Abstract
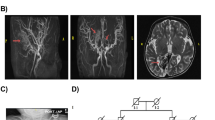
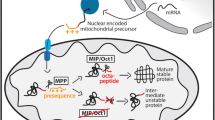
Glycosylphosphatidylinositol (GPI) functions to anchor certain proteins to the cell surface. Although defects in GPI biosynthesis can result in a wide range of phenotypes, most affected patients present with neurological abnormalities and their diseases are grouped as inherited-GPI deficiency disorders. We present two siblings with global developmental delay, brain anomalies, hypotonia, and contractures. Exome sequencing revealed a homozygous variant, NM_001035005.4:c.90dupC (p.Phe31Leufs*3) in C18orf32, a gene not previously associated with any disease in humans. The encoded protein is known to be important for GPI-inositol deacylation. Knockout of C18orf32 in HEK293 cells followed by a transfection rescue assay revealed that the PIPLC (Phosphatidylinositol-Specific Phospholipase C) sensitivity of GPI-APs (GPI-anchored proteins) was restored only by the wild type and not the mutant C18orf32. Immunofluorescence revealed that the mutant C18orf32 was localized to the endoplasmic reticulum and was also found as aggregates in the nucleus. In conclusion, we identified a pathogenic variant in C18orf32 as the cause of a novel autosomal recessive neurodevelopmental disorder with hypotonia and contractures. Our results demonstrate the importance of C18orf32 in the biosynthesis of GPI-anchors, the molecular impact of the variant on the protein function, and add a novel candidate gene to the existing repertoire of genes implicated in neurodevelopmental disorders.


Similar content being viewed by others
Availability of data and materials
Yes.
Code availability
Not applicable.
References
Bellai-Dussault K, Nguyen TTM, Baratang NV et al (2019) Clinical variability in inherited glycosylphosphatidylinositol deficiency disorders. Clin Genet 95:112–121. https://doi.org/10.1111/cge.13425
Bersuker K, Peterson CWH, To M et al (2018) A proximity labeling strategy provides insights into the composition and dynamics of lipid droplet proteomes. Dev Cell 44:97–112. https://doi.org/10.1016/j.devcel.2017.11.020
Carmody LC, Blau H, Danis D et al (2020) Significantly different clinical phenotypes associated with mutations in synthesis and transamidase+remodeling glycosylphosphatidylinositol (GPI)-anchor biosynthesis genes. Orphanet J Rare Dis 15:40. https://doi.org/10.1186/s13023-020-1313-0
Chen R, Walter EI, Parker G et al (1998) Mammalian glycophosphatidylinositol anchor transfer to proteins and posttransfer deacylation. Proc Natl Acad Sci USA 95:9512–9517. https://doi.org/10.1073/pnas.95.16.9512
Chiyonobu T, Inoue N, Morimoto M et al (2014) Glycosylphosphatidylinositol (GPI) anchor deficiency caused by mutations in PIGW is associated with West syndrome and hyperphosphatasia with mental retardation syndrome. J Med Genet 51:203–207. https://doi.org/10.1136/jmedgenet-2013-102156
Girisha KM, Shukla A, Trujillano D et al (2016) A homozygous nonsense variant in IFT52 is associated with a human skeletal ciliopathy. Clin Genet 90:536–539. https://doi.org/10.1111/cge.12762
Hansen L, Tawamie H, Murakami Y et al (2013) Hypomorphic mutations in PGAP2, encoding a GPI-anchor-remodeling protein, cause autosomal-recessive intellectual disability. Am J Hum Genet 92:575–583. https://doi.org/10.1016/j.ajhg.2013.03.008
Howard MF, Murakami Y, Pagnamenta AT et al (2014) Mutations in PGAP3 impair GPI-anchor maturation, causing a subtype of hyperphosphatasia with mental retardation. Am J Hum Genet 94:278–287. https://doi.org/10.1016/j.ajhg.2013.12.012
Ilkovski B, Pagnamenta AT, O’Grady GL et al (2015) Mutations in PIGY: expanding the phenotype of inherited glycosylphosphatidylinositol deficiencies. Hum Mol Genet 24:6146–6159. https://doi.org/10.1093/hmg/ddv331
