R and IL-10R2. Int Immunopharmacol 4:693–708
Li J, Yu S, Lu X, Cui K, Tang X, Xu Y, Liang X (2021) The phase changes of M1/M2 phenotype of microglia/macrophage following oxygen-induced retinopathy in mice. Inflamm Res 70:183–192
Liu Y, Yang Z, Lai P, Huang Z, Sun X, Zhou T, He C, Liu X (2020) Bcl-6-directed follicular helper T cells promote vascular inflammatory injury in diabetic retinopathy. Theranostics 10:4250–4264
Luo Y, Dong X, Lu S, Gao Y, Sun G, Sun X (2021) Gypenoside XVII alleviates early diabetic retinopathy by regulating Müller cell apoptosis and autophagy in db/db mice. Eur J Pharmacol 895:173893
Mattapallil MJ, Kielczewski JL, Zárate-Bladés CR, St Leger AJ, Raychaudhuri K, Silver PB, Jittayasothorn Y, Chan CC, Caspi RR (2019) Interleukin 22 ameliorates neuropathology and protects from central nervous system autoimmunity. J Autoimmun 102:65–76
Mayer-Barber KD, Barber DL (2015) Innate and Adaptive cellular immune responses to mycobacterium tuberculosis infection. Cold Spring Harb Perspect Med 5
Mossner S, Kuchner M, Fazel Modares N, Knebel B, Al-Hasani H, Floss DM, Scheller J (2020) Synthetic interleukin 22 (IL-22) signaling reveals biological activity of homodimeric IL-10 receptor 2 and functional cross-talk with the IL-6 receptor gp130. J Biol Chem 295:12378–12397
Mühl H, Bachmann M (2019) IL-18/IL-18BP and IL-22/IL-22BP: two interrelated couples with therapeutic potential. Cell Signal 63:109388
Ouyang H, Du A, Zhou L, Zhang T, Lu B, Wang Z, Ji L (2022) Chlorogenic acid improves diabetic retinopathy by alleviating blood-retinal-barrier dysfunction via inducing Nrf2 activation. Phytother Res 36:1386–1401
Ouyang H, Mei X, Zhang T, Lu B, Ji L (2018) Ursodeoxycholic acid ameliorates diabetic retinopathy via reducing retinal inflammation and reversing the breakdown of blood-retinal barrier. Eur J Pharmacol 840:20–27
Pathak JL, Fang Y, Chen Y, Ye Z, Guo X, Yan Y, Zha J, Liang D, Ke X, Yang L, Zhong W, Wang L, Wang L (2021) Downregulation of macrophage-specific Act-1 intensifies periodontitis and alveolar bone loss possibly via TNF/NF-κB signaling. Front Cell Dev Biol 9:628139
Perez LG, Kempski J, McGee HM, Pelzcar P, Agalioti T, Giannou A, Konczalla L, Brockmann L, Wahib R, Xu H, Vesely MCA, Soukou S, Steglich B, Bedke T, Manthey C, Seiz O, Diercks BP, Gnafakis S, Guse AH, Perez D, Izbicki JR, Gagliani N, Flavell RA, Huber S (2020) TGF-β signaling in Th17 cells promotes IL-22 production and colitis-associated colon cancer. Nat Commun 11:2608
Qiu AW, Bian Z, Mao PA, Liu QH (2016) IL-17A exacerbates diabetic retinopathy by impairing Müller cell function via Act1 signaling. Exp Mol Med 48:e280
Qiu AW, Liu QH, Wang JL (2017) Blocking IL-17A alleviates diabetic retinopathy in rodents. Cell Physiol Biochem 41:960–972
Rezzola S, Guerra J, Krishna Chandran AM, Loda A, Cancarini A, Sacristani P, Semeraro F, Presta M (2021) VEGF-independent activation of müller cells by the vitreous from proliferative diabetic retinopathy patients. Int J Mol Sci 22
Rodgers JM, Zhou L, Miller SD (2010) Act1, scene brain: astrocytes play a lead role. Immunity 32:302–304
Rübsam A, Parikh S, Fort PE (2018) Role of inflammation in diabetic retinopathy. Int J Mol Sci 19
Ruterbusch M, Pruner KB, Shehata L, Pepper M (2020) In Vivo CD4(+) T Cell differentiation and function: revisiting the Th1/Th2 paradigm. Annu Rev Immunol 38:705–725
Ryba-Stanisławowska M, Werner P, Brandt A, Myśliwiec M, Myśliwska J (2016) Th9 and Th22 immune response in young patients with type 1 diabetes. Immunol Res 64:730–735
Saigusa R, Winkels H, Ley K (2020) T cell subsets and functions in atherosclerosis. Nat Rev Cardiol 17:387–401
