Abstract
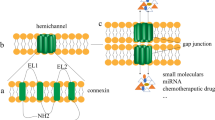
Gap junctions are considered to play a crucial role in differentiation of epithelial cells, including hepatocytes. Recently, we found that Cx32 but not Cx26 was closely related to tight junctional proteins in primary cultured rat hepatocytes (Kojima et al., Exp Cell Res 263:193–201, 2001) and that Cx32 formation and/or Cx32-mediated intercellular communication could induce expression and function of tight junctions in a mouse hepatic cell line (Kojima et al., Exp Cell Res 276:40–51, 2002). In this study, to investigate the mechanisms of induction of tight junctions by transfection with Cx32, we performed cDNA microarray analysis of Cx32 transfectants, compared with parental cells derived from Cx32-deficient hepatocytes. In cDNA microarray analysis, a 2.5-fold increase in expression of membrane-associated guanylate kinase with inverted orientation-1 (MAGI-1), which is known to be localized at adherens and tight junction regions, was observed. High expression of MAGI-1 in Cx32 transfectants was confirmed by Western blotting and RT-PCR. MAGI-1 was colocalized with occludin, claudin-2, ZO-1, and F-actin, but not with E-cadherin in the apical-most regions at cell borders of Cx32 transfectants, similar to junctional adhesion molecule-1 (JAM-1), which may play a crucial role in formation and assembly of tight junctions. Treatment with the gap junction blocker 18β-glycyrrhetinic acid did not affect expression of MAGI-1 and JAM-1 in Cx32 transfectants. These results suggest that Cx32 expression is in part related to induction of tight junctions through modulation of MAGI-1 expression in an immortalized mouse hepatic cell line.



Similar content being viewed by others
References
Decaens C, Rodriguez P, Bouchaud C, Cassio D (1996) Establishment of hepatic cell polarity in the rat hepatoma-human fibroblast hybrid WIF-B9: a biphasic phenomenon going from a simple epithelial polarized phenotype to an hepatic polarized one. J Cell Sci 109:1623–1635
Dobrosotskaya IY (2001) Identification of mNET1 as a candidate ligand for the first PDZ domain of MAGI-1. Biochem Biophys Res Commun 283:969–975
Dobrosotskaya IY, James GL (2000) MAGI-1 interacts with beta-catenin and is associated with cell–cell adhesion structures. Biochem Biophys Res Commun 270:903–909
Dobrosotskaya I, Guy RK, James GL (1997) MAGI-1, a membrane-associated guanylate kinase with a unique arrangement of protein–protein interaction domains. J Biol Chem 272:31589–31597
Ebnet K, Schulz CU, Meyer zu Brickwedde MK, Pendl GG, Vestweber D (2000) Junctional adhesion molecule interacts with the PDZ domain-containing proteins AF-6 and ZO-1. J Biol Chem 275:27979–27988
Ebnet K, Suzuki A, Horikoshi Y, Hirose T, Meyer zu Brickwedde MK, Ohno S, Vestweber D (2001) The cell polarity protein ASIP/PAR-3 directly associates with junctional adhesion molecule (JAM). EMBO J 20:3738–3748
Elias PM, Friend DS (1976) Vitamin-A-induced mucous metaplasia: an in vitro system for modulating tight and gap junction differentiation. J Cell Biol 68:173–188
Fukuhara A, Irie K, Nakanishi H, Takekuni K, Kawakatsu T, Ikeda W, Yamada A, Katata T, Honda T, Sato T, Shimizu K, Ozaki H, Horiuchi H, Kita T, Takai Y (2002) Involvement of nectin in the localization of junctional adhesion molecule at tight junctions. Oncogene 21:7642–7655
Giepmans BN (2004) Gap junctions and connexin-interacting proteins. Cardiovasc Res 62:233–245
Gumbiner BM (1993) Breaking through the tight junction barrier. J Cell Biol 123:1631–1633
Hirabayashi S, Tajima M, Yao I, Nishimura W, Mori H, Hata Y (2003) JAM4, a junctional cell adhesion molecule interacting with a tight junction protein, MAGI-1. Mol Cell Biol 23:4267–4282
Hirao K, Hata Y, Ide N, Takeuchi M, Irie M, Yao I, Deguchi M, Toyoda A, Sudhof TC, Takai Y (1998) A novel multiple PDZ domain-containing molecule interacting with N-methyl-d-aspartate receptors and neuronal cell adhesion proteins. J Biol Chem 273:21105–21110
Ide N, Hata Y, Nishioka H, Hirao K, Yao I, Deguchi M, Mizoguchi A, Nishimori H, Tokino T, Nakamura Y, Takai Y (1999) Localization of membrane-associated guanylate kinase (MAGI)-1/BAI-associated protein (BAP) 1 at tight junctions of epithelial cells. Oncogene 18:7810–7815
Itoh M, Sasaki H, Furuse M, Ozaki H, Kita T, Tsukita S (2001) Junctional adhesion molecule (JAM) binds to PAR-3: a possible mechanism for the recruitment of PAR-3 to tight junctions. J Cell Biol 154:491–497
Kojima T, Sawada N, Chiba H, Kokai Y, Yamamoto M, Urban M, Lee GH, Hertzberg EL, Mochizuki Y, Spray DC (1999) Induction of tight junctions in human connexin 32 (hCx32)-transfected mouse hepatocytes: connexin 32 interacts with occludin. Biochem Biophys Res Commun 266:222–229
