Abstract
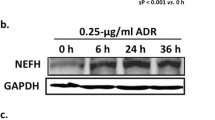
Optineurin, a cytosolic protein associated with the actin cytoskeleton, microtubules, and the Golgi complex, appears to have an important function in neurons, as mutations in its gene are causative for neurodegenerative diseases such as primary open-angle glaucoma and amyotrophic lateral sclerosis. Here, we report that optineurin is localized in podocytes of the kidney and induced upon injury following treatment with puromycin aminonucleoside. In cultured human podocytes, optineurin localizes to the Golgi complex. Optineurin depletion by RNA interference causes Golgi fragmentation. Moreover, if the Golgi complex is fragmented following microtubule destabilization induced by nocodazole treatment, optineurin dissociates from Golgi vesicles. Furthermore, optineurin colocalizes with vinculin-labeled focal contacts of cultured podocytes and with lysosome-like structures. Optineurin is essential for the survival of cultured podocytes, as optineurin depletion causes cell death. Thus, optineurin appears to play an important role in the maintenance of the podocyte Golgi complex and in the trafficking of vesicles to focal contacts and lysosomes.










Similar content being viewed by others
References
Akizuki M, Yamashita H, Uemura K, Maruyama H, Kawakami H, Ito H, Takahashi R (2013) Optineurin suppression causes neuronal cell death via NF-κB pathway. J Neurochem 126:699–704
Caviston JP, Ross JL, Antony SM, Tokito M, Holzbaur EL (2007) Huntingtin facilitates dynein/dynactin-mediated vesicle transport. Proc Natl Acad Sci U S A 104:10045–10050
De Marco N, Buono M, Troise F, Diez-Roux G (2006) Optineurin increases cell survival and translocates to the nucleus in a Rab8-dependent manner upon an apoptotic stimulus. J Biol Chem 281:16147–16156
Del Toro D, Alberch J, Lázaro-Diéguez F, Martín-Ibáñez R, Xifró X, Egea G, Canals JM (2009) Mutant huntingtin impairs post-Golgi trafficking to lysosomes by delocalizing optineurin/Rab8 complex from the Golgi apparatus. Mol Biol Cell 20:1478–1492
Geiger B (1979) A 130 K protein from chicken gizzard: its localization at the termini of microfilament bundles in cultured chicken cells. Cell 18:193–205
Gleason CE, Ordureau A, Gourlay R, Arthur JS, Cohen P (2011) Polyubiquitin binding to optineurin is required for optimal activation of TANK-binding kinase 1 and production of interferon β. J Biol Chem 286:35663–35674
Hattula K, Peränen J (2000) FIP-2, a coiled-coil protein, links Huntingtin to Rab8 and modulates cellular morphogenesis. Curr Biol 10:1603–1606
Iida H, Shibata Y (1991) Functional Golgi units in microtubule-disrupted cultured atrial myocytes. J Histochem Cytochem 39:1349–1355
Kachaner D, Filipe J, Laplantine E, Bauch A, Bennett KL, Superti-Furga G, Israël A, Weil R (2012a) Plk1-dependent phosphorylation of optineurin provides a negative feedback mechanism for mitotic progression. Mol Cell 45:553–566
Kachaner D, Génin P, Laplantine E, Weil R (2012b) Toward an integrative view of optineurin functions. Cell Cycle 11:2808–2818
Kobayashi N (2002) Mechanism of the process formation; podocytes vs. neurons. Microsc Res Tech 57:217–223
Krishnamoorthy RR, Crawford MJ, Chaturvedi MM, Jain SK, Aggarwal BB, Al-Ubaidi MR, Agarwal N (1999) Photo-oxidative stress down-modulates the activity of nuclear factor-kappaB via involvement of caspase-1, leading to apoptosis of photoreceptor cells. J Biol Chem 274:3734–3743
Kriz W, Shirato I, Nagata M, LeHir M, Lemley KV (2013) The podocyte’s response to stress: the enigma of foot process effacement. Am J Physiol Renal Physiol 304:F333–F347
Li Y, Kang J, Horwitz MS (1998) Interaction of an adenovirus E3 14.7-kilodalton protein with a novel tumor necrosis factor alpha-inducible cellular protein containing leucine zipper domains. Mol Cell Biol 18:1601–1610
