Abstract
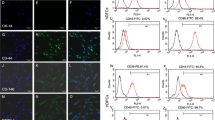
During tooth root formation, dental follicle cells (DFCs) differentiate into osteoblasts/cementoblasts when they are in contact with pre-existing dentin. Since some factors of dentin matrix were also produced by dental papilla cells (DPCs) and could induce DFCs differentiation, we hypothesized that DPCs can directly promote DFCs differentiation and that differentiation could occur in a co-culture model. To test this hypothesis, we investigated the characteristics of DFCs that are influenced by DPCs in an in vitro co-culture and in vivo heterotopic transplant model. One week into the co-culture, there were significant increases in the mRNA level of bone morphogenetic protein 2 (BMP2), osteoprotegerin (OPG), bone sialoprotein (BSP) and osteocalcin (OCN), and a decrease of the receptor activator of nuclear factor κB ligand (RANKL). Additionally, the number of BMP2-, OPG-, BSP- and OCN-positive DFCs increased whereas RANKL-positive DFCs decreased. Three weeks after co-culture, DFCs produced calcified nodules, accompanied with increased sub-cellular organelles for protein synthesis and secretion. In the heterotopic transplant model, the adult male rats were used as hosts, DFCs were transplanted into the omentum. In vivo 5-week growth of DFCs in the presence of DPCs led to the formation of bone-like tissues, positive for BSP, OCN and BMP2. In contrast, DFCs alone led to fibrous-like tissues. These results indicated that in the absence of pre-existing dentin, DPCs can stimulate osteogenesis and inhibit osteoclastogenesis in DFCs and suggested a novel strategy to promote DFCs differentiation.




Similar content being viewed by others
References
Arany S, Nakata A, Kameda T, Koyota S, Ueno Y, Sugiyama T (2006) Phenotype properties of a novel spontaneously immortalized odontoblast-lineage cell line. Biochem Biophys Res Commun 342:718–724
Balint E, Lapointe D, Drissi H, van der Meijden C, Young DW, van Wijnen AJ, Stein JL, Stein GS, Lian JB (2003) Phenotype discovery by gene expression profiling: mapping of biological processes linked to BMP-2-mediated osteoblast differentiation. J Cell Biochem 89:401–426
Bergeron E, Senta H, Mailloux A, Park H, Lord E, Faucheux N (2009) Murine preosteoblast differentiation induced by a peptide derived from bone morphogenetic proteins-9. Tissue Eng Part A 15:3341–3349
Butler WT, Brunn JC, Qin C, Mckee MD (2002) Extracellular matrix proteins and the dynamics of dentin formation. Connect Tissue Res 43:301–307
Canalis E, Economides AN, Gazzerro E (2003) Bone morphogenetic proteins, their antagonists, and the skeleton. Endocr Rev 24:218–235
Chen D, Zhao M, Mundy GR (2004) Bone morphogenetic proteins. Growth Factors 22:233–241
Fonseca JE (2008) Rebalancing bone turnover in favour of formation with strontium ranelate: implications for bone strength. Rheumatology (Oxford) 47:17–19
Ganss B, Kim RH, Sodek J (1999) Bone sialoprotein. Crit Rev Oral Biol Med 10:79–98
Katagiri T, Yamaguchi A, Komaki M, Abe E, Takahashi N, Ikeda T, Rosen V, Wozney JM, Fujisawa-Sehara A, Suda T (1994) Bone morphogenetic protein-2 converts the differentiation pathway of C2C12 myoblasts into the osteoblast lineage. J Cell Biol 127:1755–1766
