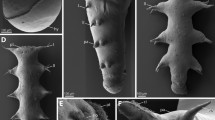
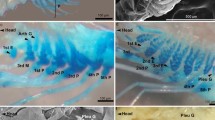
Abstract
The ontogeny of the antennal glands was studied during the embryonic and post-embryonic development of Astacus leptodactylus. The future glands arising from undifferentiated columnar cells were detectable at the metanauplius stage EI 150 μm (EI: eye index; approximately 440 μm at hatching). The tubule and labyrinth differentiated in embryos at EI 190 μm, and the bladder and coelomosac at EI 250 μm. At EI 350 μm, the tubule lengthened and divided into proximal and distal sub-regions. In later stages, the gland retained the same morpho-anatomy but the differentiation and size of each part increased. The cells of the coelomosac displayed the cytological features of podocytes in late embryonic development at EI 440 μm. Only small apical microvilli and a few mitochondria were observable in the labyrinth cells at EI 250 μm; by EI 440 μm, these cells presented well-shaped apical microvilli, formed bodies, basal infoldings and mitochondria. In the cells of the tubules and bladder, mitochondria and basal infoldings occurred at EI 440 μm and EI 250 μm, respectively. The differentiation of the tubules and bladder cells suggested that they were involved in active transport at EI 440 μm. Following hatching, the differentiation of the cells and the size of the glands increased. The ontogeny of the antennal glands thus starts in early embryos, the specific cellular functional features being differentiated in the various parts of the glands by EI 440 μm. The antennal glands are probably functional just before hatching, i.e., before the juveniles are confronted with the low osmolality of freshwater.






Similar content being viewed by others
References
Aladin NV, Potts WTW (1995) Osmoregulatory capacity of the Cladocera. J Comp Physiol [B] 164:671–683
Anderson E, Beams HW (1956) Light and electron microscopic studies on the cells of the labyrinth in the “green gland” of Cambarus sp. Iowa Acad Sci 63:681–685
Anger K (2001) The biology of the decapod crustacean larvae. Balkema, Lisse, pp 1–420
Barradas C, Dunel-Erb S, Lignon J, Péqueux A (1999a) Superimposed morphofunctional study of ion regulation and respiration in single gill filaments of the crayfish Astacus leptodactylus. J Crust Biol 19:14–25
Barradas C, Wilson JM, Dunel-Erb S (1999b) Na+,K+-ATPase activity and immunocytochemical labelling in podoranchial filament and lamina of the freshwater crayfish Astacus leptodactylus Eschscholtz: evidence for the existence of sodium transport in the filaments. Tissue Cell 31:523–528
Beams HW, Anderson E, Press N (1956) Light and electron microscopic studies on the cells of the distal portion of the crayfish nephron tubule. Cytologia 21:50–57
Bielawski J (1964) Chloride transport and water intake into isolated gills of crayfish. Comp Biochem Physiol 13:423–432
Charmantier G, Charmantier-Daures M (1994) Ontogeny of osmoregulation and salinity tolerance in the isopod crustacean Sphaeroma serratum. Mar Ecol Prog Ser 114:93–102
Charmantier G, Christophe H, Lignot JH, Charmantier-Daures M (2001) Ecophysiological adaptation to salinity throughout a life cycle: a review in homarid lobsters. J Exp Biol 204:967–977
Drach P, Tchernigovtzeff C (1967) Sur la méthode de détermination des stades d’intermue et son application générale aux crustacés. Vie Milieu 18:595–610
Dunel-Erb S, Barradas C, Lignon J (1997) Morphological evidence for the existence of two distinct types of mitochondria rich cells in the gills of the crayfish Astacus leptodactylus Eschscholtz. Acta Zool 78:195–203
