Abstract
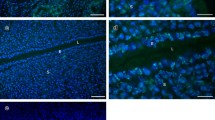
For the development of uterine receptivity, many morphological and molecular changes occur in the apical surface of luminal uterine epithelial cells (UECs) including an increase in vesicular activity. Vesicular movements for exocytosis and endocytosis are dependent on microtubules; however, changes in microtubules in UECs during early pregnancy have received little attention. β-tubulin, one of the main component of microtubules, is distributed throughout the cytoplasm of UECs on day 1 (non-receptive) of pregnancy in the rat. On day 5.5, β-tubulin is concentrated above the nuclei and by day 6 (receptive), β-tubulin is concentrated in a band-like fashion above the nucleus. Western blot analysis of isolated UECs found two bands (50 and 34 kDa) for β-tubulin in UECs during early pregnancy. The intensity of the 34 kDa band was significantly higher on day 6 compared to day 1. The increase in the 34 kDa band may be due to higher proteolytic activity associated with microtubule polymerisation during the receptive state. Transmission electron microscopy showed fragmented microtubules at the time of receptivity in UECs. This is the first study to show that microtubules are reorganised during uterine receptivity. This re-organisation likely facilitates vesicular movement and promotes the reorganisation of the apical plasma membrane for uterine receptivity.







Similar content being viewed by others
References
Allen RD, Weiss DG, Hayden JH et al (1985) Gliding movement of and bidirectional transport along single native microtubules from squid axoplasm: evidence for an active role of microtubules in cytoplasmic transport. J Cell Biol 100:1736–1752
Aplin JD (1997) Adhesion molecules in implantation. Rev Reprod 2:84–93. https://doi.org/10.1530/ror.0.0020084
Baas PW, Karabay A, Qiang L (2005) Microtubules cut and run. Trends Cell Biol 15:518–524. https://doi.org/10.1016/j.tcb.2005.08.004
Baas PW, Vidya Nadar C, Myers KA (2006) Axonal transport of microtubules: the long and short of it. Traffic 7:490–498. https://doi.org/10.1111/j.1600-0854.2006.00392.x
Bicek AD, Tüzel E, Demtchouk A et al (2009) Anterograde microtubule transport drives microtubule bending in LLC-PK1 epithelial cells. Mol Biol Cell 20:2943–2953. https://doi.org/10.1091/mbc.E08-09-0909
Cole NB, Lippincott-Schwartz J (1995) Organization of organelles and membrane traffic by microtubules. Curr Opin Cell Biol 7:55–64. https://doi.org/10.1016/0955-0674(95)80045-X
Denker HW (1990) Trophoblast-endometrial interactions at embryo implantation: a cell biological paradox. Troph Res 4:3–29
Horio T, Murata T (2014) The role of dynamic instability in microtubule organization. Front Plant Sci 5:511. https://doi.org/10.3389/fpls.2014.00511
Jolly AL, Gelfand VI (2010) Cytoplasmic microtubule sliding. Commun Integr Biol 3:589–591. https://doi.org/10.4161/cib.3.6.13212
Kraemer J, Schmitz F, Drenckhahn D (1999) Cytoplasmic dynein and dynactin as likely candidates for microtubule-dependent apical targeting of pancreatic zymogen granules. Eur J Cell Biol 78:265–277. https://doi.org/10.1016/S0171-9335(99)80060-0
Lodish H, Berk A, Zipursky S L, Matsudaira P (2000) Molecular cell biology, 4th edition
Lunam CA, Murphy CR (1983) Alterations in microvilli of uterine epithelial cells after colchicine treatment. Z Mikrosk Anat Forsch 97:1005–1008
Mandelkow E, Thomas J, Cohen C (1977) Microtubule structure at low resolution by x-ray diffraction. Proc Natl Acad Sci U S A 74:3370–3374
Mitchison T, Kirschner M (1984) Dynamic instability of microtubule growth. Nature 312:237–242. https://doi.org/10.1038/312237a0
Moore CL, Cheng D, Shami GJ, Murphy CR (2016) Correlated light and electron microscopy observations of the uterine epithelial cell actin cytoskeleton using fluorescently labeled resin-embedded sections. Micron 84:61–66. https://doi.org/10.1016/j.micron.2016.02.010
Muroyama A, Lechler T (2017) Microtubule organization, dynamics and functions in differentiated cells. Development 144:3012–3021. https://doi.org/10.1242/dev.153171
Murphy C (1994) Plasma membrane transformation: a common response of uterine epithelial cells during the peri-implantation period. Cell Biol Int 18:1115–1128. https://doi.org/10.1006/cbir.1994.1038
Murphy CR (1993) The plasma membrane of uterine epithelial cells: structure and Histochemistry. Prog Histochem Cytochem 27:1–66. https://doi.org/10.1016/S0079-6336(11)80004-5
Murphy CR (2004) Uterine receptivity and the plasma membrane transformation. Cell Res 14:259–267. https://doi.org/10.1038/sj.cr.7290227
Murphy CR, Martin B (1985) Cholesterol in the plasma membrane of uterine epithelial cells: a freeze-fracture cytochemical study with digitonin. J Cell Sci 78:163–172
