Abstract
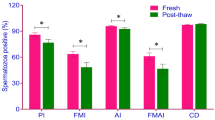
Deciphering sperm transcriptome is the key to understanding the molecular mechanisms governing peri-fertilization, embryonic development, and pregnancy establishment. This study aimed to profile sperm transcriptome to identify signature transcripts regulating male fertility. Semen samples were collected from 47 bulls with varied fertility rates. The sperm total RNA was isolated (n = 8) and subjected to transcriptome sequencing. Based on the expression pattern obtained from RNA profiling, the bulls were grouped (p = 0.03) into high-fertile and sub-fertile, and signature transcripts controlling sperm functions and fertility were identified. The results were validated using the OMIM database, qPCR, and sperm function tests. The sperm contains 1100 to 1700 intact transcripts, of which BCL2L11 and CAPZA3 were abundant and associated (p < 0.05) with spermatogenesis and post-embryonic organ morphogenesis. The upregulated genes in the acrosome integrity and functional membrane integrity groups had a close association with the fertility rate. The biological functions of these upregulated genes (p < 0.05) in the high-fertile bulls were associated with spermatogenesis (AFF4 and BRIP1), sperm motility (AK6 and ATP6V1G3), capacitation and zona binding (AGFG1), embryo development (TCF7 and AKIRIN2), and placental development (KRT19). The transcripts involved in pathways regulating embryonic development such as translation (EEF1B2 and MTIF3, p = 8.87E−05) and nonsense-mediated decay (RPL23 and RPL7A, p = 5.01E−27) were upregulated in high-fertile bulls. The identified transcripts may significantly impact oocyte function, embryogenesis, trophectoderm development, and pregnancy establishment. In addition, the study also reveals that the genes governing sperm functional membrane integrity and acrosome integrity have a prospective effect on male fertility.





Similar content being viewed by others
References
Agarwal A, Mulgund A, Hamada A, Chyatte MR (2015) A unique view on male infertility around the globe. Reprod Biol Endocrinol 13(1):37. https://doi.org/10.1186/s12958-015-0032-1
Akakura S, Gelman IH (2012) Pivotal role of AKAP12 in the regulation of cellular adhesion dynamics: control of cytoskeletal architecture cell migration and mitogenic signaling. J Signal Transduct. https://doi.org/10.1155/2012/529179
Assou S, Haouzi D, Mahmoud K, Aouacheria A, Guillemin Y, Pantesco V, Reme T, Dechaud H, De Vos J, Hamamah S (2008) A non-invasive test for assessing embryo potential by gene expression profiles of human cumulus cells: a proof of concept study. Mol Hum Reprod 14(12):711–719. https://doi.org/10.1093/molehr/gan067
Behm-Ansmant I, Gatfield D, Rehwinkel J, Hilgers V, Izaurralde E (2007) A conserved role for cytoplasmic poly (A)-binding protein 1 (PABPC1) in nonsense-mediated mRNA decay. EMBO J 26(6):1591–1601. https://doi.org/10.1038/sj.emboj.7601588
Bissonnette N, Lévesque-Sergerie JP, Thibault C, Boissonneault G (2009) Spermatozoal transcriptome profiling for bull sperm motility: a potential tool to evaluate semen quality. Reproduction 138(1):65. https://doi.org/10.1530/REP-08-0503
Boumela I, Assou S, Aouacheria A, Haouzi D, Dechaud H, De Vos J, Handyside A, Hamamah S (2011) Involvement of BCL2 family members in the regulation of human oocyte and early embryo survival and death: gene expression and beyond. Reproduction 141(5):549–561. https://doi.org/10.1530/REP-10-0504
Burl RB, Clough S, Sendler E, Estill M, Krawetz SA (2018) Sperm RNA element as markers of health. Syst Biol Reprod Med 64(1):25–38. https://doi.org/10.1080/19396368.2017.1393583
Caballero-Campo P, Buffone MG, Benencia F, Conejo-García JR, Rinaudo PF, Gerton GL (2014) A role for the chemokine receptor CCR6 in mammalian sperm motility and chemotaxis. J Cell Physiol 229(1):68–78. https://doi.org/10.1002/jcp.24418
