Abstract
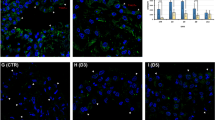
Following Freund’s adjuvant-induced acute inflammation, the regeneration of rat mesothelium is accompanied by the reduction of cell organelles. The aim of the present study is to test whether autophagy may play a role in the recovery process of mesothelial cells by eliminating accumulated cell organelles and also to investigate the presence of potential inducers and molecular transmitters of the process. Control and treated (from day 2 to day 11; D2–D11) mesothelial cells (n = 16 samples/group) obtained from male rats were isolated and phenotypically characterized. Morphological studies included light and electron microscopy. Biochemical studies performed on tissue samples as well as isolated cells were used to evaluate the dynamics of autophagy and also to detect the expression levels of TNF-α, LC3B, estrogen receptors (ER-α and GPR30) and Erk1/2. Gene expression was measured by individual Taqman assays on quantitative RT-PCR. Protein expression study was performed by Western blotting and immunolabeling. Estradiol concentration was measured both in peritoneal fluid and plasma samples in control and treated animals (n = 3–10 animals per group). Our conventional electron microscopic and morphometric results showed a progressive autophagosome formation with a peak by the termination of inflammation (D5). Subsequently, autophagolysosome formation dominated between D6 and D8 with a concomitant expression of LC3B proved by immunoblotting. We further observed the reduction of cell compartments by D11 parallel with the morphological restitution of mesothelium. Estradiol showed a sustained level in the peritoneal fluid but not in plasma samples between D3 and D11 compared to levels obtained from untreated animals. The mRNA expression of TNF-α was increased between D2 and D11 compared to control. Western blot analysis showed a constitutive expression of GPR30, while ER-α could not be detected between D6 and D11. Erk1/2 was activated by phosphorylation with a peak at D6. Considering our present in vivo results, we hypothesize that the facilitated autophagy might play an important role in the removal of cytoplasmic organelles during the recovery of mesothelium, while our results also suggest that the detected peritoneal estradiol as well as TNF-α may contribute to this process.








Similar content being viewed by others
References
Balogh P, Szabó A, Katz S, Likó I, Patócs A, Kiss AL (2013) Estrogen receptor alpha is expressed in mesenteric mesothelial cells and is internalized in caveolae upon Freund’s adjuvant treatment. PLoS ONE 11, e79508
Barbati C, Pierdominici M, Gambardella L, LbediMF KHR, Rosano G, Malorni W, Ortona E (2012) Cell surface estrogen receptor alpha is upregulated during subchronic metabolic stress and inhibits neuronal cell degeneration. PLoS ONE. doi:10.1371/journal.pone.0042339
Bayard F, Clamens S, Delsol G, Blaes N, Maret A, Faye JC (1995) Oestrogen biosynthesis, oestrogen metabolism and functional oestrogen receptors in bovine aortic endothelial cells. Ciba Found Symp 191:122–132
Beato M, Herrlich P, Schutz G (1995) Steroid hormon receptors: many actors insearch of a plot. Cell 83:851–857
Bi R, Broutman G, Foy MR, Thompson RF, Baudry M (2000) The tyrosine kinase and mitogen-activated protein kinase pathways mediate multiple effects of estrogen in hippocampus. Proc Natl Acad Sci U S A 97:3602–3607
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brailoiu E, Dun SL, Brailoiu GC, Mizuo K, Sklar LA, Oprea TI, Prossnitz ER, Kawata M (2007) Expression of GPR30, a G protein-coupled membrane estrogen receptor, in oxytocin neurons of the rat paraventricular and supraoptic nuclei. Endocrinology 148:4853–4864
Bruch HR, Wolf L, Budde R, Romalo G, Schweikert HU (1992) Androstenedione metabolism in cultured human osteoblast-like cells. J Clin Endocrinol Metab 75:101–105
Chakrabati S, Davidge ST (2012) G-protein coupled receptor 30 (GPR30): A novel regulator of endothelial inflammation. PLoS ONE 7(12), e52357
