Abstract
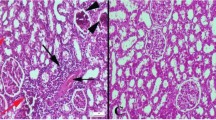
Phenoloxidase (PO) activity was examined in the tunic tissue of Ciona intestinalis following lipopolysaccharide (LPS) intratunic injection. Tunic homogenate supernatant (THS), assayed with the Dopa-MBTH reaction, displayed Ca2+-independent PO activity that was raised by LPS and further enhanced by proteases. Specific inhibitors (tropolone, phenylthiourea, diethylthiocarbamate) supported the specificity of the reaction. Assay with soybean trypsin inhibitor showed that, in the tunic, PO activation with trypsin was not significantly inhibited suggesting that proteases diverse from serine proteases were involved. In vivo experiments were carried out by injecting isosmotic medium or LPS, and THS was assayed for its PO activity. Analysis of variance of the time-course profiles showed that LPS was more effective in activating proPO. To disclose the PO response at the injured site, an assay with Dopa-MBTH was performed in vitro. Quinones were mainly contained in the tunic matrix enriched with inflammatory cells around the injection site. Microscopic observations and immunohistochemistry with anti-CinPO-2 antibodies showed granulocytes and unilocular refractile granulocytes containing PO, whereas few morula cells were stained. In THS zymograms (SDS-polyacrylamide gel electrophoresis), PO activity linked to 90-kDa and 120-kDa bands was observed as an effect of LPS injection, whereas the density of 170-kDa PO was weak. A third presumptive PO enzyme (CinPO-3) containing the CinPO-2 peptide was identified in the recent Ciona genome version. Presumably, LPS stimulated the production and dimerization (120 kDa) of CinPO-3 (66 kDa). Thus, the activated proPO system includes several POs that are distinguishable by size and that are contained and presumably released by tunic inflammatory cells and hemocytes of the pharynx bars.




Similar content being viewed by others
References
Arizza V, Cammarata M, Tomasino MC, Parrinello N (1995) Phenoloxidase characterization in vacuolar hemocytes from the solitary ascidians Styela plicata. J Invert Pathol 66:297–302
Aspan A, Söderhäll K (1991) Purification of prophenoloxidase from crayfish blood cells and its activation by an endogeneous serine proteinase. Insect Biochem 21:363–373
Aspan A, Sturtevant J, Smith VJ, Söderhäll K (1990) Purification and characterization of a prophenoloxidase activating enzyme from crayfish blood cells. Insect Biochem 20:709–718
Aspan A, Huang TS, Cerenius L, Söderhäll K (1995) cDNA cloning of prophenoloxidase from the freshwater crayfish Pacifastacus leniusculus and its activation. Proc Natl Acad Sci USA 92:939–943
Ballarin L, Cima F, Sabbadin A (1994) Phenoloxidase in the colonial ascidian Botryllus schlosseri (Urochordata, Ascidiacea). Anim Biol 3:41–48
Ballarin L, Cima F, Sabbadin A (1996) Morula cells and histocompatibility in the colonial ascidian Botryllus schlosseri. Zool Sci 12:757–764
Ballarin L, Cima F, Floreani M, Sabbadin A (2002) Oxidative stress induces cytotoxicity during rejection reaction in the compound ascidian Botryllus schlosseri. Comp Biochem Physiol C Toxicol Pharmacol 133:411–418
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cammarata M, Arizza V, Vazzana M, Parrinello N (1996) Prophenoloxidase activating system in tunicate hemolymph. It J Zool 63:345–351
Cammarata M, Arizza V, Candore G, Caruso C, Parrinello N (1997) Phenoloxidase-dependent cytotoxic mechanism in ascidian Styela plicata hemocyte active against erythrocytes and K562 tumour cells. Eur J Cell Biol 74:302–307
Cerenius L, Söderhäll K (2004) The prophenoloxidase-activating system in invertebrates. Immunol Rev 198:116–126
De Leo G, Parrinello N, Di Bella MA (1987) Fine structure of blood system in Ciona intestinalis (Tunicata). Vessel and hemocytes in pharyngeal wall. Arch Biol 98:35–52
Dunnett CW (1955) A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc 50:1096–1121
Ermak TH (1976) The hematogenic tissues of tunicates. In: Wright RK, Cooper EL (eds) Phylogeny of thymus and bone marrow-bursa cells. Elsevier/North-Holland, Amsterdam, pp 45–56
Fuke MT (1980) “Contact reactions” between xenogeneic or allogeneic coelomic cells of solitary ascidians. Biol Bull 158:304–315
Hata S, Azumi K, Yokosawa H (1998) Ascidian phenoloxidase: its release from hemocytes, isolation, characterization and physiological roles. Comp Biochem Physiol [B] Biochem Mol Biol 119:769–776
Immesberger A, Burmester T (2004) Putative phenoloxidases in the tunicate Ciona intestinalis and the origin of the arthropod hemocyanin superfamily. J Comp Physiol [B] 174:169–180
Jackson AD, Smith VJ, Peddie CM (1993) In vitro phenoloxidase activity in the blood of Ciona intestinalis and other ascidians. Dev Comp Immunol 17:97–108
Johansson MW, Söderhäll K (1989) Cellular immunity in crustaceans and proPO system. Parasitol Today 5:171–176
Kahn V (1985) Tropolone a compound that can aid in differentiating between tyrosinase and peroxidase. Biochemistry 24:915–920