Dzeja P, Terzic A (2009) Adenylate kinase and AMP signaling networks: metabolic monitoring signal communication and body energy sensing. Int J Mol Sci 10(4):1729–1772. https://doi.org/10.3390/ijms10041729
Eisa AA, De S, Detwiler A, Gilker E, Ignatious AC, Vijayaraghavan S, Kline D (2019) YWHA (14–3-3) protein isoforms and their interactions with CDC25B phosphatase in mouse oogenesis and oocyte maturation. BMC Dev Biol 19(1):20. https://doi.org/10.1186/s12861-019-0200-1
Ekstrand J, Razuvaev A, Folkerse L, Roy J, Hedin U (2010) Tissue factor pathway inhibitor-2 is induced by fluid shear stress in vascular smooth muscle cells and affects cell proliferation and survival. J Vasc Surg 52(1):167–175. https://doi.org/10.1016/j.jvs.2010.02.282
Feugang JM, Rodriguez-Osorio N, Kaya A, Wang H, Page G, Ostermeier GC, Topper EK, Memili E (2010) Transcriptome analysis of bull spermatozoa: implications for male fertility. Reprod Biomed Online 21(3):312–324. https://doi.org/10.1016/j.rbmo.2010.06.022
Friedrich BM, Jülicher F (2007) Chemotaxis of sperm cells. Proc Natl Acad Sci USA 104(33):13256–13261. https://doi.org/10.1073/pnas.0703530104
Grabow S, Kueh AJ, Ke F, Vanyai HK, Sheikh BN, Dengler MA, Chiang W, Eccles S, Smyth IM, Jones LK, De Sauvage FJ (2018) Subtle changes in the levels of BCL-2 proteins cause severe craniofacial abnormalities. Cell Rep 24(12):3285–3295. https://doi.org/10.1016/j.celrep.2018.08.048
Gunes S, Al-Sadaan M, Agarwal A (2015) Spermatogenesis DNA damage DNA repair mechanisms in male infertility. Reprod Biomed Online 31(3):309–319. https://doi.org/10.1016/j.rbmo.2015.06.010
Herrada G, Wolgemuth DJ (1997) The mouse transcription factor Stat4 is expressed in haploid male germ cells is present in the perinuclear theca of spermatozoa. J Cell Sci 110(14):1543–1553
Jodar M, Sendler E, Moskovtsev SI, Librach CL, Goodrich R, Swanson S, Hauser R, Diamond MP, Krawetz SA (2015) Absence of sperm RNA elements correlates with idiopathic male infertility. Sci Trans Med 7(295re6):295re6-296. https://doi.org/10.1126/scitranslmed.aab1287
Kabartan E, Gunes S, Arslan MA, Asci R (2019) Investigating the relationship between BRCA1 and BRCA2 genes methylation profile and sperm DNA fragmentation in infertile men. Andrologia 51(7):e13308. https://doi.org/10.1111/and.13308
Kumar M, Ardekani AM (2019) Effect of external shear flow on sperm motility. Soft Matter 15(31):6269–6277. https://doi.org/10.1039/C9SM00717B
Lalancette C, Thibault C, Bachand I, Caron N, Bissonnette N (2008) Transcriptome analysis of bull semen with extreme nonreturn rate: use of suppression-subtractive hybridization to identify functional markers for fertility. Biol Reprod 78(4):618–635. https://doi.org/10.1095/biolreprod.106.059030
Lange S, S, Tomida J, Boulware KS, Bhetawal S, Wood RD, (2016) The polymerase activity of mammalian DNA pol zeta is specifically required for cell and embryonic viability. PLoS Genet 12(1):e1005759. https://doi.org/10.1371/journal.pgen.1005759
Liu G, Shi QW, Lu GX (2010) A newly discovered mutation in PICK1 in a human with globozoospermia. Asian J Androl 12(4):556–560. https://doi.org/10.1038/aja.2010.47
Merrick BA, Phadke DP, Auerbach SS, Mav D, Stiegelmeyer SM, Shah RR, Tice RR (2013) RNA-Seq profiling reveals novel hepatic gene expression pattern in aflatoxin B1 treated rats. PLoS ONE 8(4):e61768. https://doi.org/10.1371/journal.pone.0061768
Miller D (2015) Confrontation consolidation and recognition: the oocyte’s perspective on the incoming sperm. Cold Spring Harb Perspect Med 5(8):a023408. https://doi.org/10.1101/cshperspect.a023408
