Abstract
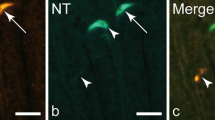
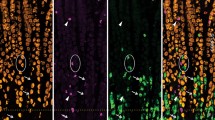
Both ghrelin and obestatin are derived from preproghrelin by post-translational processing. We have morphologically characterized the cells that produce obestatin and ghrelin in new-born and adult Sprague-Dawley rats that were freely fed, fasted, or subjected to gastric bypass surgery or reserpine treatment. Tissue samples collected from the gastrointestinal tract and pancreas were examined by double-immunofluorescence staining, immunoelectron microscopy, and conventional electron microscopy. Obestatin was present in the stomach, duodenum, jejunum, colon, and pancreas. In the stomach, differences were noted in the development of obestatin- and preproghrelin-immunreactive (IR) cells on the one hand and ghrelin-IR cells on the other, particularly 2 weeks after birth. Preproghrelin- and obestatin-IR cells were more numerous than ghrelin-IR cells in the stomach, suggesting the lack of ghrelin in some A-like cells. Most obestatin-producing cells in the stomach were distributed in the basal part of the oxyntic mucosa; these cells co-localized with chromogranin A (pancreastatin) and vesicle monoamine transporters type 1 and 2, but not with serotonin or histidine decarboxylase. Immunoelectron microscopy revealed the obestatin- and ghrelin-producing cells to be A-like cells, characterized by numerous highly electron-dense granules containing ghrelin and obestatin. Some granules exhibited an even electron density with thin electron-lucent halos, suggestive of monoamines. Feeding status, gastric bypass surgery, and reserpine treatment had no obvious effect on the A-like cells. In the pancreas, obestatin was present in the peripheral part of the islets, with a distribution distinct from that of glucagon-producing A cells, insulin-producing beta cells, and cells producing pancreatic polypeptide Y. Thus, obestatin and ghrelin co-localize with an anticipated monoamine in A-like cells in the stomach, and obestatin is found in pancreatic islets.









Similar content being viewed by others
References
Alumets J, Sundler F, Håkanson R (1977) Distribution, ontogeny and ultrastructure of somatostatin immunoreactive cells in the pancreas and gut. Cell Tissue Res 185:465–479
Andersson K, Zhao CM, Chen D, Sundler F, Håkanson R (1996) Effect of alpha-fluoromethylhistidine-evoked histamine depletion on ultrastructure of endocrine cells in acid-producing mucosa of stomach in mouse, rat and hamster. Cell Tissue Res 286:375–384
Bassil AK, Haglund Y, Brown J, Rudholm T, Hellstrom PM, Naslund E, Lee K, Sanger GJ (2007) Little or no ability of obestatin to interact with ghrelin or modify motility in the rat gastrointestinal tract. Br J Pharmacol 150:58–64
Björkqvist M, Dornoville de la Cour C, Zhao CM, Gagnemo-Persson R, Håkanson R, Norlén P (2002) Role of gastrin in the development of gastric mucosa. ECL cells and A-like cells in newborn and young rats. Regul Pept 108:73–82
Bresciani E, Rapetti D, Dona F, Bulgarelli I, Tamiazzo L, Locatelli V, Torsello A (2006) Obestatin inhibits feeding but does not modulate GH and corticosterone secretion in the rat. J Endocrinol Invest 29:RC16–RC18
Canese MG, Bussolati G (1977) Immuno-electron-cytochemical localization of the somatostatin cells in the human antral mucosa. J Histochem Cytochem 25:1111–1118
Carlini VP, Schioth HB, Debarioglio SR (2007) Obestatin improves memory performance and causes anxiolytic effects in rats. Biochem Biophys Res Commun 352:907–912
Chanoine JP, Wong AC (2004) Ghrelin gene expression is markedly higher in fetal pancreas compared to fetal stomach: effect of maternal fasting. Endocrinology 145:3813–3820
Chanoine JP, Wong AC, Barrios V (2006) Obestatin, acylated and total ghrelin concentrations in the perinatal rat pancreas. Horm Res 66:81–88