Kelly K, Cooper EL, Raftos DA (1992) Purification and characterization of a humoral opsonin from the solitary urochordate Styela clava. Comp Biochem Physiol [B] 103:749–753
Klabunde T, Eicken C, Sacchettini JC, Krebs B (1998) Crystal structure of a plant catechol oxidase containing a dicopper center. Nat Struct Biol 5:1084–1090
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of Bacteriophage T4. Nature 227:680–685
Leonard C, Söderhäll K, Ratcliffe NA (1985) Studies on prophenoloxidase and protease activity of Blaberus craniifer. Insect Biochem 15:803–810
Ling E, Shirai K, Kanehatsu R, Kiguchi K (2005) Reexamination of phenoloxidase in larval circulating hemocytes of the silkworm Bombyx mori. Tissue Cell 37:101–107
Millar RH (1953) Ciona. LMBC Mem Typ Br Mar Plants Anim 35:1–123
Nappi AJ, Ottaviani E (2000) Cytotoxicity and cytotoxic molecules in invertebrates. Bioessays 22:469–480
Nappi AJ, Seymur J (1991) Hemolyph phenoloxidases in Drosophila melanogaster, Locusta migratoria and Austropotamobius pallipes. Biochem Biophys Res Comun 180:748–754
Nappi AJ, Vass E (1993) Melanogenesis and the generation of cytotoxic molecules during insect cellular immune reactions. Pigment Cell Res 6:117–126
Nellaiappan K, Vinayagam A (1986) A rapid method for detection of tyrosinase activity in electrophoresis. Stain Technol 61:269–272
Pang Q, Zhang S, Wang C, Shi X, Sun Y (2004) Presence of prophenoloxidase in the humoral fluid of amphioxus Branchiostoma belcheri tsingtauense. Fish Shellfish Immunol 17:477–487
Parrinello N (1981) The reaction of Ciona intestinalis L. to subcuticular erythrocyte and protein injection. Dev Comp Immunol 5:105–110
Parrinello N, Patricolo E, Canicattí C (1984a) Inflammatory-like reaction in the tunic of Ciona intestinalis (Tunicata). Encapsulation and tissue injury I. Biol Bull 167:229–237
Parrinello N, Patricolo E, Canicattí C (1984b) Inflammatory-like reaction in the tunic of Ciona intestinalis (Tunicata). Encapsulation tissue injury II. Biol Bull 167:238–250
Parrinello N, De Leo G, Di Bella MA (1990) Fine structural observations of the granulocytes involved in the tunic inflammatory-like reaction of Ciona intestinalis (Tunicata). J Invert Pathol 56:181–189
Parrinello N, Cammarata M, Vazzana M, Arizza V, Vizzini A, Cooper EL (2001) Immunological activity of ascidian hemocytes. In: Yokosawa H, Lambert CC (eds) The Biology of Ascidians. Springer, Tokyo, pp 395–401
Parrinello N, Arizza V, Chinnici C, Parrinello D, Cammarata M (2003) Phenoloxidases in ascidian hemocytes: characterization of the pro-phenoloxidase activating system. Comp Biochem Physiol [B] Biochem Mol Biol 135:583–591
Parrinello N, Arizza V, Cammarata M, Giaramita FT, Pergolizzi M, Vazzana M, Vizzini A, Parrinello D (2007) Inducible lectins with galectin properties and human IL1α epitopes opsonize yeasts in the ascidian Ciona intestinalis inflammatory response. Cell Tissue Res 329:79–90
Plagemann PGW (2005) Epitope specificity of monoclonal antibodies to the N-protein of porcine reproductive and respiratory syndrome virus by ELISA with synthetic peptides. Vet Immunol Immunophatol 104:50–68
Raftos DA, Tait NN, Briscoe DA (1987a) Allograft rejection and alloimmune memory in the solitary urochordate, Styela plicata. Dev Comp Immunol 11:343–351
Raftos DA, Tait NN, Briscoe DA (1987b) Cellular basis of allograft rejection in the solitary urochordate, Styela plicata. Dev Comp Immunol 11:713–725
Raftos DA, Briscoe DA, Tait NN (1988) The mode of recognition of allogeneic tissue in the solitary urochordate Styela plicata. Transplantation 45:1123–1126
Smith VJ, Peddie CM (1992) Cell cooperation during host defense in the solitary tunicate Ciona intestinalis (L). Biol Bull 183:211–219
Smith VJ, Söderhäll K (1991) A comparison of phenoloxidase activity in the blood of marine invertebrates. Dev Comp Immunol 15:251–261
Söderhäll K, Cerenius L (1998) Role of the prophenoloxidase-activating system in invertebrate immunity. Curr Opin Immunol 10:23–28
Sugumaran M (1996) Roles of the insect cuticle in host defence reactions. In: Söderhäll K, Iwanaga S, Vasta GR (eds) New directions in invertebrates immunology. SOS, Fair Haven, pp 355–374
Vizzini A, Pergolizzi M, Vazzana M, Salerno G, Di Sano C, Macaluso P, Arizza V, Parrinello D, Cammarata M, Parrinello N (2008) FACIT collagen (1 alpha-chain) is expressed by hemocytes and epidermis during the inflammatory response of the ascidian Ciona intestinalis. Dev Comp Immunol 32:682–692
Winder J, Harris H (1991) New assays for tyrosine hydroxylase and dopa oxidase activities of tyrosinase. Eur J Biochem 198:317–326
Acknowledgements
Our thanks are due to Mr. G. Miceli for collecting ascidians.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the Italian MIUR (PRIN 2004 and 2006).
Rights and permissions
About this article
Cite this article
Cammarata, M., Arizza, V., Cianciolo, C. et al. The prophenoloxidase system is activated during the tunic inflammatory reaction of Ciona intestinalis . Cell Tissue Res 333, 481–492 (2008). https://doi.org/10.1007/s00441-008-0649-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-008-0649-x