Kojima T, Sawada N, Duffy HS, Spray DC (2001a) Gap and tight junctions in liver: composition, regulation and function. In: Arias IM, Fausto N, Chisari FV, Shafritz DA, Boyer JL (eds) The liver: biology and pathobiology, vol. 4. Raven, New York, pp 29–46
Kojima T, Kokai Y, Chiba H, Yamamoto M, Mochizuki Y, Sawada N (2001b) Cx32 but not Cx26 is associated with tight junctions in primary cultures of rat hepatocytes. Exp Cell Res 263:193–201
Kojima T, Spray DC, Kokai Y, Chiba H, Mochizuki Y, Sawada N (2002) Cx32 formation and/or Cx32-mediated intercellular communication induces expression and function of tight junctions in hepatocytic cell line. Exp Cell Res 276:40–51
Kumar NM, Gilula NB (1996) The gap junction communication channel. Cell 84:381–388
Lin D, Edwards AS, Fawcett JP, Mbamalu G, Scott JD, Pawson T (2000) A mammalian PAR-3-PAR-6 complex implicated in Cdc42/Rac1 and aPKC signalling and cell polarity. Nat Cell Biol 2:540–547
Loewenstein WR (1979) Junctional intercellular communication and the control of growth. Biochim Biophys Acta 560:1–65
Martin-Padura I, Lostaglio S, Schneemann M, Williams L, Romano M, Fruscella P, Panzeri C, Stoppacciaro A, Ruco L, Villa A, Simmons D, Dejana E (1998) Junctional adhesion molecule, a novel member of the immunoglobulin superfamily that distributes at intercellular junctions and modulates monocyte transmigration. J Cell Biol 142:117–127
Mino A, Ohtsuka T, Inoue E, Takai Y (2000) Membrane-associated guanylate kinase with inverted orientation (MAGI)-1/brain angiogenesis inhibitor 1-associated protein (BAP1) as a scaffolding molecule for Rap small G protein GDP/GTP exchange protein at tight junctions. Genes Cells 5:1009–1016
Nishimura W, Iizuka T, Hirabayashi S, Tanaka N, Hata Y (2000) Localization of BAI-associated protein1/membrane-associated guanylate kinase-1 at adherens junctions in normal rat kidney cells: polarized targeting mediated by the carboxyl-terminal PDZ domains. J Cell Physiol 185:358–365
Ozaki H, Ishii K, Horiuchi H, Arai H, Kawamoto T, Okawa K, Iwamatsu A, Kita T (1999) Cutting edge: combined treatment of TNF-alpha and IFN-gamma causes redistribution of junctional adhesion molecule in human endothelial cells. J Immunol 163:553–557
Patrie KM, Drescher AJ, Goyal M, Wiggins RC, Margolis B (2001) The membrane-associated guanylate kinase protein MAGI-1 binds megalin and is present in glomerular podocytes. J Am Soc Nephrol 12:667–677
Patrie KM, Drescher AJ, Welihinda A, Mundel P, Margolis B (2002) Interaction of two actin-binding proteins, synaptopodin and α-actinin-4, with the tight junction protein MAGI-1. J Biol Chem 277:30183–30190
Saez JC, Spray DC, Nairn AC, Hertzberg E, Greengard P, Bennett MV (1986) cAMP increases junctional conductance and stimulates phosphorylation of the 27-kDa principal gap junction polypeptide. Proc Natl Acad Sci U S A 83:2473–2477
Schneeberger EE, Lynch RD (2004) The tight junction: a multifunctional complex. Am J Physiol Cell Physiol 286:C1213–C1228
Toyofuku T, Yabuki M, Otsu K, Kuzuya T, Hori M, Tada M (1998) Direct association of the gap junction protein connexin-43 with ZO-1 in cardiac myocytes. J Biol Chem 273:12725–12731
Trosko JE, Ruch RJ (1998) Cell–cell communication in carcinogenesis. Front Biosci 3:D208–D236
Tsukita S, Furuse M (2002) Claudin-based barrier in simple and stratified cellular sheets. Curr Opin Cell Biol.14:531–536
Tsukita S, Furuse M, Itoh M (2001) Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol 2:285–293
Yamasaki H, Naus CC (1996) Role of connexin genes in growth control. Carcinogenesis 17:1199–1213
Acknowledgements
We are grateful to Dr Y. Hata (Tokyo Medical and Dental University) for the MAGI-1 antibody and Dr T. Kita (Kyoto University) for the JAM-1 antibody.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported in by Grants-in-Aid from the Ministry of Education, Culture, Sports, Science Technology and the Ministry of Health, Labour and Welfare of Japan, and by the Kato Memorial Bioscience Foundation, the Uehara Memorial Foundation, the Suhara Memorial Foundation, the Smoking Research Foundation, and the Long-Range Research Initiative Project of the Japan Chemical Industry Association.
Rights and permissions
About this article
Cite this article
Murata, M., Kojima, T., Yamamoto, T. et al. Tight junction protein MAGI-1 is up-regulated by transfection with connexin 32 in an immortalized mouse hepatic cell line: cDNA microarray analysis. Cell Tissue Res 319, 341–347 (2005). https://doi.org/10.1007/s00441-004-1017-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-004-1017-0