Sheu A, Chan Y, Ferguson A, Bakhtyari MB, Hawke W, White C, Chan YF, Bertolino PJ, Woon HG, Palendira U, Sierro F, Lau SM (2018) A proinflammatory CD4(+) T cell phenotype in gestational diabetes mellitus. Diabetologia 61:1633–1643
Shi L, Ji Q, Liu L, Shi Y, Lu Z, Ye J, Zeng T, Xue Y, Yang Z, Liu Y, Lu J, Huang X, Qin Q, Li T, Lin YZ (2020) IL-22 produced by Th22 cells aggravates atherosclerosis development in ApoE(-/-) mice by enhancing DC-induced Th17 cell proliferation. J Cell Mol Med 24:3064–3078
Sonnenberg GF, Fouser LA, Artis D (2010) Functional biology of the IL-22-IL-22R pathway in regulating immunity and inflammation at barrier surfaces. Adv Immunol 107:1–29
Sugita S, Kawazoe Y, Imai A, Usui Y, Takahashi M, Mochizuki M (2013) Suppression of IL-22-producing T helper 22 cells by RPE cells via PD-L1/PD-1 interactions. Invest Ophthalmol vis Sci 54:6926–6933
Sun H, Zhong D, Wang C, Sun Y, Zhao J, Li G (2018) MiR-298 exacerbates ischemia/reperfusion injury following ischemic stroke by targeting Act1. Cell Physiol Biochem 48:528–539
Suzuki Y, Adachi K, Maeda N, Tanabu R, Kudo T, Nakazawa M (2020) Proliferative diabetic retinopathy without preoperative pan-retinal photocoagulation is associated with higher levels of intravitreal IL-6 and postoperative inflammation. Int J Retina Vitreous 6:24
Taguchi M, Someya H, Inada M, Nishio Y, Takayama K, Harimoto K, Karasawa Y, Ito M, Takeuchi M (2020) Retinal changes in mice spontaneously developing diabetes by Th17-cell deviation. Exp Eye Res 198:108155
Takeuchi M, Sato T, Sakurai Y, Taguchi M, Harimoto K, Karasawa Y, Ito M (2017) Association between aqueous humor and vitreous fluid levels of Th17 cell-related cytokines in patients with proliferative diabetic retinopathy. PLoS ONE 12:e0178230
Tang J, Kern TS (2011) Inflammation in diabetic retinopathy. Prog Retin Eye Res 30:343–358
Taurone S, Ralli M, Nebbioso M, Greco A, Artico M, Attanasio G, Gharbiya M, Plateroti AM, Zamai L, Micera A (2020) The role of inflammation in diabetic retinopathy: a review. Eur Rev Med Pharmacol Sci 24:10319–10329
Ucgun NI, Zeki-Fikret C, Yildirim Z (2020) Inflammation and diabetic retinopathy. Mol vis 26:718–721
Wang JJ, Zhu M, Le YZ (2015) Functions of Müller cell-derived vascular endothelial growth factor in diabetic retinopathy. World J Diabetes 6:726–733
Wang T, Zhang C, Xie H, Jiang M, Tian H, Lu L, Xu GT, Liu L, Zhang J (2021) Anti-VEGF therapy prevents Müller intracellular edema by decreasing VEGF-A in diabetic retinopathy. Eye vis (lond) 8:13
Wang Y, Liu X, Zhu L, Li W, Li Z, Lu X, Liu J, Hua W, Zhou Y, Gu Y, Zhu M (2020) PG545 alleviates diabetic retinopathy by promoting retinal Müller cell autophagy to inhibit the inflammatory response. Biochem Biophys Res Commun 531:452–458
Wei HX, Wang B, Li B (2020) IL-10 and IL-22 in mucosal immunity: driving protection and pathology. Front Immunol 11:1315
Whitehead M, Osborne A, Widdowson PS, Yu-Wai-Man P, Martin KR (2019) Angiopoietins in diabetic retinopathy: current understanding and therapeutic potential. J Diabetes Res 2019:5140521
Wu H, Wang M, Li X, Shao Y (2021) The metaflammatory and immunometabolic role of macrophages and microglia in diabetic retinopathy. Hum Cell 34:1617–1628
Xie H, Zhang C, Liu D, Yang Q, Tang L, Wang T, Tian H, Lu L, Xu JY, Gao F, Wang J, Jin C, Li W, Xu G, Xu GT, Zhang J (2021) Erythropoietin protects the inner blood-retinal barrier by inhibiting microglia phagocytosis via Src/Akt/cofilin signalling in experimental diabetic retinopathy. Diabetologia 64:211–225
Xu H, Cai M, Zhang X (2015) Effect of the blockade of the IL-23-Th17-IL-17A pathway on streptozotocin-induced diabetic retinopathy in rats. Graefes Arch Clin Exp Ophthalmol 253:1485–1492