Edwards JG (1928) Studies on aglomerular and glomerular kidneys. Am J Anat 42:75
Fuller EG, Highison GJ, Brown F, Bayer C (1989) Ultrastructure of the crayfish antennal gland revealed by scanning and transmission electron microscopy combined with ultrasonic microdissection. J Morphol 200:9–15
Holdich DM, Horlioglu MM, Firkins I (1997) Salinity adaptations of crayfish in British waters with particular reference to Austropotamobius pallipes, Astacus leptodactylus and Pacifastacus leniusculus. Estuar Coast Shelf Sci 44:147–154
Holliday CW, Miller DS (1984) Cellular mechanisms of organic anion transport in crustacean renal tissue. Am Zool 24:275–284
Hubschman JH (1971) Transient larval glands in Palamonetes. In: Crisp DJ (ed) Fourth European Marine Biology Symposium. Cambridge University Press, New York, pp 295–300
Ischiguro J (1975) Fine structure of the antennal gland of the crayfish Procambarus clarkii. J Electron Microsc 24:191
Johnson PT (1980). Histology of the blue crab Callinectes sapidus. Praeger, New York, pp 1–440
Kamemoto FI, Ono JK (1968) Urine flow determination by continuous collection in the crayfish Procambarus clarkii. Comp Biochem Physiol 27:851–857
Khodabandeh S, Kutnik M, Aujoulat F, Charmantier G, Charmantier-Daures M (2004) Ontogeny of the antennal glands in the crayfish, Astacus leptodactylus (Crustacea, Decapoda): immunolocalization of Na+,K+-ATPase. Cell Tissue Res DOI 10.1007/s00441-004-0970-y
Kirschner LB, Wagner S (1965) The site and permeability of the filtration locus in the crayfish antennal gland. J Exp Biol 43:385–395
Kümmel G (1964) Morphological indication of a filtration process in the antennal gland of Cambarus affinis. Naturwissenschaften 51:200
Lopez J, Cuesta N, Martinez A, Montuenga L, Cuttitta F (1999) Proadrenomedullin N-terminal 20 peptide (PAMP) immunoreactivity in vertebrate juxtaglomerular granular cells identified by both light and electron microscopy. Gen Comp Endocrinol 116:192–203
Luft JH (1961) Improvements in epoxy resin embedding materials. J Biophys Biochem Cytol 9:409–414
Mantel LH, Farmer LL (1983) Osmotic and ionic regulation. In: Bliss DE (ed) The biology of Crustacea. Internal anatomy and physiological regulations. Academic Press, London, pp 53–161
Martoja R, Martoja-Pierson M (1967) Initiation aux techniques de l’histologie animale. Masson et Cie, Paris, p 345
Miyawaki M, Ukeshima A (1967) On the ultrastructure of the antennal gland epithelium of the crayfish, Procambarus clarkii. Kumamoto J Sci 8:59–73
Miyawaki M, Ura T (1969) Absorption and secretion of experimentally injected protein silver by kidney cells of the crayfish. Ann Zool Jpn 42:56–62
Morrit D, Spicer JI (1995) Changes in the pattern of osmoregulation in the brackish water amphipod Gammarus duebeni Lilljeborg (Crustacea) during embryonic development. J Exp Zool 273:271–281
Morrit D, Spicer JI (1996a) Developmental ecophysiology of the beachflea Orchestia gammarellus (Pallas) (Crustacea: Amphipoda). I. Female control of the embryonic environment. J Exp Mar Biol Ecol 207:191–203
Morrit D, Spicer JI (1996b) Developmental ecophysiology of the beachflea Orchestia gammarellus (Pallas) (Crustacea: Amphipoda). Embryonic osmoregulation. J Exp Mar Biol Ecol 207:205–216
Morrit D, Spicer JI (1999) Developmental ecophysiology of the beachflea Orchestia gammarellus (Pallas) (Crustacea: Amphipoda: Talitridae) III. Physiological competency as a possible explanation for timing of hatchling release. J Exp Mar Biol Ecol 232:275–283
Péqueux A (1995) Osmotic regulation in crustaceans. J Crust Biol 15:1–60