Chartrel N, Alvear-Perez R, Leprince J, Iturrioz X, Reaux-Le Goazigo A, Audinot V, Chomarat P, Coge F, Nosjean O, Rodriguez M, Galizzi JP, Boutin JA, Vaudry H, Llorens-Cortes C (2007) Comment on “Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin’s effects on food intake”. Science 315:766
Chen D, Monstein HJ, Nylander AG, Zhao CM, Sundler F, Håkanson R (1994) Acute responses of rat stomach enterochromaffinlike cells to gastrin: secretory activation and adaptation. Gastroenterology 107:18–27
Chen D, Zhao CM, Norlén P, Björkqvist M, Ding XQ, Kitano M, Håkanson R (2000) Effect of cholecystokinin-2 receptor blockade on rat stomach ECL cells. A histochemical, electron-microscopic and chemical study. Cell Tissue Res 299:81–95
Cummings DE (2006) Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol Behav 89:71–84
Date Y, Kojima M, Hosoda H, Sawaguchi A, Mondal MS, Suganuma T, Matsukura S, Kangawa K, Nakazato M (2000) Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 141:4255–4261
Date Y, Nakazato M, Hashiguchi S, Dezaki K, Mondal MS, Hosoda H, Kojima M, Kangawa K, Arima T, Matsuo H, Yada T, Matsukura S (2002) Ghrelin is present in pancreatic alpha-cells of humans and rats and stimulates insulin secretion. Diabetes 51:124–129
Dornonville de la Cour C, Björkqvist M, Sandvik AK, Bakke I, Zhao CM, Chen D, Håkanson R (2001) A-like cells in the rat stomach contain ghrelin and do not operate under gastrin control. Regul Pept 99:141–150
Dun SL, Brailoiu GC, Brailoiu E, Yang J, Chang JK, Dun NJ (2006) Distribution and biological activity of obestatin in the rat. J Endocrinol 191:481–489
Egido EM, Rodriguez-Gallardo J, Silvestre RA, Marco J (2002) Inhibitory effect of ghrelin on insulin and pancreatic somatostatin secretion. Eur J Endocrinol 146:241–244
Erickson JD, Schafer MK, Bonner TI, Eiden LE, Weihe E (1996) Distinct pharmacological properties and distribution in neurons and endocrine cells of two isoforms of the human vesicular monoamine transporter. Proc Natl Acad Sci USA 93:5166–5171
Fischer-Colbrie R, Lassmann H, Hagn C, Winkler H (1985) Immunological studies on the distribution of chromogranin A and B in endocrine and nervous tissues. Neuroscience 16:547–555
Furnes WM, Stenström B, Tømmerås K, Skoglund T, Dickson SL, Kulseng B, Zhao CM, Chen D (2007) Feeding behaviour in rats subjected to gastrectomy or gastric bypass surgery. Eur Surg Res (in press)
Gil-Campos M, Aguilera CM, Canete R, Gil A (2006) Ghrelin: a hormone regulating food intake and energy homeostasis. Br J Nutr 96:201–226
Gourcerol G, Million M, Adelson DW, Wang Y, Wang L, Rivier J, St-Pierre DH, Taché Y (2006) Lack of interaction between peripheral injection of CCK and obestatin in the regulation of gastric satiety signaling in rodents. Peptides 27:2811–2819
Gourcerol G, St-Pierre DH, Taché Y (2007) Lack of obestatin effects on food intake: should obestatin be renamed ghrelin-associated peptide (GAP)? Regul Pept 141:1–7
Granata R, Settanni F, Biancone L, Trovato L, Nano R, Bertuzzi F, Destefanis S, Annunziata M, Martinetti M, Catapano F, Ghe C, Isgaard J, Papotti M, Ghigo E, Muccioli G (2007) Acylated and unacylated ghrelin promote proliferation and inhibit apoptosis of pancreatic beta-cells and human islets: involvement of 3′,5′-cyclic adenosine monophosphate/protein kinase A, extracellular signal-regulated kinase 1/2, and phosphatidyl inositol 3-kinase/Akt signaling. Endocrinology 148:512–529
Green BD, Irwin N, Flatt PR (2007) Direct and indirect effects of obestatin peptides on food intake and the regulation of glucose homeostasis and insulin secretion in mice. Peptides 28:981–987
Gualillo O, Lago F, Casanueva FF, Dieguez C (2006) One ancestor, several peptides: posttranslational modifications of preproghrelin generate several peptides with antithetical effects. Mol Cell Endocrinol 256:1–8
Håkanson R, Owman C (1967) Concomitant histochemical demonstration of histamine and catecholamines in enterochromaffin-like cells of gastric mucosa. Life Sci 6:759–766