Yang W, Chen X, Hu H (2020) CD4(+) T-cell differentiation in vitro. Methods Mol Biol 2111:91–99
Yao Y, Du J, Li R, Zhao L, Luo N, Zhai JY, Long L (2019a) Association between ICAM-1 level and diabetic retinopathy: a review and meta-analysis. Postgrad Med J 95:162–168
Yao Y, Li R, Du J, Long L, Li X, Luo N (2019b) Interleukin-6 and diabetic retinopathy: a systematic review and meta-analysis. Curr Eye Res 44:564–574
Ye Z, Zhao L, Gao Q, Jiang Y, Jiang Z, Chu CQ (2020) Analysis of IL-22 and Th22 cells by flow cytometry in systemic lupus erythematosus. Methods Mol Biol 2108:29–42
Yi C, Yi Y, Wei J, Jin Q, Li J, Sacitharan PK (2021) Targeting IL-22 and IL-22R protects against experimental osteoarthritis. Cell Mol Immunol 18:1329–1331
Zenewicz LA (2021) IL-22 Binding protein (IL-22BP) in the regulation of IL-22 biology. Front Immunol 12:766586
Zhang CJ, Wang C, Jiang M, Gu C, Xiao J, Chen X, Martin BN, Tang F, Yamamoto E, Xian Y, Wang H, Li F, Sartor RB, Smith H, Husni ME, Shi FD, Gao J, Carman J, Dongre A, McKarns SC, Coppieters K, Jørgensen TN, Leonard WJ, Li X (2018) Act1 is a negative regulator in T and B cells via direct inhibition of STAT3. Nat Commun 9:2745
Zhang T, Ouyang H, Mei X, Lu B, Yu Z, Chen K, Wang Z, Ji L (2019) Erianin alleviates diabetic retinopathy by reducing retinal inflammation initiated by microglial cells via inhibiting hyperglycemia-mediated ERK1/2-NF-κB signaling pathway. Faseb j 33:11776–11790
Zhang Y, Wang X, Mao L, Yang D, Gao W, Tian Z, Zhang M, Yang X, Ma K, Wu Y, Ni B (2017) Dual roles of IL-22 at ischemia-reperfusion injury and acute rejection stages of rat allograft liver transplantation. Oncotarget 8:115384–115397
Zhao R, Tang D, Yi S, Li W, Wu C, Lu Y, Hou X, Song J, Lin P, Chen L, Sun L (2014a) Elevated peripheral frequencies of Th22 cells: a novel potent participant in obesity and type 2 diabetes. PLoS ONE 9:e85770
Zhao RX, Li WJ, Lu YR, Qin J, Wu CL, Tian M, He TY, Yi SN, Tang DQ, Sun L, Chen L (2014b) Increased peripheral proinflammatory T helper subsets contribute to cardiovascular complications in diabetic patients. Mediators Inflamm 2014:596967
Zhou L, Chong MM, Littman DR (2009) Plasticity of CD4+ T cell lineage differentiation. Immunity 30:646–655
Zhu X, Zhu J (2020) CD4 T helper cell subsets and related human immunological disorders. Int J Mol Sci 21
Zuo ZF, Zhang Q, Liu XZ (2013) Protective effects of curcumin on retinal Müller cell in early diabetic rats. Int J Ophthalmol 6:422–424
Funding
This study was supported by the China Postdoctoral Science Foundation (No. 2017M612870) and Natural Science Foundation of Liaoning Province (No. 2019-ZD-0807).
Author information
Authors and Affiliations
Contributions
YFW and HDY contributed to experiments, data collection, manuscript writing. JL, WQL and SXY provide help for experimental testing and theoretical support. LPZ, LP and XBW help collect experimental data. XZL and ZFZ contributed to experimental conception, data interpretation and manuscript revision. All authors have read and approved the submission and publication of the final version of manuscript. The authors vouch for the accuracy and completeness of the experiment.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Ethics approval for all animal experiments was obtained from the institutional animal care and use Committee of Jinzhou Medical University [SYXK• [ Liao] •2019–0007].
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Yu, H., Li, J. et al. Th22 cells induce Müller cell activation via the Act1/TRAF6 pathway in diabetic retinopathy. Cell Tissue Res 390, 367–383 (2022). https://doi.org/10.1007/s00441-022-03689-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-022-03689-8