Johnston JJ, Gropman AL, Sapp JC et al (2012) The phenotype of a germline mutation in PIGA: the gene somatically mutated in paroxysmal nocturnal hemoglobinuria. Am J Hum Genet 90:295–300. https://doi.org/10.1016/j.ajhg.2011.11.031
Kinoshita T (2016) Glycosylphosphatidylinositol (GPI) anchors: biochemistry and cell biology: introduction to a thematic review series. J Lipid Res 57:4–5. https://doi.org/10.1194/jlr.E065417
Kinoshita T, Fujita M (2016) Biosynthesis of GPI-anchored proteins: special emphasis on GPI lipid remodeling. J Lipid Res 57:6–24. https://doi.org/10.1194/jlr.R063313
Krawitz PM, Schweiger MR, Rödelsperger C et al (2010) Identity-by-descent filtering of exome sequence data identifies PIGV mutations in hyperphosphatasia mental retardation syndrome. Nat Genet 42:827–829. https://doi.org/10.1038/ng.653
Krawitz PM, Murakami Y, Hecht J et al (2012) Mutations in PIGO, a member of the GPI-anchor-synthesis pathway, cause hyperphosphatasia with mental retardation. Am J Hum Genet 91:146–151. https://doi.org/10.1016/j.ajhg.2012.05.004
Kvarnung M, Nilsson D, Lindstrand A et al (2013) A novel intellectual disability syndrome caused by GPI anchor deficiency due to homozygous mutations in PIGT. J Med Genet 50:521–528. https://doi.org/10.1136/jmedgenet-2013-101654
Liu Y-S, Guo X-Y, Hirata T et al (2018) N-Glycan-dependent protein folding and endoplasmic reticulum retention regulate GPI-anchor processing. J Cell Biol 217:585–599. https://doi.org/10.1083/jcb.201706135
Maydan G, Noyman I, Har-Zahav A et al (2011) Multiple congenital anomalies-hypotonia-seizures syndrome is caused by a mutation in PIGN. J Med Genet 48:383–389. https://doi.org/10.1136/jmg.2010.087114
Murakami Y, Siripanyapinyo U, Hong Y et al (2003) PIG-W is critical for inositol acylation but not for flipping of glycosylphosphatidylinositol-anchor. Mol Biol Cell 14:4285–4295. https://doi.org/10.1091/mbc.e03-03-0193
Murakami Y, Kanzawa N, Saito K et al (2012) Mechanism for release of alkaline phosphatase caused by glycosylphosphatidylinositol deficiency in patients with hyperphosphatasia mental retardation syndrome. J Biol Chem 287:6318–6325. https://doi.org/10.1074/jbc.M111.331090
Tanaka S, Maeda Y, Tashima Y, Kinoshita T (2004) Inositol deacylation of glycosylphosphatidylinositol-anchored proteins is mediated by mammalian PGAP1 and yeast Bst1p. J Biol Chem 279:14256–14263. https://doi.org/10.1074/jbc.M313755200
UniProt Consortium (2015) UniProt: a hub for protein information. Nucleic Acids Res 43:D204-212. https://doi.org/10.1093/nar/gku989
Acknowledgements
The authors are grateful to the family for cooperating in this study.
Funding
This work was supported by grants-in-aid from the National Natural Science Foundation of China 31770853 (MF). ‘Clinical and molecular characterization of leukodystrophies in Indian children’ (V.25011/379/2015-GIA/HR) was funded by Department of Health Research, Government of India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that the authors do not have any conflict of interest and all have read and approved the final manuscript.
Ethics approval
This study was approved by the CHU Sainte-Justine institutional review board.
Consent to participate
Written informed consent was taken.
Consent for publication
Consent for publication has been obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salian, S., Guo, XY., Murakami, Y. et al. C18orf32 loss-of-function is associated with a neurodevelopmental disorder with hypotonia and contractures. Hum Genet 141, 1423–1429 (2022). https://doi.org/10.1007/s00439-022-02433-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-022-02433-0