Abstract
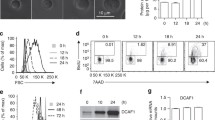
SEPTIN9 (SEPT9) is a filament-forming protein involved in numerous cellular processes. We have used a conditional knock out allele of Sept9 to specifically delete Sept9 in T-cells. As shown by fluorescence-activated cell sorting, loss of Sept9 at an early thymocyte stage in the thymus results in increased numbers of double-negative cells indicating that SEPT9 is involved in the transition from the double-negative stage during T-cell development. Accordingly, the relative numbers of mature T-cells in the periphery are decreased in mice with a T-cell-specific deletion of Sept9. Proliferation of Sept9-deleted CD8+ T-cells from the spleen is decreased upon stimulation in culture. The altered T-cell homeostasis caused by the loss of Sept9 results in an increase of CD8+ central memory T-cells.






Similar content being viewed by others
References
Ammirati E, Cianflone D, Vecchio V, Banfi M, Vermi AC, De Metrio M, Grigore L, Pellegatta F, Pirillo A, Garlaschelli K, Manfredi AA, Catapano AL, Maseri A, Palini AG, Norata GD (2012) Effector memory T cells are associated with atherosclerosis in humans and animal models. J Am Heart Assoc 1:27–41
Billadeau DD, Nolz JC, Gomez TS (2007) Regulation of T-cell activation by the cytoskeleton. Nat Rev Immunol 7:131–143
Crotzer VL, Blum JS (2009) Autophagy and its role in MHC-mediated antigen presentation. J Immunol 182:3335–3341
Dent J, Kato K, Peng XR, Martinez C, Cattaneo M, Poujol C, Nurden P, Nurden A, Trimble WS, Ware J (2002) A prototypic platelet septin and its participation in secretion. Proc Natl Acad Sci USA 99:3064–3069
Estey MP, Di Ciano-Oliveira C, Froese CD, Bejide MT, Trimble WS (2010) Distinct roles of septins in cytokinesis: SEPT9 mediates midbody abscission. J Cell Biol 191:741–749
Foulds KE, Zenewicz LA, Shedlock DJ, Jiang J, Troy AE, Shen H (2002) Cutting edge: CD4 and CD8 T cells are intrinsically different in their proliferative responses. J Immunol 168:1528–1532
Füchtbauer A, Lassen LB, Jensen AB, Howard J, Quiroga Ade S, Warming S, Sørensen AB, Pedersen FS, Füchtbauer EM (2011) Septin9 is involved in septin filament formation and cellular stability. Biol Chem 392:769–777
Hall PA, Russell SE (2004) The pathobiology of the septin gene family. J Pathol 204:489–505
Hall PA, Russell SE (2012) Mammalian septins: dynamic heteromers with roles in cellular morphogenesis and compartmentalization. J Pathol 226:287–299
Hall PA, Jung K, Hillan KJ, Russell SE (2005) Expression profiling the human septin gene family. J Pathol 206:269–278
Hall PA, Russell SEH, Pringle JR (2008) The septins. Wiley, New York
Hartwell LH (1971) Genetic control of the cell division cycle in yeast. IV. Genes controlling bud emergence and cytokinesis. Exp Cell Res 69:265–276
Hu Q, Milenkovic L, Jin H, Scott MP, Nachury MV, Spiliotis ET, Nelson WJ (2010) A septin diffusion barrier at the base of the primary cilium maintains ciliary membrane protein distribution. Science 329:436–439
Ihara M, Kinoshita A, Yamada S, Tanaka H, Tanigaki A, Kitano A, Goto M, Okubo K, Nishiyama H, Ogawa O, Takahashi C, Itohara S, Nishimune Y, Noda M, Kinoshita M (2005) Cortical organization by the septin cytoskeleton is essential for structural and mechanical integrity of mammalian spermatozoa. Dev Cell 8:343–352
Ihara M, Yamasaki N, Hagiwara A, Tanigaki A, Kitano A, Hikawa R, Tomimoto H, Noda M, Takanashi M, Mori H, Hattori N, Miyakawa T, Kinoshita M (2007) Sept4, a component of presynaptic scaffold and Lewy bodies, is required for the suppression of alpha-synuclein neurotoxicity. Neuron 53:519–533
Kim MS, Froese CD, Estey MP, Trimble WS (2011) SEPT9 occupies the terminal positions in septin octamers and mediates polymerization-dependent functions in abscission. J Cell Biol 195:815–826
Kinoshita M, Field CM, Coughlin ML, Straight AF, Mitchison TJ (2002) Self- and actin-templated assembly of mammalian septins. Dev Cell 3:791–802