Li X, Magenheimer BS, Xia S, Johnson T, Wallace DP, Calvet JP, Li R (2008) A tumor necrosis factor-alpha-mediated pathway promoting autosomal dominant polycystic kidney disease. Nat Med 14:863–868
Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y, Kinoshita Y, Kamada M, Nodera H, Suzuki H, Komure O, Matsuura S, Kobatake K, Morimoto N, Abe K, Suzuki N, Aoki M, Kawata A, Hirai T, Kato T, Ogasawara K, Hirano A, Takumi T, Kusaka H, Hagiwara K, Kaji R, Kawakami H (2010) Mutations of optineurin in amyotrophic lateral sclerosis. Nature 465:223–226
Mundel P, Heid HW, Mundel TM, Krüger M, Reiser J, Kriz W (1997) Synaptopodin: an actin-associated protein in telencephalic dendrites and renal podocytes. J Cell Biol 139:193–204
Quigley HA (2011) Glaucoma. Lancet 377:1367–1377
Rezaie T, Child A, Hitchings R, Brice G, Miller L, Coca-Prados M, Héon E, Krupin T, Ritch R, Kreutzer (2002) Adult-onset primary open-angle glaucoma caused by mutations in optineurin. Science 295:1077–1079
Rezaie T, Waitzman DM, Seeman JL, Kaufman PL, Sarfarazi M (2005) Molecular cloning and expression profiling of optineurin in the rhesus monkey. Invest Ophthalmol Vis Sci 46:2404–2410
Rowland LP, Shneider NA (2001) Amyotrophic lateral sclerosis. N Engl J Med 344:1688–1700
Sahlender DA, Roberts RC, Arden SD, Spudich G, Taylor MJ, Luzio JP, Kendrick-Jones J, Buss F (2005) Optineurin links myosin VI to the Golgi complex and is involved in Golgi organization and exocytosis. J Cell Biol 169:285–295
Saleem MA, O’Hare MJ, Reiser J, Coward RJ, Inward CD, Farren T, Xing CY, Ni L, Mathieson PW, Mundel P (2002) A conditionally immortalized human podocyte cell line demonstrating nephrin and podocin expression. J Am Soc Nephrol 13:630–638
Schiwek D, Endlich N, Holzman L, Holthöfer H, Kriz W, Endlich K (2004) Stable expression of nephrin and localization to cell-cell contacts in novel murine podocyte cell lines. Kidney Int 66:91–101
Schwamborn K, Weil R, Courtois G, Whiteside ST, Israël A (2000) Phorbol esters and cytokines regulate the expression of the NEMO-related protein, a molecule involved in a NF-kappa B-independent pathway. J Biol Chem 275:22780–22789
Sippl C, Bosserhoff AK, Fischer D, Tamm ER (2011) Depletion of optineurin in RGC-5 cells derived from retinal neurons causes apoptosis and reduces the secretion of neurotrophins. Exp Eye Res 93:669–680
Sudhakar C, Nagabhushana A, Jain N, Swarup G (2009) NF-kappaB mediates tumor necrosis factor alpha-induced expression of optineurin, a negative regulator of NF-kappaB. PLoS ONE 4:e5114
Tufro A, Veron D (2012) VEGF and podocytes in diabetic nephropathy. Semin Nephrol 32:385–393
Visa N, Percipalle P (2010) Nuclear functions of actin. Cold Spring Harb Perspect Biol 2:a000620
Vittitow J, Borrás T (2002) Expression of optineurin, a glaucoma-linked gene, is influenced by elevated intraocular pressure. Biochem Biophys Res Commun 298:67–74
Whiteside CI, Cameron R, Munk S, Levy J (1993) Podocytic cytoskeletal disaggregation and basement-membrane detachment in puromycin aminonucleoside nephrosis. Am J Pathol 142:1641–1653
Wild P, Farhan H, McEwan DG, Wagner S, Rogov VV, Brady NR, Richter B, Korac J, Waidmann O, Choudhary C, Dötsch V, Bumann D, Dikic I (2011) Phosphorylation of the autophagy receptor optineurin restricts Salmonella growth. Science 333:228–233
Zhu G, Wu CJ, Zhao Y, Ashwell JD (2007) Optineurin negatively regulates TNFalpha-induced NF-kappaB activation by competing with NEMO for ubiquitinated RIP. Curr Biol 17:1438–1443
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sippl, C., Zeilbeck, L.F., Fuchshofer, R. et al. Optineurin associates with the podocyte Golgi complex to maintain its structure. Cell Tissue Res 358, 567–583 (2014). https://doi.org/10.1007/s00441-014-1968-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-1968-8