Kémoun P, Laurencin-Dalicieux S, Rue J, Farges JC, Gennero I, Conte-Auriol F, Briand-Mesange F, Gadelorge M, Arzate H, Narayanan AH, Brunel G (2007) Human dental follicle cells acquire cementoblast features under stimulation by BMP-2/-7 and enamel matrix derivatives (EMD) in vitro. Cell Tissue Res 329:283–294
Kin T, Korbutt GS, Rajotte RV (2003) Survival and metabolic function of syngeneic rat islet grafts transplanted in the omental pouch. Am J Transplant 3:281–285
Korbutt GS, Elliott JF, Rajotte RV (1997) Cotransplantation of allogeneic islets with allogeneic testicular cell aggregates allows long-term graft survival without systemic immunosuppression. Diabetes 46:317–322
Kong YY, Yoshida H, Sarosi I, Tan HL, Timms E, Capparelli C, Morony S, Oliveira-dos-Santos AJ, Van G, Itie A, Khoo W, Wakeham A, Dunstan CR, Lacey DL, Mak TW, Boyle WJ, Penninger JM (1999) OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397:315–323
LeDouarin NM, Dupin E, Ziller C (1994) Genetic and epigenetic control in neural crest development. Curr Opin Gen Dev 4:685–695
Lee MK, Choi H, Gil M, Nikodem VM (2006) Regulation of osteoblast differentiation by Nurr1 in MC3T3-E1 cell line and mouse calvarial osteoblasts. J Cell Biochem 99:986–994
Luan X, Ito Y, Dangaria S, Diekwisch TGH (2006) Dental follicle progenitor cell heterogeneity in the developing mouse periodontium. Stem Cells Dev 15:595–608
Lumsden AGS (1988) Spatial organization of the epithelium and the role of neural crest cells in the initiation of the mammalian tooth germ. Development 103(suppl):155–169
Ma HJ, Yue XZ, Wang DG, Li CR, Zhu WY (2006) A modified method for purifying amelanotic melanocytes from human hair follicles. J Dermatol 33:239–248
Marie PJ (2007) Strontium ranelate: New insights into its dual mode of action. Bone 40:S5–8
Mizuno N, Ozeki Y, Shiba H, Kajiya M, Nagahara T, Takeda K, Kawaguchi H, Abiko Y, Kurihara H (2008) Humoral factors released from human periodontal ligament cells influence calcification and proliferation in human bone marrow mesenchymal stem cells. J Periodontol 79:2361–2370
Morsczeck C, Götz W, Schierholz J, Zeilhofer F, Kühn U, Möhl C, Sippel C, Hoffmann KH (2005) Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol 24:155–165
Nishimura M, Suzuki S, Satoh T, Naito S (2009) Tissue-specific mRNA expression profiles of human solute carrier 35 transporters. Drug Metab Pharmacokinet 24:91–99
Pispa J, Thesleff I (2003) Mechanisms of ectodermal organogenesis. Dev Biol 262:195–205
Rickard DJ, Kassem M, Hefferan TE, Sarkar G, Spelsberg TC, Riggs BL (1996) Isolation and characterization of osteoblast precursor cells from human bone marrow. J Bone Miner Res 11:312–324
Ripamonti U, Renton L (2006) Bone morphogenetic proteins and the induction of periodontal tissue regeneration. Periodontol 2000 41: 73–87
Saito M, Handa K, Kiyono T, Hattori S, Yokoi T, Tsubakimoto T, Harada H, Noguchi T, Toyoda M, Sato S, Teranaka T (2005) Immortalization of cementoblast progenitor cells with Bmi-1 and TERT. J Bone Miner Res 20:50–57
Saugspier M, Felthaus O, Viale-Bouroncle S, Driemel O, Reichert TE, Schmalz G, Morsczeck C (2010) The differentiation and gene expression profile of human dental follicle cells. Stem Cells Dev 19:707–717
Silva TA, Rosa AL, Lara VS (2004) Dentin matrix proteins and soluble factors: intrinsic regulatory signals for healing and resorption of dental and periodontal tissues? Oral Dis 10:63–74