Perkins HC (1972) Developmental rates at various temperatures of embryos of the northern lobster (Homarus americanus Milne-Edwards). Fish Bull 70:95–99
Peterson DR, Loizzi RF (1973) Regional cytology and cytochemistry of the crayfish kidney tubule. J Morphol 141:133–146
Peterson DR, Loizzi RF (1974) Ultrastructure of the crayfish kidney, coelomosac, labyrinth, and nephridial canal. J Morphol 142:241–264
Riegel JA (1963) Micropuncture studies of chloride concentrations and osmotic pressure in the crayfish antennal gland. J Exp Biol 40:487–492
Riegel JA (1966) Analysis of formed bodies in urine removed from the crayfish antennal gland by micropuncture. J Exp Biol 44:387–395
Riegel JA (1970) A new model of transepithelial fluid movement with detailed application to fluid movement in the crayfish antennal gland. Comp Biochem Physiol 36:403–410
Riegel JA (1977) Fluid movement through the crayfish antennal gland. In: Gupta BL, Moreton RB, Osshman JL, Wall BJ (eds) Transport of ions and water in animals. Academic Press, London, pp 613–631
Riegel JA, Cook MA (1975) Recent studies of excretion in crustaceans. Fortschr Zool 23:48–75
Riegel JA, Kirschner LB (1960) The excretion of inulin and glucose by the crayfish antennal gland. Biol Bull 118:296–307
Schaffner A, Rodewald R (1978) Filtration barriers in the coelomic sac of the crayfish, Procambarus clarkii. J Ultrastruct Res 65:36–47
Sesma P, Bayona C, Villaro AC, Vazquez JJ (1983) A microscopic study on the antennal gland of Antrapotamobius ballines (Crustacea, Decapoda). Morfol Normal Patologica 7:289–301
Shivers RR, Chauvin WJ (1977) Intercellular junctions of antennal gland epithelial cells in the crayfish Orconectes virilis. Cell Tissue Res 175:425
Susanto GN (2000) Adaptation des écrevisses à l’eau douce: ontogenèse de l’osmorégulation chez Astacus leptodactylus. Ph.D thesis. University of Montpellier 2, France, pp 1–129
Susanto GN, Charmantier G (2000) Ontogeny of osmoregulation in the crayfish Astacus leptodactylus. Physiol Biochem Zool 73:169–176
Susanto GN, Charmantier G (2001) Crayfish freshwater adaptation starts in eggs: ontogeny of osmoregulation in embryos of Astacus leptodactylus. J Exp Zool 289:433–440
Trask T (1974) Laboratory-reared larvae of Cancer anthonyi (Decapoda: Brachyura) with a brief description of the internal anatomy of the megalopa. Mar Biol 27:63–74
Wheatly MG, Gannon TA (1995) Ion regulation in crayfish: freshwater adaptations and the problem of molting. Am Zool 35:49–59
Yamada E (1955) The fine structure of the renal glomerulus of the mouse. J Biophys Biochem Cytol 1:551
Zehnder H (1934) Über die Embryonalentwicklung des Flusskrebses. Acta Zool 15:261–408
Acknowledgements
We thank Mr. F. Aujoulat, University of Montpellier II, for his technical help, and anonymous referees for helping to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Thanks are due to the University of Tarbiat Modarres and Ministry of Science, Research and Technology, Islamic Republic of Iran, for financial aid and support. Special thanks are also extended to the Société Française d’Exportation des Ressources Educatives (SFERE) for a scholarship to S.K.
Rights and permissions
About this article
Cite this article
Khodabandeh, S., Charmantier, G., Blasco, C. et al. Ontogeny of the antennal glands in the crayfish Astacus leptodactylus (Crustacea, Decapoda): anatomical and cell differentiation. Cell Tissue Res 319, 153–165 (2005). https://doi.org/10.1007/s00441-004-0982-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-004-0982-7