Håkanson R, Böttcher G, Ekblad E, Panula P, Simonsson M, Dohlsten M, Hallberg T, Sundler F (1986) Histamine in endocrine cells in the stomach. A survey of several species using a panel of histamine antibodies. Histochemistry 86:5–17
Håkanson R, Chen D, Sundler F (1994) The ECL cells. In: Johnson LR (ed) Physiology of the gastrointestinal tract, vol 2, 3rd edn. Raven Press, New York, pp 1171–1184
Hashimoto H, Fujihara H, Kawasaki M, Saito T, Shibata M, Otsubo H, Takei Y, Ueta Y (2007) Centrally and peripherally administered ghrelin potently inhibits water intake in rats. Endocrinology 148:1638–1647
Hawkins KL, Lloyd RV, Toy KA (1989) Immunohistochemical localization of chromogranin A in normal tissues from laboratory animals. Vet Pathol 26:488–498
Holst B, Egerod KL, Schild E, Vickers SP, Cheetham S, Gerlach LO, Storjohann L, Stidsen CE, Jones R, Beck-Sickinger AG, Schwartz TW (2007) GPR39 signaling is stimulated by zinc ions but not by obestatin. Endocrinology 148:13–20
Inokuchi H, Kawai K, Takeuchi Y, Sano Y (1982) Immunohistochemical demonstration of EC cells in rat gastrointestinal tract. Histochemistry 74:453–456
Kojima M, Kangawa K (2006) Drug insight: the functions of ghrelin and its potential as a multitherapeutic hormone. Nat Clin Pract Endocrinol Metab 2:80–88
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402:656–660
Lagaud GJ, Young A, Acena A, Morton MF, Barrett TD, Shankley NP (2007) Obestatin reduces food intake and suppresses body weight gain in rodents. Biochem Biophys Res Commun 357:264–269
Lauwers E, Landuyt B, Arckens L, Schoofs L, Luyten W (2006) Obestatin does not activate orphan G protein-coupled receptor GPR39. Biochem Biophys Res Commun 351:21–25
Liu Y, Edwards R (1997) The role of vesicular transport proteins in synaptic transmission and neural degeneration. Annu Rev Neurosci 20:125–156
Pearse AGE (1969) The cytochemistry and ultrastructure of polypeptide hormone-producing cells of the APUD series and the embryologic, physiologic and pathologic implications of the concept. J Histochem Cytochem 17:303–313
Pearse AG, Bussolati G (1972) The identification of gastrin cells as G cells. Virchows Arch [A] Pathol Pathol Anat 355:99–104
Prado CL, Pugh-Bernard AE, Elghazi L, Sosa-Pineda B, Sussel L (2004) Ghrelin cells replace insulin-producing beta cells in two mouse models of pancreas development. Proc Natl Acad Sci USA 101:2924–2929
Nogueiras R, Pfluger P, Tovar S, Arnold M, Mitchell S, Morris A, Perez-Tilve D, Vazquez MJ, Wiedmer P, Castaneda TR, DiMarchi R, Tschop M, Schurmann A, Joost HG, Williams LM, Langhans W, Dieguez C (2007) Effects of obestatin on energy balance and growth hormone secretion in rodents. Endocrinology 148:21–26
Norlén P, Curry WJ, Chen D, Zhao CM, Johnston CF, Håkanson R (1997) Expression of the chromogranin A-derived peptides pancreastatin and WE14 in rat stomach ECL cells. Regul Pept 70:121–133
Rindi G, Necchi V, Savio A, Torsello A, Zoli M, Locatelli V, Raimondo F, Cocchi D, Solcia E (2002) Characterisation of gastric ghrelin cells in man and other mammals: studies in adult and fetal tissues. Histochem Cell Biol 117:511–519
Sakata I, Nakamura K, Yamazaki M, Matsubara M, Hayashi Y, Kangawa K, Sakai T (2002) Ghrelin-producing cells exist as two types of cells, closed- and opened-type cells, in the rat gastrointestinal tract. Peptides 23:531–536
Samson WK, White MM, Price C, Ferguson AV (2007) Obestatin acts in brain to inhibit thirst. Am J Physiol Regul Integr Comp Physiol 292:R637–643
Schuldiner S, Shirvan A, Linial M (1995) Vesicular neurotransmitter transporters: from bacteria to humans. Physiological Rev 75:369–392
Stenström B, Zhao CM, Tømmerås K, Arum CJ, Chen D (2006a) Is gastrin partially responsible for body weight reduction after gastric bypass? Eur Surg Res 38:94–101
Stenström B, Furnes MW, Tømmerås K, Syversen U, Zhao CM, Chen D (2006b) Mechanism of gastric bypass-induced body weight loss: one-year follow-up after micro-gastric bypass in rats. J Gastrointest Surg 10:1384–1391