Morita M, Ler LW, Fabian MR, Siddiqui N, Mullin M, Henderson VC, Alain T, Fonseca BD, Karashchuk G, Bennett CF, Kabuta T (2012) A novel 4EHP-GIGYF2 translational repressor complex is essential for mammalian development. Mol Cell Biol 32(17):3585–3593. https://doi.org/10.1128/MCB.00455-12
Moye AR, Bedoni N, Cunningham JG, Sanzhaeva U, Tucker ES, Mathers P, Peter VG, Quinodoz M, Paris LP, Coutinho-Santos L, Camacho P (2019) Mutations in ARL2BP a protein required for ciliary microtubule structure cause syndromic male infertility in humans and mice. PLoS Genet 15(8):e1008315. https://doi.org/10.1371/journal.pgen.1008315
Mühlemann O (2016) Spermatogenesis studies reveal a distinct nonsense-mediated mRNA decay (NMD) mechanism for mRNAs with long 3′ UTRs. PLoS Genet 12(5):e1005979. https://doi.org/10.1371/journal.pgen.1005979
Ostermeier GC, Dix DJ, Miller D, Khatri P, Krawetz SA (2002) Spermatozoal RNA profiles of normal fertile men. Lancet 360(9335):772–777. https://doi.org/10.1016/S0140-6736(02)09899-9
Ostermeier GC, Miller D, Huntriss JD, Diamond MP, Krawetz SA (2004) Delivering spermatozoan RNA to the oocyte. Nature 429(6988):154–154. https://doi.org/10.1038/429154a
Parthipan S, Selvaraju S, Somashekar L, Kolte AP, Arangasamy A, Ravindra JP (2015) Spermatozoa input concentrations and RNA isolation methods on RNA yield and quality in bull (Bos taurus). Ana Biochem 482:32–39. https://doi.org/10.1016/j.ab.2015.03.022
Qu HM, Qu LP, Pan XZ, Mu LS (2018) Upregulated miR-222 targets BCL2L11 and promotes apoptosis of mesenchymal stem cells in preeclampsia patients in response to severe hypoxia. Int J Clin Exp Pathol 11(1):110
Rando OJ (2012) Daddy issues: paternal effects on phenotype. Cell 151(4):702–708. https://doi.org/10.1016/j.cell.2012.10.020
Ren X, Chen X, Wang Z, Wang D (2017) Is transcription in sperm stationary or dynamic? J Reprod Develop 63(5):439–443. https://doi.org/10.1262/jrd.2016-093
Selvaraju S, Parthipan S, Somashekar L, Kolte AP, Binsila BK, Arangasamy A, Ravindra JP (2017) Occurrence and functional significance of the transcriptome in bovine (Bos taurus) spermatozoa. Sci Rep 7:42392. https://doi.org/10.1038/srep42392
Selvaraju S, Ravindra J, Ghosh J, Gupta P, Suresh K (2008) Evaluation of sperm functional attributes in relation to in vitro sperm-zona pellucida binding ability and cleavage rate in assessing frozen thawed buffalo (Bubalus bubalis) semen quality. Anim Reprod Sci 106(3–4):311–321. https://doi.org/10.1016/j.anireprosci.2007.05.005
Selvaraju S, Sivasubramani T, Raghavendra B, Raju P, Rao S, Dineshkumar D, Ravindra JP (2012) Effect of dietary energy on seminal plasma insulin-like growth factor-I (IGF-I) serum IGF-I and testosterone levels semen quality and fertility in adult rams. Theriogenology 78(3):646–655. https://doi.org/10.1016/j.theriogenology.2012.03.010
Shawki HH, Kigoshi T, Katoh Y, Matsuda M, Ugboma CM, Takahashi S, Oishi H, Kawashima A (2016) Identification localization and functional analysis of the homologues of mouse CABS1 protein in porcine testis. Exp Anim 65(3):253–265. https://doi.org/10.1538/expanim.15-0104
Shen YR, Wang HY, Kuo YC, Shih SC, Hsu CH, Chen YR, Wu SR, Wang CY, Kuo PL (2017) SEPT12 phosphorylation results in loss of the septin ring/sperm annulus defective sperm motility and poor male fertility. PLoS Genet 13(3):e1006631. https://doi.org/10.1371/journal.pgen.1006631
Somashekar L, Selvaraju S, Parthipan S, Patil S, Binsila B, Venkataswamy M, Karthik Bhat S, Ravindra JP (2017) Comparative sperm protein profiling in bulls differing in fertility and identification of phosphatidylethanolamine-binding protein 4 a potential fertility marker. Andrology 5(5):1032–1051. https://doi.org/10.1111/andr.12404
Sosnik J, Buffone M, G, Visconti P, E, (2010) Analysis of CAPZA3 localization reveals temporally discrete events during the acrosome reaction. J Cell Physiol 224(3):575–580. https://doi.org/10.1002/jcp.22211