Cheng Y, Qiu F, Tashiro S, Onodera S, Ikejima T (2008) ERK and JNK mediate TNFalpha-induced p53 activation in apoptotic and autophagic L929 cell death. Biochem Biophys Res Commun 376:483–488
Choi AJS, Ryter SW (2011) Autophagy in inflammatory diseases. Int J Cell Biol. doi:10.1155/2011/732798
Corcelle E, Nebout M, Bekri S, Gauthier N, Hofman P, Poujeol P, Fenichel P, Mograbi B (2006) Disruption of autophagy at the maturation step by the carcinogen lindane is associated with the sustained mitogen-activated proein kinase/extracellular signal-regulated kinase activity. Cancer Res 66:6861–6870
Corcelle E, Djerbi N, Mari M, Nebout M, Fiorini C et al (2007) Control of the autophagy maturation step by the MAPK ERK and p38: lessons from environmental carcinogens. Autophagy 3:57–59
Deretic V, Saitoh T, Akira S (2013) Autophagy in infection, inflammation and immunity. Nat Rev Immunol 13:722–737
Djavaheri-Mergny M, Amelotti M, Mathieu J, Besançon F, Bauvy C, Souquère S, Pierron G, Codogno P (2006) NF-kappaB activation represses tumor necrosis factor-alpha-induced autophagy. J Biol Chem 281:30373–30382
Fang CC, Yen CJ, Chen YM, Shyu RS, Tsai TJ, Lee PH, Hsieh BS (2000) Pentoxifylline inhibits human peritoneal mesothelial cell growth and collagen synthesis: Effects on TGF-β. Nature 57:2626–2633
Filardo EJ, Quinn JA, Bland KI, Frackelton AR Jr (2000) Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol Endocrinol 14:1649–1660
Gajewska M, Zielniok K, Motyl T (2013) Autophagy in development and remodelling of mammary gland. In: Bailly Y (ed) Autophagy – A double-edged sword-cell survival or death? InTech Chapter 20
Keller CW, Fokken C, Turville SG, Lünemann A, Schmidt J, Münz C, Lünemann JD (2011) TNF-α induces macroautophagy and regulates MHC class II expression in human skeletal muscle cells. J Biol Chem 286:3970–3980
Kilpatrick LE, SunS MD, Baik F, Li H, Korchak HM (2006) Regulation of TNF mediated antiapoptotic signaling in human neutrophils: role of delta-PKC and ERK1/2. J Leukoc Biol 80:1512–1521
Klionsky DJ (2007) Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol 8:931
Kroemer G, Marino G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40:280–293
Labrie F, Belanger A, Cusan L, Candas B (1997a) Physiological changes in dehydroepiandrosterone are not reflected by serum levels of active androgens and estrogens but of their metabolites: intracrinology. J Clin Endocrinol Metab 82:2403–2409
Labrie F, Belanger A, Cusan L, Gomez JL, Candas B (1997b) Marked decline serum concentrations of adrenal C19 sex steroid precursors and conjugated androgen metabolites during aging. J Clin Endocrinol Metab 82:2396–2402
Labrie F, Belanger A, Luu-The V, Labrie C, Simond J, Cusan L, Gomez JL, Candas B (1998) DHEA and the intracrine formation of androgens and estrogens in peripheral target tissues: its role during aging. Steroids 63:322–328
Levin ER (2009) Plasma membrane estrogen receptors. Trends Endocrinol Metab 20:477–482
Levine B, Klionsy DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6:463–477
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132:27–42
Levine B, Mizushima N, Virgin HW (2011) Autophagy in immunity and inflammation. Nature 469:323–335
Mauvais-Jarvis F (2012) Estrogen sulfotransferase: Intracrinology meets metabolic diseases. Diabetes 61:1353–1354
Migliaccio A, Di Domenico M, Castoria G, de Falco A, Bontempo B et al (1996) Tyrosine kinase/p21 ras/MAP-kinase pathway activation by estradiol-receptor complex in MCF-7 cells. EMBO J 15:1292–1300
Mizushima N (2005) The pleitotropic role of autophagy: from protein metabolism to bactericide. Cell Death Differ 12(Suppl 2):1535–1542
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873
Mizushima N, Komatsu M (2011) Autophagy: Renovation of cells and tissues. Cell 147:728–741
Mizushima M, Yoshimori T (2007) How to interpret LC3 immunoblotting. Autophagy 3(6):542–545
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy figths disease through cellular self-digestioin. Nature 451:1069–1075
Morita T, Mayanagi T, Sobue K (2007) Dual roles of myocardin-related transcription factors in epithelial-mesenchymal transition via slug induction and actin remodeling. J Cell Biol 179:1027–1042