Sundler F, Håkanson R (1991) Gastric endocrine cell typing at the light microscopic level. In: Håkanson R, Sundler F (eds) The stomach as an endocrine organ. Elsevier, Amsterdam, pp 9–26
Torbergsen K, Wiksen H, Johansen K, Rahimipoor S, Falkmer UG, Zhao CM (2005) Immunoreactivity of gastric ECL and A-like cells in fasted and fed rats and mice. Biotech Histochem 80:21–30
Urbé S, Dittié A, Tooze SA (1997) pH-dependent processing of secretogranin II by the endopeptidase PC2 in isolated immature secretory granules. Biochem J 321:65–74
Van de Ven WJ, Roebroek AJ, Van Duijnhoven HL (1993) Structure and function of eukaryotic proprotein processing enzymes of the subtilisin family of serine proteases. Crit Rev Oncog 4:115–136
Volante M, Allia E, Gugliotta P, Funaro A, Broglio F, Deghenghi R, Muccioli G, Ghigo E, Papotti M (2002) Expression of ghrelin and of the GH secretagogue receptor by pancreatic islet cells and related endocrine tumors. J Clin Endocrinol Metab 87:1300–1308
Weihe E, Eiden LE (2000) Chemical neuroanatomy of the vesicular amine transporters. FASEB J 14:2435–2449
Wierup N, Sundler F (2005) Ultrastructure of islet ghrelin cells in the human fetus. Cell Tissue Res 319:423–428
Wierup N, Svensson H, Mulder H, Sundler F (2002) The ghrelin cell: a novel developmentally regulated islet cell in the human pancreas. Regul Pept 107:63-69
Wierup N, Yang S, McEvilly RJ, Mulder H, Sundler F (2004) Ghrelin is expressed in a novel endocrine cell type in developing rat islets and inhibits insulin secretion from INS-1 (832/13) cells. J Histochem Cytochem 52:301–310
Yabuki A, Ojima T, Kojima M, Nishi Y, Mifune H, Matsumoto M, Kamimura R, Masuyama T, Suzuki S (2004) Characterization and species differences in gastric ghrelin cells from mice, rats and hamsters. J Anat 205:239–246
Zhang JV, Ren P-G, Avsian-Kretchmer O, Luo C-W, Rauch R, Klein C, Hsueh AJW (2005) Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin’s effects on food intake. Science 310:996–999
Zhao CM, Chen D, Lintunen M, Panula P, Håkanson R (1999a) Effects of reserpine on ECL-cell ultrastructure and histamine compartmentalization in the rat stomach. Cell Tissue Res 295:131–140
Zhao CM, Chen D, Lintunen M, Panula P, Håkanson R (1999b) Secretory organelles in ECL cells of the rat stomach: an immunohistochemical and electron-microscopic study. Cell Tissue Res 298:457–470
Zhao CM, Bakke I, Tostrup-Skogaker N, Waldum HL, Håkanson R, Chen D (2001) Functionally impaired, hypertrophic ECL cells accumulate vacuoles and lipofuscin bodies. An ultrastructural study of ECL cells isolated from hypergastrinemic rats. Cell Tissue Res 303:415–422
Zhou A, Martin S, Lipkind G, LaMendola J, Steiner DF (1998) Regulatory roles of the P domain of the subtilisin-like prohormone convertases. J Biol Chem 273:11107–11114
Zhu X, Cao Y, Voogd K, Steiner DF (2006) On the processing of proghrelin to ghrelin. J Biol Chem 281:38867–38870
Zizzari P, Longchamps R, Epelbaum J, Bluet-Pajot MT (2007) Obestatin partially affects ghrelin stimulation of food intake and GH secretion in rodents. Endocrinology 148:1648–1653
Acknowledgements
We thank Trine Skoglund at the Department of Cancer Research and Molecular Medicine, Norwegian University of Science and Technology, Trondheim, Norway, for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by a grant from the Cancer Foundation of St. Olav’s Hospital, Trondheim, Norway.
Rights and permissions
About this article
Cite this article
Zhao, CM., Furnes, M.W., Stenström, B. et al. Characterization of obestatin- and ghrelin-producing cells in the gastrointestinal tract and pancreas of rats: an immunohistochemical and electron-microscopic study. Cell Tissue Res 331, 575–587 (2008). https://doi.org/10.1007/s00441-007-0514-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-007-0514-3