Sutovsky P, Manandhar G, McCauley TC, Caamaño JN, Sutovsky M, Thompson WE, Day BN (2004) Proteasomal interference prevents zona pellucida penetration and fertilization in mammals. Biol Reprod 71(5):1625–1637. https://doi.org/10.1095/biolreprod.104.032532
Vera M, Pani B, Griffiths LA, Muchardt C, Abbott CM, Singer RH, Nudler E (2014) The translation elongation factor eEF1A1 couples transcription to translation during heat shock response. Elife 3:e03164. https://doi.org/10.7554/eLife.03164
Wickliffe KE, Williamson A, Meyer HJ, Kelly A, Rape M (2011) K11-linked ubiquitin chains as novel regulators of cell division. Trends Cell Biol 21(11):656–663. https://doi.org/10.1016/j.tcb.2011.08.008
Winet H, Bernstein GS, Head J (1984) Observations on the response of human spermatozoa to gravity, boundaries and fluid shear. Reproduction 70(2):511–523. https://doi.org/10.1530/jrf.0.070051
Yeung C, Cooper T, Nieschlag E (1997) Human epididymal secreted protein CD52 on ejaculated spermatozoa: correlations with semen characteristics and the effect of its antibody. Mol Hum Reprod 3(12):1045–1051. https://doi.org/10.1093/molehr/3.12.1045
Yu B, Zhang T, Xia P, Gong X, Qiu X, Huang J (2018) CCDC134 serves a crucial role in embryonic development. Int J Mol Med 41(1):381–390. https://doi.org/10.3892/ijmm.2017.3196
Zhang Q, Liang X, Zhu B, Dong Q, Xu L, Xia L, Hu J, Fu J, Liu M (2006) Effects of fluid shear stress on mRNA expression of carbonic anhydrase II in polarized rat osteoclasts. Cell Biol Int 30(9):714–720. https://doi.org/10.1016/j.cellbi.2006.05.002
Zhu F, Yan P, Zhang J, Cui Y, Zheng M, Cheng Y, Guo Y, Yang X, Guo X, Zhu H (2019) Deficiency of TPPP2, a factor linked to oligoasthenozoospermia, causes subfertility in male mice. J cell Mol Med 23(4):2583–2594. https://doi.org/10.1111/jcmm.14149
Acknowledgements
The authors sincerely acknowledge Dr. Raghavendra Bhatta, Director, ICAR-NIANP, Bengaluru, India, for his critical technical inputs, drafting the manuscript and providing necessary facilities to carry out this work. The authors are grateful to Dr. J.P. Ravindra, Former Head, Animal Physiology Division, ICAR-NIANP, for his technical contribution and proofreading of the manuscript. The authors are also thankful to Dr. Maharajan Lavanya for critical reviewing of the manuscript.
Funding
This research was carried out under the ICAR-National Fellow project funded by the Indian Council of Agricultural Research, Government of India. RNA seq data was generated as a part of DBT project (PR3587) funded by the Department of Biotechnology, Government of India. Dr. S. Selvaraju is supported by the ICAR-National Fellow project, ICAR, Ministry of Agriculture, Government of India.
Author information
Authors and Affiliations
Contributions
Conceived, designed the experiment, and bioinformatics analysis: SS, LR, SP, APK, and DS. Performed the experiments: SS, SP, LR, and DS. LR, DS, SP, and APK, and BBK analyzed and drafted the manuscript. All authors: reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animal experiments performed by any of the authors. The experiment does not require Institutional Animal Ethics committee approval, since the experiment was conducted in fresh and frozen semen samples purchased from the commercial semen bank. However, collecting the semen samples and data, all applicable Institutional Animal Ethics guidelines for the care of animals were followed.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Selvaraju, S., Ramya, L., Parthipan, S. et al. Deciphering the complexity of sperm transcriptome reveals genes governing functional membrane and acrosome integrities potentially influence fertility. Cell Tissue Res 385, 207–222 (2021). https://doi.org/10.1007/s00441-021-03443-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-021-03443-6