Murphy CR, Martin B (1987) Digitonin cytochemistry reveals cholesterol-rich vesicles in uterine epithelial cells. Acta Histochem 81:143–147. https://doi.org/10.1016/S0065-1281(87)80003-X
Müsch A (2004) Microtubule organization and function in epithelial cells. Traffic 5:1–9. https://doi.org/10.1111/j.1600-0854.2003.00149.x
Ng YH, Rome S, Jalabert A et al (2013) Endometrial Exosomes/microvesicles in the uterine microenvironment: a new paradigm for embryo-endometrial cross talk at implantation. PLoS One 8:e58502. https://doi.org/10.1371/journal.pone.0058502
Noordstra I, Akhmanova A (2017) Linking cortical microtubule attachment and exocytosis. F1000Research 6:469. Doi: https://doi.org/10.12688/f1000research.10729.1
Oriolo AS, Wald FA, Ramsauer VP, Salas PJI (2007) Intermediate filaments: a role in epithelial polarity. Exp Cell Res 313:2255–2264. https://doi.org/10.1016/j.yexcr.2007.02.030
Parr MB (1980) Endocytosis at the basal and lateral membranes of rat uterine epithelial cells during early pregnancy. J Reprod Fertil 60:95–99
Parr MM (1982) Apical vesicles in the rat uterine epithelium during early pregnancy: a morphometric study. Biol Reprod 26:915–924
Pelham HR (2001) SNAREs and the specificity of membrane fusion. Trends Cell Biol 11:99–101
Png FY, Murphy CR (2000) Closure of the uterine lumen and the plasma membrane transformation do not require blastocyst implantation. Eur J Morphol 38:122–127. https://doi.org/10.1076/0924-3860(200004)38:2;1-F;FT122
Roll-Mecak A, McNally FJ (2010) Microtubule-severing enzymes. Curr Opin Cell Biol 22:96–103. https://doi.org/10.1016/j.ceb.2009.11.001
Sackett DL, Wolff J (1986) Proteolysis of tubulin and the substructure of the tubulin dimer. J Biol Chem 261:9070–9076
Schnapp BJ, Vale RD, Sheetz MP, Reese TS (1985) Single microtubules from squid axoplasm support bidirectional movement of organelles. Cell 40:455–462. https://doi.org/10.1016/0092-8674(85)90160-6
Serrano L, Wandosell F, De J et al (1988) Effect of specific proteolytic cleavages on tubulin polymer formation. Biochem J 252:683–691
Serrano L, Wandosell F, de la Torre J, Avila J (1986) Proteolytic modification of tubulin. Methods Enzymol 134:179–190. https://doi.org/10.1016/0076-6879(86)34087-4
Sharp DJ, Ross JL (2012) Microtubule-severing enzymes at the cutting edge. J Cell Sci 125:2561–2569. https://doi.org/10.1242/jcs.101139
Sheetz MP, Vale R, Schnapp B et al (1987) Movements of vesicles on microtubules. Ann N Y Acad Sci 493:409–416. https://doi.org/10.1111/j.1749-6632.1987.tb27227.x
Shekhar N, Neelam S, Wu J et al (2013) Fluctuating motor forces bend growing microtubules. Cell Mol Bioeng 6:120–129. https://doi.org/10.1007/s12195-013-0281-z
Slautterback DB (1963) Cytoplasmic microtubules. I Hydra J Cell Biol 18:367–388. https://doi.org/10.1083/jcb.18.2.367
Tai AW, Chuang JZ, Bode C et al (1999) Rhodopsin’s carboxy-terminal cytoplasmic tail acts as a membrane receptor for cytoplasmic dynein by binding to the dynein light chain Tctex-1. Cell 97:877–887
Wacławek E, Włoga D (2011) Microtubule severing proteins—structure and regulation of activity. Postepy Biochem 62
Welte MA (2004) Bidirectional transport along microtubules. Curr Biol 14:R525–R537. https://doi.org/10.1016/j.cub.2004.06.045
Yeaman C, Grindstaff KK, Nelson WJ (1999) New perspectives on mechanisms involved in generating epithelial cell polarity. Physiol Rev 79:73–98. https://doi.org/10.1002/(sici)1097-4644(19960616)61:4<514::aid-jcb4>3.0.co
Yoon Y, Torok N, Krueger E et al (1998) Ethanol-induced alterations of the microtubule cytoskeleton in hepatocytes. Am J Phys 274:G757–G766
Acknowledgements
The authors acknowledge the support and facilities of the Bosch Institute Advanced Microscopy Facility, The University of Sydney and the assistance provided by Dr. Louise Cole. The authors also acknowledge the support and facilities provided by the Bosch Institute Molecular Biology Facility and the scientific and technical assistance of the Australian Microscopy & Microanalysis Research Facility at the Australian Centre for Microscopy & Microanalysis, The University of Sydney. Financial support was provided by the Australian Research Council, The Ann Macintosh Foundation of the Discipline of Anatomy and Histology and the Murphy Laboratory.
Author information
Authors and Affiliations
Contributions
S.N.K. designed the study, performed all experiments, analysed data and took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Rights and permissions
About this article
Cite this article
Kalam, S.N., Dowland, S., Lindsay, L. et al. Microtubules are reorganised and fragmented for uterine receptivity. Cell Tissue Res 374, 667–677 (2018). https://doi.org/10.1007/s00441-018-2887-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-2887-x