Song AM, Shu R, Xie YF, Song ZC, Li HY, Liu XF, Zhang XL (2007) A study of enamel matrix proteins on differentiation of porcine bone marrow stromal cells into cementoblasts. Cell Prolif 40:381–396
Sun ZL, Fang DN, Wu XY, Ritchie HH, Bègue-Kirn C, Wataha JC, Hanks CT, Butler WT (1998) Expression of dentin sialoprotein (DSP) and other molecular determinants by a new cell line from dental papillae, MDPC-23. Connect Tissue Res 37:251–261
Takagi M (2005) Cell processing engineering for ex-vivo expansion of hematopoietic cells. J Biosci Bioeng 99:189–196
Thesleff I, Mikkol M (2002) The role of growth factors in tooth development. Int Rev Cytol Suppl 217:93–135
Wise GE, Frazier-Bowers S, D-Souza RN (2002) Cellular, molecular, and genetic determinants of tooth eruption. Crit Rev Oral Biol Med 13:323–334
Wise GE, Lin F, Fan W (1992) Culture and characterization of dental follicle cells from rat molars. Cell Tissue Res 267:483–492
Yamaguchi A, Ishizuya T, Kintou N, Wada Y, Katagiri T, Wozney JM, Rosen V, Yoshiki S (1996) Effects of BMP2, BMP4 and BMP6 on osteoblastic differentiation of bone marrow-derived stromal cell lines, ST2 and MC3T3-G2/PA6. Biochem Biophys Res Commun 220:366–371
Yao S, Pan F, Prpic V, Wise GE (2008) Differentiation of stem cells in the dental follicle. J Dent Res 87:767–771
Yokoi T, Saito M, Kiyono T, Iseki S, Kosaka K, Nishida E, Tsubakimoto T, Harada H, Eto K, Noguchi T, Teranaka T (2007) Establishment of immortalized dental follicle cells for generating periodontal ligament in vivo. Cell Tissue Res 327:301–311
Yokose S, Kakokura H, Tajima Y, Fujieda K, Katayama I, Matsuoka T, Katayama T (2000) Establishment and characterization of a culture system for enzymatically released rat dental pulp cells. Calcif Tissue Int 66:139–144
Young CS, Abukawa H, Asrican R, Ravens M, Troulis MJ, Kaban LB, Vacanti JP, Yelick PC (2005) Tissue-engineered hybrid tooth and bone. Tissue Eng 11:1599–1610
Yu J, Deng Z, Shi J, Zhai H, Nie X, Zhuang H, Li Y, Jin Y (2006) Differentiation of dental pulp stem cells into regular-shaped dentin-pulp complex induced by tooth germ cell conditioned medium. Tissue Eng 12:3097–3105
Zhang QX, Magovern CJ, Mack CA, Budenbender KT, Ko W, Rosengart TK (1997) Vascular endothelial growth factor is the major angiogenic factor in omentum: mechanism of the omentum-mediated angiogenesis. J Surg Res 67:147–154
Zhao M, Xiao G, Berry JE, Franceschi RT, Reddi A, Somerman MJ (2002) Bone morphogenetic protein 2 induces dental follicle cells to differentiate toward a cementoblast/osteoblast phenotype. J Bone Miner Res 17:1441–1451
Acknowledgments
We are grateful to Dr. Takashi Muramatsu and Yasunobu Enokiya (Department of Pathology, Tokyo Dental College) for kindly providing BMP2 primer and antibody. We are also grateful to Dr. Fumiko Fukuhara, Dr. Sachie Sennzui (Department of Pediatric Dentistry, Tokyo Dental College), Mr. Tatokoro and Ms. Takano for advice and technology help in the experiments.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yudi Bai and Yuxiang Bai contributed equally to this work.
This work was supported partly by the Sasagawa Fellowship.
Rights and permissions
About this article
Cite this article
Bai, Y., Bai, Y., Matsuzaka, K. et al. Formation of bone-like tissue by dental follicle cells co-cultured with dental papilla cells. Cell Tissue Res 342, 221–231 (2010). https://doi.org/10.1007/s00441-010-1046-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-1046-9