Kuhlenbaumer G, Hannibal MC, Nelis E, Schirmacher A, Verpoorten N, Meuleman J, Watts GD, De Vriendt E, Young P, Stogbauer F, Halfter H, Irobi J, Goossens D, Del-Favero J, Betz BG, Hor H, Kurlemann G, Bird TD, Airaksinen E, Mononen T, Serradell AP, Prats JM, Van Broeckhoven C, De Jonghe P, Timmerman V, Ringelstein EB, Chance PF (2005) Mutations in SEPT9 cause hereditary neuralgic amyotrophy. Nat Genet 37:1044–1046
Kuhn R, Schwenk F, Aguet M, Rajewsky K (1995) Inducible gene targeting in mice. Science 269:1427–1429
Laouar A, Manocha M, Haridas V, Manjunath N (2008) Concurrent generation of effector and central memory CD8 T cells during vaccinia virus infection. PLoS One 3:e4089
McMurray MA, Thorner J (2009) Reuse, replace, recycle. Specificity in subunit inheritance and assembly of higher-order septin structures during mitotic and meiotic division in budding yeast. Cell Cycle 8:195–203
Mostowy S, Cossart P (2012) Septins: the fourth component of the cytoskeleton. Nat Rev Mol Cell Biol 13:183–194
Mostowy S, Bonazzi M, Hamon MA, Tham TN, Mallet A, Lelek M, Gouin E, Demangel C, Brosch R, Zimmer C, Sartori A, Kinoshita M, Lecuit M, Cossart P (2010) Entrapment of intracytosolic bacteria by septin cage-like structures. Cell Host Microbe 8:433–444
Orban PC, Chui D, Marth JD (1992) Tissue- and site-specific DNA recombination in transgenic mice. Proc Natl Acad Sci USA 89:6861–6865
Roseler S, Sandrock K, Bartsch I, Busse A, Omran H, Loges NT, Zieger B (2011) Lethal phenotype of mice carrying a Sept11 null mutation. Biol Chem 392:779–781
Russell SE, Hall PA (2011) Septin genomics: a road less travelled. Biol Chem 392:763–767
Sellin ME, Holmfeldt P, Stenmark S, Gullberg M (2011) Microtubules support a disk-like septin arrangement at the plasma membrane of mammalian cells. Mol Biol Cell 22:4588–4601
Sirajuddin M, Farkasovsky M, Hauer F, Kuhlmann D, Macara IG, Weyand M, Stark H, Wittinghofer A (2007) Structural insight into filament formation by mammalian septins. Nature 449:311–315
Sørensen AB, Lund AH, Ethelberg S, Copeland NG, Jenkins NA, Pedersen FS (2000) Sint1, a common integration site in SL3-3-induced T-cell lymphomas, harbors a putative proto-oncogene with homology to the septin gene family. J Virol 74:2161–2168
Sørensen AB, Warming S, Fuchtbauer EM, Pedersen FS (2002) Alternative splicing, expression, and gene structure of the septin-like putative proto-oncogene Sint1. Gene 285:79–89
Suzuki G, Harper KM, Hiramoto T, Sawamura T, Lee M, Kang G, Tanigaki K, Buell M, Geyer MA, Trimble WS, Agatsuma S, Hiroi N (2009) Sept5 deficiency exerts pleiotropic influence on affective behaviors and cognitive functions in mice. Hum Mol Genet 18:1652–1660
Takada K, Jameson SC (2009) Naive T cell homeostasis: from awareness of space to a sense of place. Nat Rev Immunol 9:823–832
Tooley AJ, Gilden J, Jacobelli J, Beemiller P, Trimble WS, Kinoshita M, Krummel MF (2009) Amoeboid T lymphocytes require the septin cytoskeleton for cortical integrity and persistent motility. Nat Cell Biol 11:17–26
Ware J, Martínez C, Zieger B (2008) Septins and platelets. In: Hall PA, Russell SEH, Pringle JR (eds)The septins. Wiley, New York, pp 269–280
Acknowledgments
The authors thank Lone Højgaard Nielsen for technical assistance, and Charlotte Christie Petersen, Rodrigo Labouriau and Bo Porse for advice regarding the set-up and analysis of experiments. Flow cytometry/cell sorting was performed at the FACS Core Facility, The Faculty of Health Sciences, Aarhus University, Denmark.
Author information
Authors and Affiliations
Corresponding author
Additional information
Finn Skou Pedersen and Ernst-Martin Füchtbauer equally contributed to this work.
This study was supported in part by grants from the Danish Cancer Society, the Danish Medical Research Council, the Novo Nordisk Foundation, and the Danish Genetically Modified Animal Resource (DAGMAR) funded by the Danish Agency for Science, Technology, and Innovation.
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lassen, L.B., Füchtbauer, A., Schmitz, A. et al. Septin9 is involved in T-cell development and CD8+ T-cell homeostasis. Cell Tissue Res 352, 695–705 (2013). https://doi.org/10.1007/s00441-013-1618-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-013-1618-6