Morris PG, Hudis CA, Giri D, Morrow M, Falcone DJ, Zhou XK, Du B, Brogi E, Crawford CB, Kopelovich L, Subbaramaiah K, Dannenberg AJ (2011) Inflammation and increased aromatase expression occur in the breast tissue of obese women with breast cancer. Cancer Prev Res 4:1021–1029. doi:10.1158/1940-6207.CAPR-11-0110
Murakami H, Sasano H, Satoh A, Satomi S, Nagura H, Harada N (1998) Aromatase in human aortic tissue. 79th Annual Meeting of the Endocrine Society, Minneapolis, MN, USA p 212 (Abstract)
Nelson DR, Koymans L, Kamataki T, Stegeman JJ, Feyereisen R, Waxman DJ, Waterman MR, Gotoh O, Coon MJ, Estabrook RW, Gunsalus IC, Nebert DW (1996) P450 superfamily: update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics 6:1–42
Ohshiro K, Schwartz AM, Levine PH, Kumar R (2012) Alternate estrogen receptor promote invasion of inflammatory breast cancer cells via non-genomic signaling. PLoS ONE 7(1), e30725
Ohsumi Y (2001) Molecular dissection of autophagy: two ubiquitin-like systems. Nat Rev Mol Cell Biol 2:211–216
Prossnitz ER, Oprea TI, Sklar LA, Arterburn JB (2008) The ins and outs of GPR30: A transmembrane estrogen receptor. J Steroid Biochem Mol Biol 109:350–353
Razandi M, Pedram A, Greene GL, Levin ER (1999) Cell membrane and nuclear estrogen receptors derive from a single transcript: studies of ERα and ERβ expressed in CHO cells. Mol Endocrinol 13:307–319
Simoncini T, Mannella P, Fornari L, Caruso A, Varone G, Genazzani AR (2004) Genomic and non-genomic effects of estrogens on endothelial cells. Steroids 69:537–542
Simpson E, Rubin G, Clyne C, Robertson K, O’Donnell L, Davis S, Jones M (1999) Local estrogen biosynthesis in males and females. Endocr Relat Cancer 6:131–137
Simpson ER, Rubin G, Clyne C, Robertson K, O’Donnell L, Jones M, Davis S (2000) The role of local estrogen byosynthesis in males and females. Trends Endocrinol Metab 11:184–188
Song RX, Santen RJ (2006) Membrane initiated estrogen signaling in breast cancer. Biol Reprod 75:9–16
Song RX, Robert A, McPherson LA, Yongde B et al (2002) Linkage of rapid estrogen action to MAPK activation by ERα-Shc association and Shc pathway activation. Mol Endocrinol 16:116–127
Sridharan S, Jain K, Basu A (2011) Regulation of autophagy by kinases. Cancers 3:2630–2654
Thomas P, Pang Y, Filardo EJ, Dong J (2005) Identity of an estrogen membrane receptor coupled to a G-protein in human breast cancer cells. Endocrinology 146:624–632
Tsai MJ, O’Malley BW (1994) Molecular mechanism of action of steroid/thyroid receptor superfamily members. Annu Rev Biochem 63:451–486
Vague J (1981) Sardo Aromatization of androgens (author's transl). Sem Hop 57:1467–1476
Weibel ER, Kiseler GS, Scherle WF (1966) Practical stereological method for morphometric cytology. J Cell Biol 30:23–38
Xie Z, Klionsky DJ (2007) Autophagosome formation: core machinery and adaptations. Nat Cell Biol 9:1102–1109
Yang YH, Chen K, Li B, Chen JW, Zheng XF, Wang YR, Jiang SD, Jiang LS (2013) Estradiol inhibits osteoblast apoptosis via promotion of autophagy through the ER-ERK-mTOR pathway. Apoptosis 18:1363–1375
Acknowledgments
We would like to express our thankfulness to Professor Pál Röhlich for carefully reviewing and lecturing the manuscript. Thanks to Dr. Márk Oláh who generously offered anti-pErk1/2 antibody for the experiments. Thanks are also due to Katalin Lőcsey and Nikoletta Dóczi for their valuable technical help. The authors acknowledge the financial support from the Hungarian Scientific Research Fund [OTKA, PD100648 (AP)] and Technology Innovation Fund, National Developmental Agency (KTIA-AIK-2012-12-1-0010). The authors highly value the useful critical comments of the reviewers by which the quality of the manuscript was greatly enhanced.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balogh, P., Szabó, A., Likó, I. et al. Autophagy may contribute to the recovery of rat mesothelium following acute inflammation in vivo. Cell Tissue Res 362, 127–137 (2015). https://doi.org/10.1007/s00441-015-2188-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-015-2188-6